Abstract
Identifying coals suitable for blast furnace injection has become increasingly important due to rising injection rates. This review of traditional pulverised coal reactivity testing equipment reveals that no agreed-upon evaluation standard exists and that different reactor types are employed for testing. Therefore, reference blast furnace conversion conditions are defined, followed by a discussion of their influence on the coal conversion process as illustrated by conceptual conversion models. Critical process parameters are temperature, heating rate and pressure, while other effects can be calibrated. Evaluating the currently employed test equipment with regard to these process parameters shows that only specially designed drop-tube furnaces and flow reactors provide conversion conditions near to blast furnace conditions. For consistent injection coal testing, special reactors complying with the previously defined critical process parameters must be established.
Introduction
Injecting pulverised coal (PC) into blast furnace raceway zones has become a popular approach to reduce metallurgic coke consumption. The best available techniques (BAT) document for the European Union's iron and steel industry [1] qualifies this direct injection of hydrocarbons as a promising technology to increase efficiency and reduce greenhouse gas emissions from blast furnaces. All forms of hydrocarbons reduce the metallurgic coke consumption and the gross energy consumption, e.g. by 3.6% at PC injection rates of 180 kg/t
Metallurgic coke and PC compete for a limited amount of oxygen; therefore, high conversion rates and high PC consumption reduce the metallurgic coke consumption in the vicinity of the tuyeres. The disadvantage of direct injection is a decrease in the raceway temperature, which must be compensated by feeding O drying, devolatilisation, volatiles combustion, gasification, and burnout.


The initial step of the coal conversion process involves drying and is followed by devolatilisation. During devolatilisation, volatile compounds degas from the solid carbon matrix within the coal particle. The volatiles diffuse to the particle surface and are eventually transferred to the bulk gas phase, where they ignite if the bulk temperature exceeds their auto-ignition temperature and sufficient O




Mass transfer of the gaseous reactant from the bulk gas phase to the external solid fuel surface. Intra-particle processes: mass diffusion of the gaseous reactant from the external surface to the actual reaction site; adsorption of the gaseous reactant to the reaction site; chemical reaction of the adsorbed gaseous reactant and the solid; desorption of the gaseous product from the reaction site; mass diffusion of the gaseous product from the reaction site to the external surface. Mass transfer of the gaseous product from the external surface to the bulk gas phase.
Gasification and burnout steps are critical for the overall conversion time of coal particles [3], and both can be limited either by mass transfer from the gas phase to the reaction site or the chemical kinetic. Recent results suggest that even the coal preparation process, e.g. milling or grinding, affects the coal conversion process by changing surface chemistry and mineral phases [5].
In order to obtain comprehensive evaluation results of PCs and their applicability to blast furnaces, the employed test equipment should reproduce the fundamental features of the pulverised coal injection (PCI) process. Currently, PC performance in blast furnaces is evaluated in diverse equipment or test reactors. However, aside from Li et al. [6, 7], limited literature questions the applicability to reproduce blast furnace conditions. Because current knowledge indicates that coal conversion is decisively influenced by conversion conditions [2, 3, 8–14], evaluating coals under different operation conditions is assumed to significantly alter the outcome. Coals evaluated as suitable may cause problems when applied to the blast furnace and suitable coals might be rejected because of misleading evaluation results.
This work aims to identify the potential key parameters to reproduce raceway zone conditions in test equipment. First, representative raceway conditions are discussed and, subsequently, the coal conversion process and its dependence on the surrounding conditions is evaluated. Next, the currently employed testing equipment is discussed and its applicability to reproduce raceway conditions is evaluated.
Defining raceway conditions
Figure 1 shows representative modelling results for the thermo-chemical coal conversion within the raceway zone for temperatures (a), heating rates (b), velocities (c), and particle conversion characteristics (d) versus particle residence time [16, 17]. Hot blast and coal temperatures are around 1250 and 200

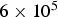
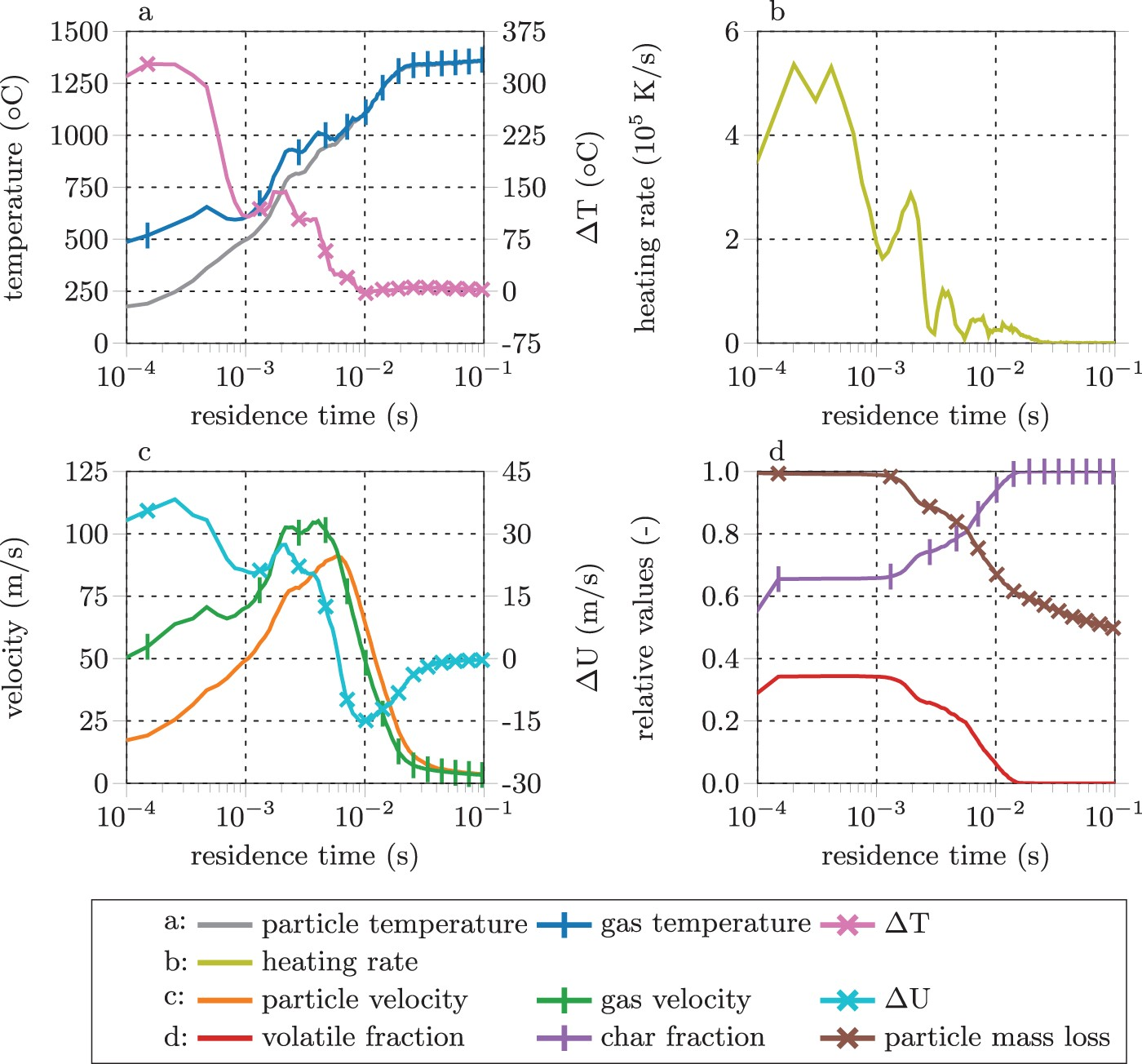
Dependences of PC conversion
To isolate the effects of main operating parameters (temperature, pressure), heating rates, fluid–solid interface, and particle size on PC conversion, a theoretical analysis first describes well-established mechanistic modelling concepts to outline the dependences on coal conversion rates accompanied by literature challenging the analysis. This analysis is followed by a presentation of the key parameters for reproducing blast furnace operating conditions.
Temperature
Temperature directly affects the intrinsic reaction rate of hetero- and homogeneous reactions, which are typically modelled using the Arrhenius form. Pressure and concentration effects are incorporated by functional expressions [22]: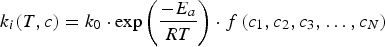
The parameters 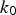
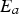
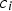
Rumpel [23, 24] proposes a conceptual model for the gas–solid conversion of porous solids based on the contribution of diffusion sub-processes: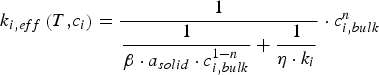
Intrinsic rates are significantly affected by temperature. The first term in the denominator on the right-hand side of Equation (2) represents the contribution of the boundary layer diffusion from the bulk to the particle surface, while the second term represents the contribution of the pore diffusion and the intrinsic reaction rate. β is the mass transfer coefficient between the surrounding and particle, 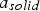
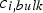
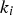
Mehrabian et al. [26] propose a conversion model for dense particles, assuming infinitesimal reaction layers moving from the solid surface to its centre, based on the layer approach proposed by Thunman et al. [27]: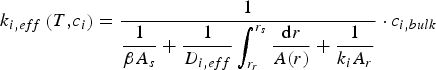
In this approach, A and r represent the area and the radial position of the surface s and the reaction layer r, respectively. The terms in the denominator denote the boundary layer diffusion rate, the pore diffusion rate and the actual reaction rate, respectively. In contrast to the model proposed by Rumpel [23, 24], Mehrabian et al. explicitly consider pore diffusion from the surface to the reaction side.
Diffusion within solid particles is controlled by molecular and/or Knudsen diffusion, depending on the ratio between the mean free molecule path (λ) and the characteristic length scale (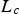
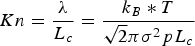
Here, 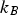
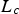
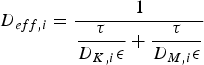
Both diffusivities are corrected by the ratio of particle porosity (ε) and particle tortuosity (τ) to incorporate pore effects on diffusion rates [28]. Knudsen diffusion is a function of the pore size (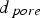
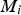
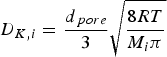
The molecular diffusion coefficient of specie i in the gas mixture (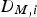
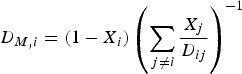
In this mixture rule, X denotes the mole fraction of species i or j and 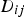
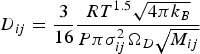
The binary diffusion coefficients depend on T, the temperature-dependent collision integral (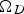
Summarising the above points, temperature affects gas–solid conversion rates by changing the intrinsic reaction rates and the diffusion rates. Reaction rates depend exponentially on temperatures (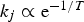
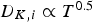
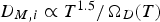
Since both mass transport from the bulk gas phase to the reaction site and the intrinsic conversion rates strongly depend on temperature, suitable experimental temperatures are necessary to obtain reliable results for the conversion behaviour of PC in blast furnaces.
Heating rate
PC heating rates range between 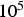
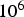
Fragmentation is reported to be caused either by thermal stresses due to intra-particle temperature gradients [11] or by internal pressure gradients from the volatiles emerging during devolatilisation [11, 13] or a combination of both. Literature indicates that vitrinite-rich coals are prone to fragmentation [9, 10, 12, 14] since vitrinite is one of the most brittle coal macerals [15]. Kim et al. [14] and Friedmann et al. [9, 10] investigate the fragmentation behaviour of different coal type particles, and both indicate a higher fragmentation probability for anthracite-like coals (high vitrinite content). Moreover, Friedmann et al. [9, 10] also indicate that fragmentation increases with increasing heating rates. Fragmentation due to thermal stresses significantly depends on the particle heating rates and the physical properties of the coal.
The probability of intra-particle gradients is characterised by the Biot number (Bi), which denotes the ratio between the external and internal heat transfer of the fuel particle [28]: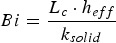
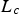
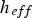
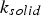
The external pyrolysis number (Py) compares the reaction and heat transfer time scales [31]: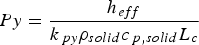
Here, 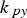
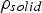
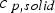
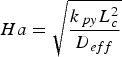
Ha is the Hatta number and 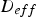
Pressure
Ambient pressure affects the species transport from the bulk fluid to the reaction site, the intrinsic reaction rate, the fragmentation behaviour, product yields, and morphology during thermo-chemical coal conversion [8, 11]. At higher pressures, binary diffusion (Equation (8)) and effective diffusion rates decrease within the fluid and particle pores (Equation (5)). Mass diffusion rates are inverse proportional to the pressure (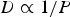
Intrinsic reaction rates depend on reactant species' partial pressures or concentrations, while adsorption rates to active surface reaction sites increase at higher partial pressures. The dependence of the intrinsic rate on the pressure is proportional to the partial pressure of the educt species (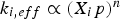
The effect of the intra-particle pressure on the fragmentation behaviour has been studied by Stanmore et al. [11], among others. By varying the ambient pressure, they show that intra-particle pressure gradients affect fragmentation probability. They ascribe this to a reduced differential pressure between the interior and surrounding at elevated pyrolysis pressures. However, Wall et al. [8] report a decrease in pyrolysis gases, with a simultaneous increase of solid yields at elevated pressures. The reason for the lower fragmentation probability might be a combination of both effects. Wall et al. also report pressure effects such as smaller residual ash particles and reduced conversion duration at elevated pressures. A more detailed discussion of these issues can be found elsewhere [8, 11].
Fluid–solid interface
Heat and mass transfer are dominated by the fluid surrounding the coal particles. Boundary layers for momentum (velocity/flow), heat and mass transfer emerge in the vicinity of particle surfaces [28, 32]. They develop according to fluid properties and the surrounding fluid flow situation. Boundary layers of these three transport phenomena are compared by dimensionless numbers, e.g. the Prandtl (Pr), the Schmidt (Sc), and the Lewis number (Le). Pr relates to the fluid dynamic and heat transfer boundary layer. If Pr<<1, the heat transfer layer is thicker than the fluid dynamic one. The Prandtl number relates the kinematic viscosity (ν) and the thermal diffusion rate (α):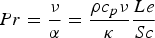
The thermal diffusion rate is based on physical gas properties, where ρ is the bulk density, 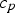
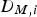
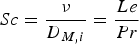
The Lewis number is the ratio of the thermal diffusivity and the mass diffusivity and relates the thermal and mass transport boundary layer thickness to each other: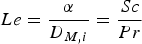
For ideal gases at blast furnace conditions, the characteristic quantities are Pr, Sc, and Re number versus temperature at blast furnace conditions. 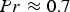
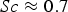
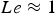
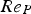
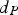
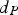
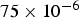
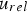
The convective and diffusive heat and mass transfer from the bulk to the particle surface are characterised by the Nusselt (Nu) and Sherwood (Sh) numbers, respectively. Both numbers are defined as the ratio of the actual transfer rate to the transfer rate in case of pure diffusion. They can be expressed by correlations of 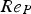
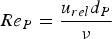
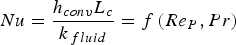
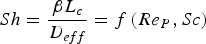
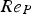
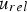
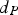
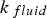
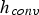
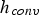
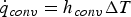
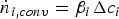
Since Pr and Sc numbers are constant at raceway conditions, Nu and Sh depend solely on 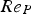
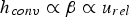

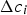
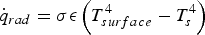
Here, σ is the Stefan–Boltzmann constant, ε is the emission coefficient, 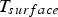
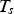
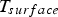
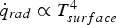
Particle size
Particle size has an influence on most of the above-discussed parameters. Effectiveness factors or pore diffusion from the particle surface to the reaction site depend on particle size (see Section ‘Temperature’). The characteristic length scale 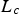
Summary conversion effects
As discussed in the previous subsections, the intense conditions within the blast furnace raceway zone significantly affect the coal conversion characteristics. The combination of high temperatures, high heating rates, and elevated pressure pose challenges for designing suitable test reactors. Evaluating the involved processes in PC conversion, in turn, enables the evaluation of the contribution to the overall conversion process. For clarity, effective reaction and diffusion rates as well as heat and mass transfer coefficients are shown for typical conditions. Figure 3 visualises the dependences of the effective reaction rate (Equation (2)) on temperature, pressure, and relative velocity between gas and solid. Increasing temperature, pressure and relative velocity promotes coal conversion rates. Effective reaction rate of a coal particle with O

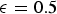
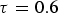
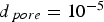
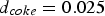
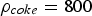
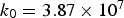
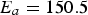
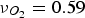
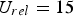
Figure 4 illustrates the effects of temperature and pressure on the diffusivity within porous coal particles. Effective O Effective diffusion coefficient of O


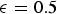
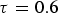
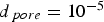
Furthermore, Figure 5 shows the relations between heat and mass transfer coefficient and temperature, pressure, particle size, and relative velocity. It indicates a decrease of heat transfer coefficients at higher pressures, while mass transfer coefficients increase. This contradictory behaviour is caused by the physical fluid properties. Relative velocity and particle size have a more pronounced effect on heat and mass transfer than temperature and pressure. Since both transfer coefficients have been derived by employing the analogy between heat and mass transfer, they show a similar dependence on particle size and velocity. The lowest specific transfer rates occur at low relative velocities for large particles. Decreasing particle sizes and increasing relative velocities improves transfer rates. Heat transfer coefficients (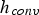
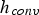

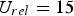
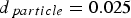
Overview of the discussed coherences between experimental conditions and coal conversion characteristics.
Test reactors should correctly reproduce the key operating parameters when screening coals for the application in the metallurgical industry. Based on the involved phenomena and the results of Table 3, the following parameter set has been identified: (blast) temperature, heating rate, pressure, particle size, gas–particle interface (boundary layer), and residence time.
Recommended design criteria are process temperature, heating rates, and pressure. Remaining close to the actual blast furnace operating conditions ensures that all important conversion characteristics are preserved. It is also recommended to employ similar particle size distributions, although they are subject to proper sample pre-processing. Gas–particle interface and residence time are assumed to be of minor importance when designing a test reactor. Keeping gas–particle relative velocities at the injection point is expensive in terms of consumables, while they equalise in a short time. Both residence times and gas–particle interface effects can be calibrated by comparison with known coals.
Current coal reactivity testing facilities and methods
This section outlines selected institutions active in the field of PC characterisation. Given this article's specific focus, the list is not exhaustive. In addition, institutions' employed testing methods are discussed and compared to the reference blast furnace operating conditions as defined in Section ‘Defining raceway conditions’. Subsequently, their applicability for PC injection characterisation is evaluated.
Overview testing facilities
Overview of selected institutions researching pulverised coal conversion (sorted by country).
a DTF, drop-tube furnace; FR, flow reactor; IR, injection rig; TGA, thermo gravimetric analysis; WR, wire-mesh reactor.
Five different reactor concepts are employed for the lab-scale evaluation of PCI coals by the discussed institutions:
drop-tube furnaces (DTF), flow reactors (FR), injection rigs (IR), thermo-gravimetric analysis (TGA), and wire-mesh reactors (WR).
Coal reactivity testing methods
The following subsection briefly restates the different reactor types employed for the PC classification of Table 4 and summarises the capabilities of the employed equipment.
Drop-tube furnace
Drop-tube furnaces (DTF) are essentially vertical pipes heated from the outside, and solid samples pass the reactor due to gravitational acceleration. Particle and gas phase samples are typically taken at the reactor outlet or in a vertical direction along the reactor for varying residence times. The sample heating rates in DTFs range from Schematic drop-tube furnace (DTF); 1, fuel feeder; 2, reactor; 3, heating elements; 4, sampling ports along reactor; 5, sampling at reactor end.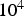

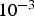
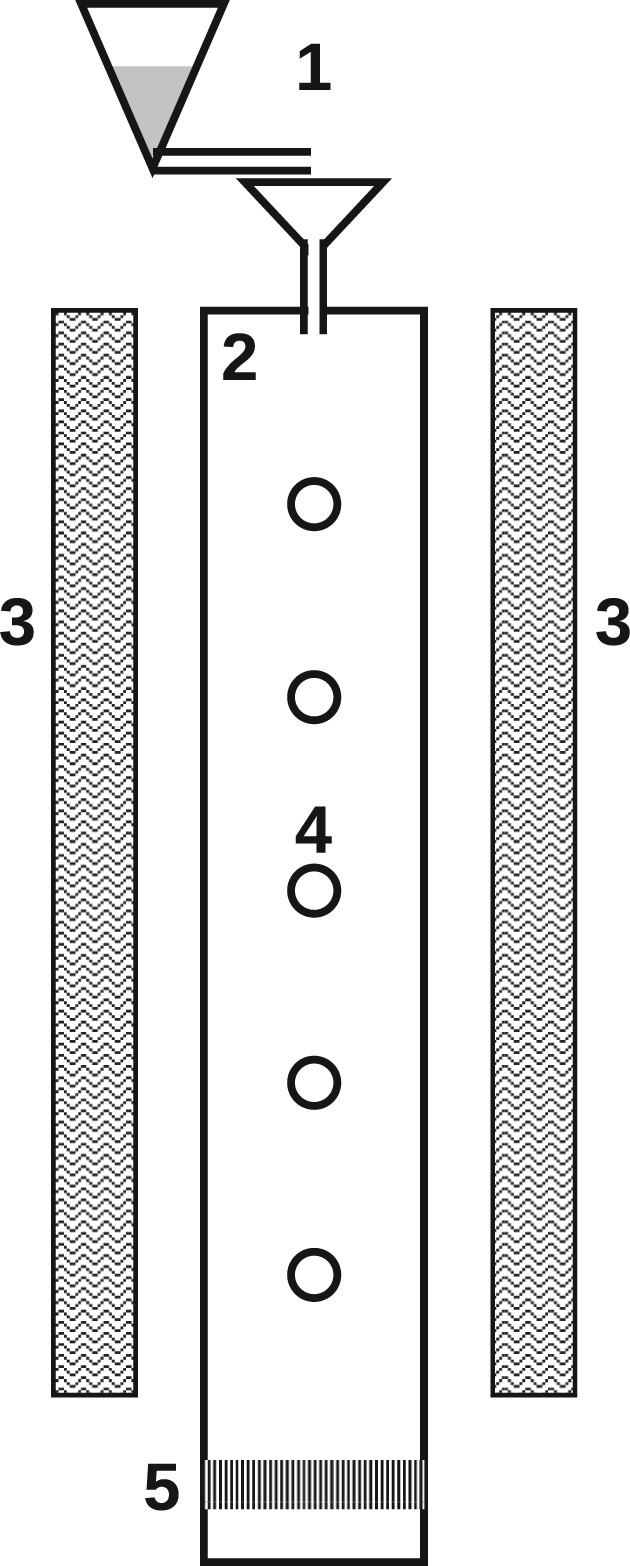
Flow reactor
Flow reactors (FR) are either vertically or horizontally aligned and fuel particles are pneumatically shot through the reactor. Temperature, pressure, and residence time ranges are similar to those of DTFs [14, 39]. However, particle heating rates within FRs are higher than in DTFs and reach values between Schematic flow reactor (FR); 1, fuel feeder; 2, gas co-flow; 3, reactor; 4, heating elements; 5, sampling ports along reactor; 6, sampling at reactor end.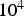
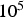
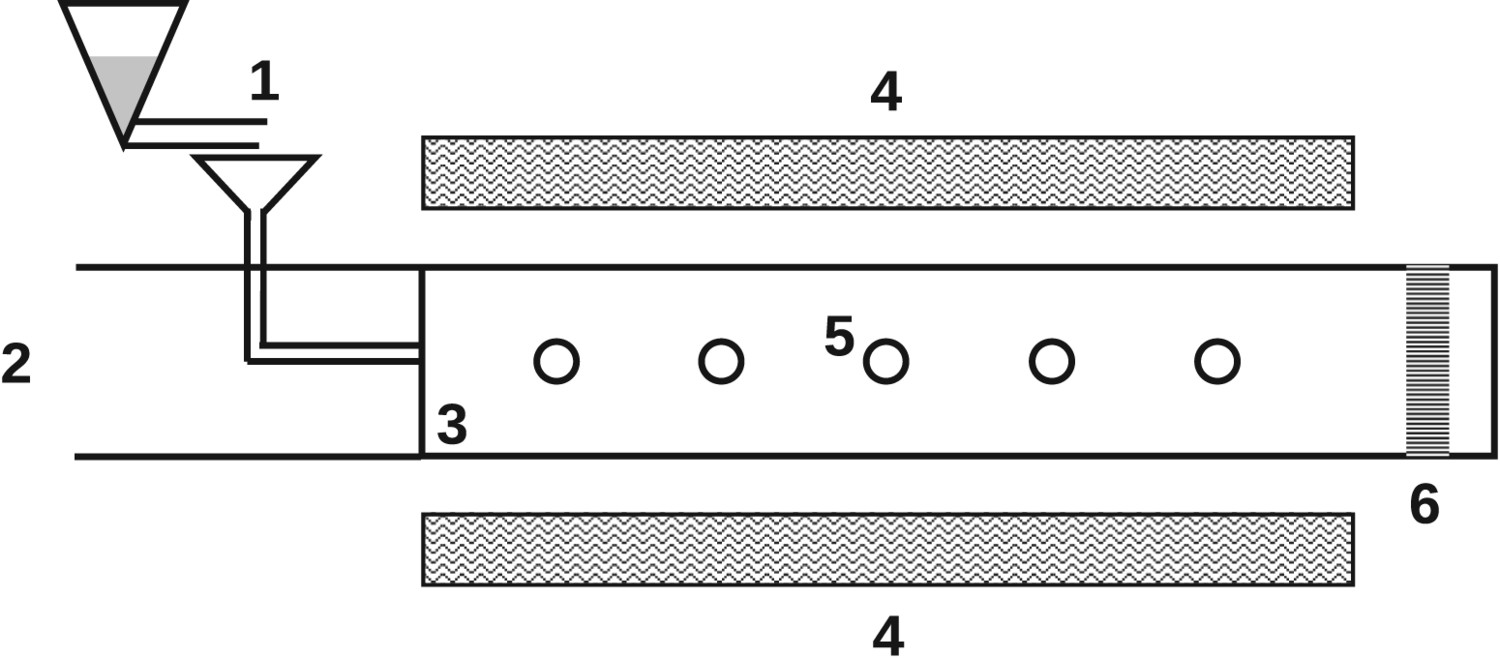
Injection rig
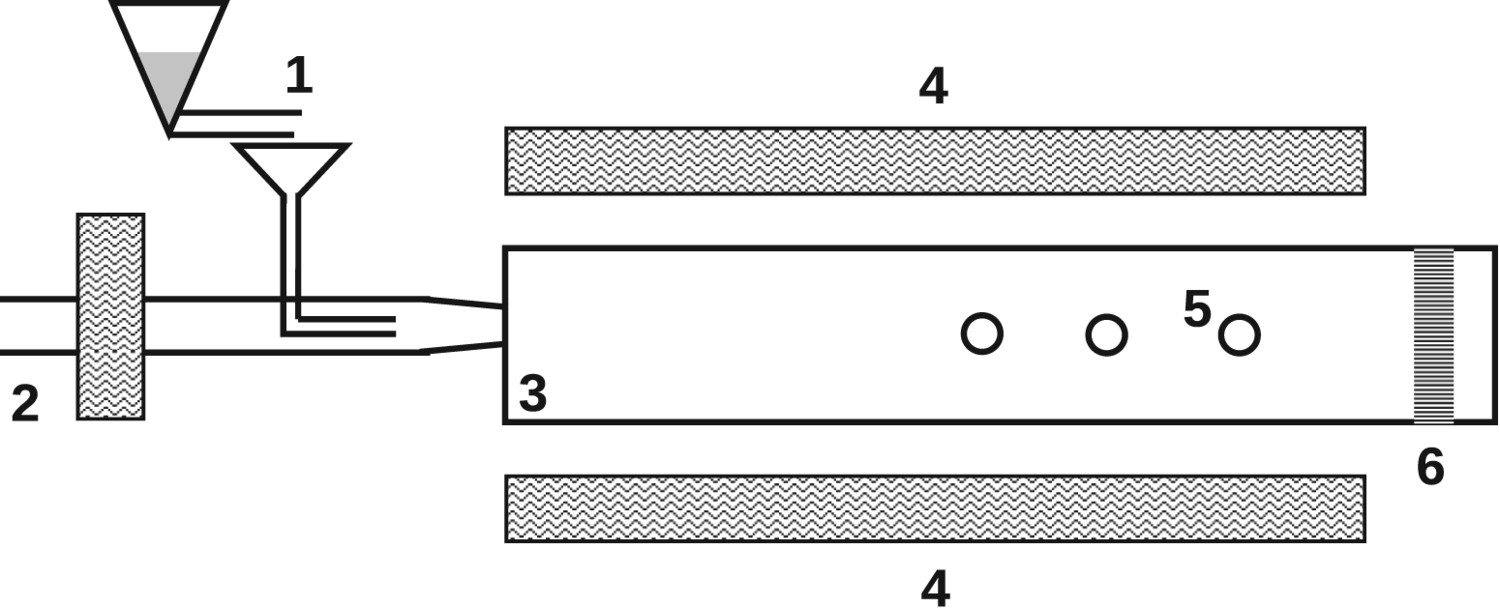
Injection rigs (IR) are similar to FRs; however, they use coal mass flows orders of magnitude higher than FRs (0.2–150 kg/h) [18, 38, 43, 65]. Consequently, gas flow rates must also be significantly increased compared to FRs (up to several hundreds of N m3/h [34, 35]). The high velocities within IRs result in residence times between 20 and 140 ms [18, 36]. PC particles are continuously injected into a pre-heated combustion chamber surrounded by a pre-heated co-flow. The relative velocity at the injection point is at the same order of magnitude as in PC combustion. Therefore, heating rates within IRs are in the order of Schematic injection rig (IR); 1, fuel feeder; 2, blast pre-heater; 3, combustion chamber; 4, heating elements; 5, sampling ports along reactor; 6, sampling at reactor end.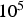
Special IRs are operated in batch mode, performing single sample shots into a pre-heated chamber [18, 41–45]. These batch rigs often require sample masses in the range of several milligrams (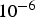
Thermo-gravimetric analysis
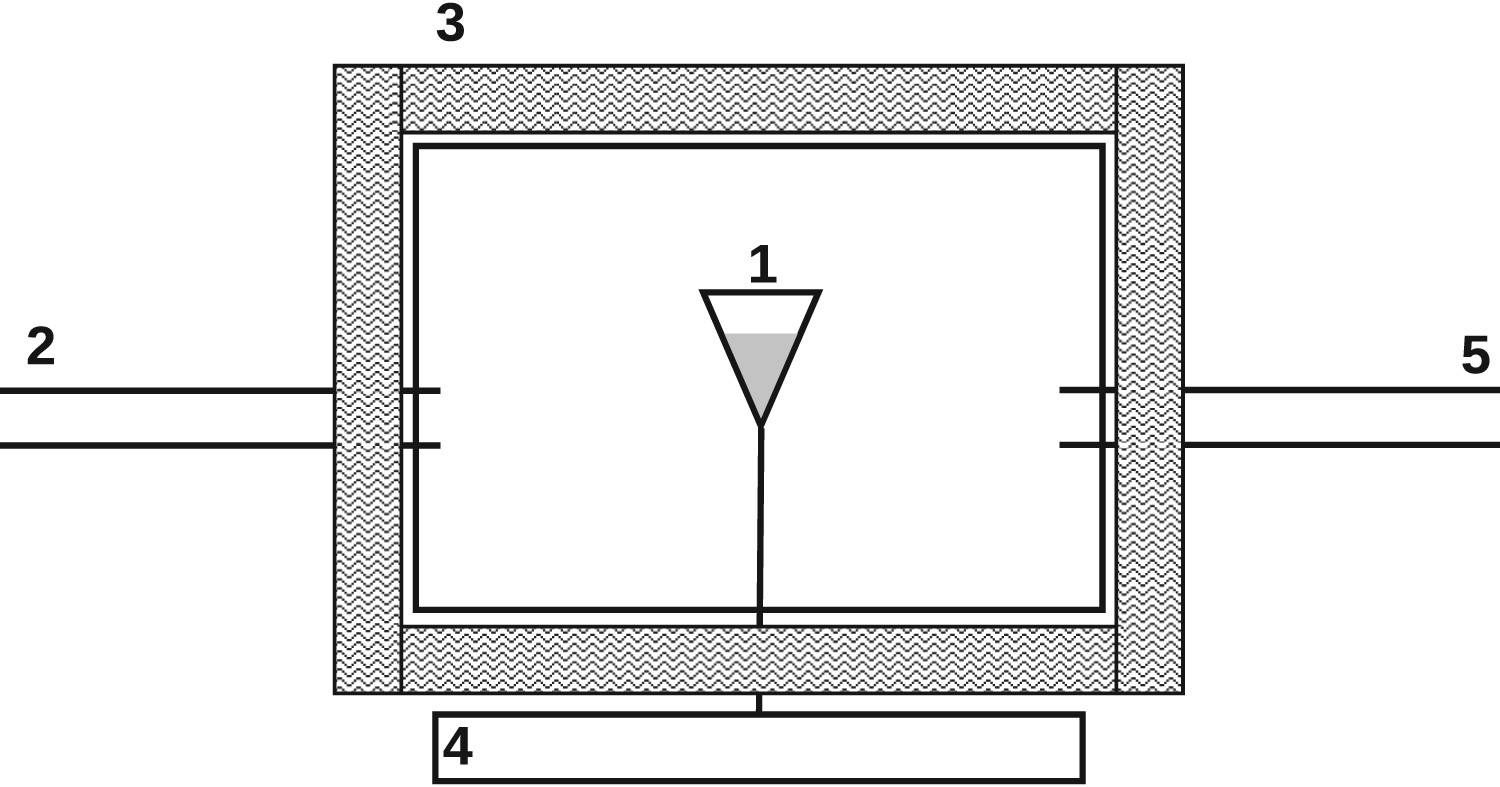
Thermo-gravimetric analysis (TGA) is a common, commercially available device, and is a default method for characterising gas–solid reaction kinetics or the characterisation of combustion and pyrolysis residues. Several specialised TGA appliances exist, including high-pressure TGAs or high-heating rate TGAs. Compared to the other employed reactor concepts, TGA heating rates are the lowest (ranging between 0.5 and Schematic thermo-gravimetric analysis (TGA); 1, fuel sample; 2, pre-defined reaction gas; 3, external heating; 4, balance; 5, gas sampling.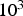

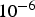
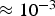
Wire-mesh reactor
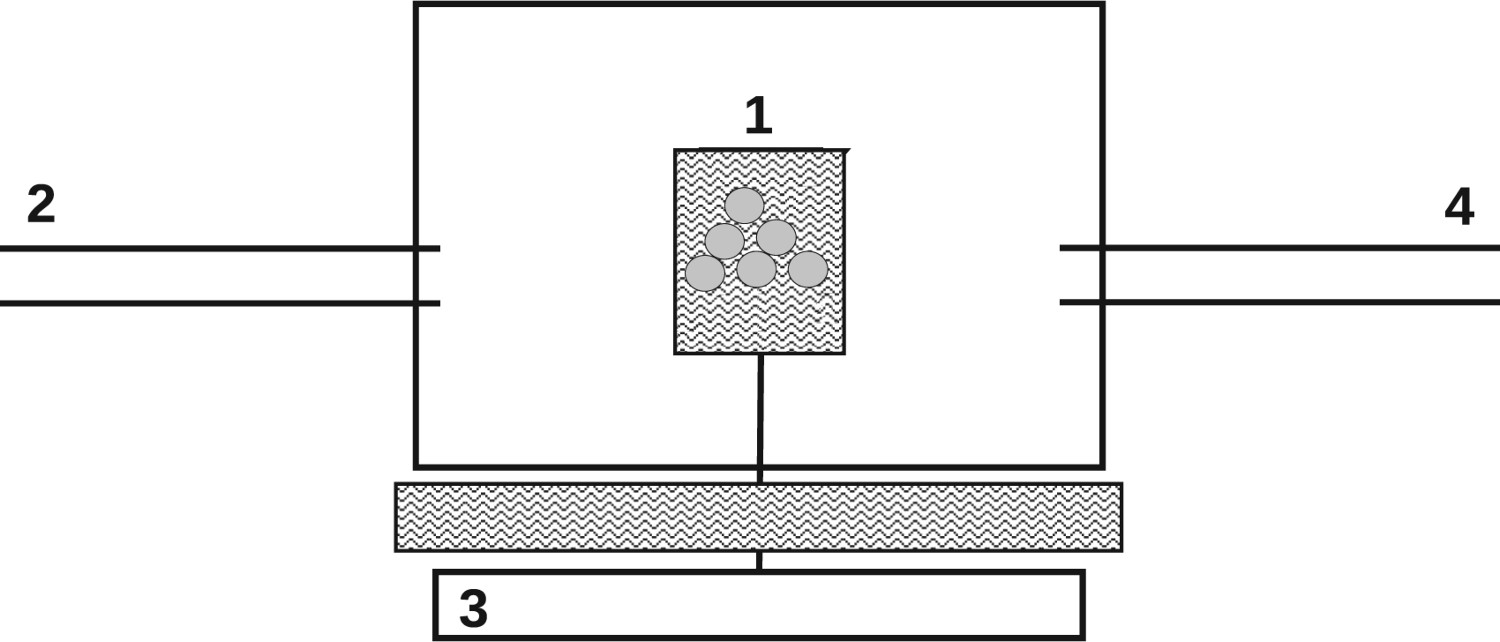
Sample holders in wire-mesh reactors (WR) are resistance-heated metal meshes. Therefore, the sample is fully perfused by the reaction gas. This configuration enables heating rates of around Schematic wire-mesh reactor (WR); 1, heated wire-mesh sample holder; 2, pre-defined reaction gas; 3, balance; 4, gas sampling.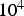

Comparison of characterisation methods
Properties of employed test reactor types in the pulverised coal characterisation under blast furnace conditions.


Advantages and disadvantages of the different characterisation methods for pulverised coal characterisation.

Summary and conclusion
Diverse reactor types are currently employed for evaluating PCI across the world. Special drop-tube furnaces (DTF) and flow reactors (FR) are capable of reproducing similar conversion conditions as in the raceway zone, while others fail to provide reasonable results, e.g. thermo-gravimetric analysis (TGA) and wire-mesh reactors (WR). Larger injection rigs (IR) can simulate raceway zone conditions at ambient pressures, thus these IRs disregard the importance of the operating pressure on coal conversion [8]. The number of specialised DTFs/FRs is limited and testing capacity is low. Comparing results from different test rigs is not recommended, as there is no agreed-upon, standardised procedure for testing PC and, moreover, the different testing equipment characteristics differ (see Section ‘Dependences of PC conversion’).
The preceding discusses key parameters to provide similar conversion conditions in test equipment, as supported by a conceptual analysis of the coal conversion process. These parameters were defined as the conversion temperature, employed heating rates, and pressure, while the particle boundary layer or ambient gas phase is of lesser importance and can be calibrated. A suitable reactor concept for PC testing under blast furnace conditions and a testing protocol will be developed based on these findings. This could contribute to more efficient evaluation of PCI coals in terms of their conversion behaviour and energy release under blast furnace conditions with the final goal of finding PCI coals with optimal performance. An increased PCI injection level during the blast furnace process would lead to decreased CO
The presented work partly contradicts the findings of Li et al. [6, 7], who postulated that DTFs can be used instead of IRs to characterise injection coals. Based on the thorough evaluation of the involved phenomena and additional literature, DTFs generally provide different results than IRs and, therefore, should not be employed for the evaluation of injection coals. The deviating operating conditions can significantly alter the evaluation results.
Footnotes
References




