Abstract
Melting and reduction of fine iron ore particles in the gas environment of a HIsarna smelting cyclone is a critically important topic, but very limited information is currently available except for some experimental data from high temperature drop tube furnace (HTDF). This work discusses the equilibrium state of reacting iron ore in the HTDF environment by thermodynamic calculations to strengthen the understanding of the HIsarna process. The limit of reduction termination of the ore particles was estimated in the calculation for the thermal decomposition and topochemical gas reduction. The theoretical calculation results are compared with the experimental data from the previous studies. Furthermore, variation of slag composition and iron valence states were estimated theoretically to understand the effects of post combustion ratio value and hydrogen/carbon ratio on the equilibrium state of the ore particles in the reducing gas.
Introduction
HIsarna is an emerging alternative ironmaking process aiming for up to 80% CO2 reduction by 2050 in combination with CO2 capture and storage (CCS) or usage (CCU). Without CCS/CCU, a reduction of up to 20% is feasible due to the removal of a number of pre-processing steps and less requirement on the quality of the raw materials in the HIsarna process [1,2]. HIsarna uses a smelting cyclone to melt and pre-reduce fine and un-agglomerated iron ore particles. Final reduction takes place in the smelting reduction vessel (SRV) directly beneath the smelt cyclone. Inside the smelting cyclone, the fine iron ore experiences very high temperatures. The ore particles are pre-reduced by around 20% through thermal decomposition and reduction in flight by the SRV off-gas [3]. The reduction kinetics of fine iron ore particles has been studied recently in a specially designed laboratory furnace, the high temperature drop tube furnace (HTDF) [3–7]. Meanwhile, the thermodynamics of pure haematite in the inert and reducing gas was investigated in their research. It shows that the thermal decomposition of haematite is much easier than magnetite because of more negative Gibbs energy change in haematite at the same temperature. Furthermore, it is suggested that the equilibrium state depends on the oxygen partial pressure [6]. The equilibrium reduction degree of haematite was also predicted by the Fe–O–C equilibrium diagram [8]. Here the reduction degree is defined as the ratio of lost oxygen of the ore to the initial oxygen content in the ore. The diagram suggests an increase of the reduction degree with the decreasing post combustion ratio (PCR) value of reducing gas. Qu et al. [4] also indicated the equilibrium state could only be reached in HTDF experiments at high temperature after reaction for 1 or 2 s. PCR of the gas is defined as follows: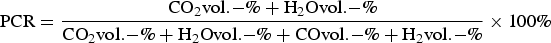
Thermodynamic calculation
Composition of haematite ore particles.
Gas composition.
Calculation results and discussion
As previously mentioned, the high temperature reaction of haematite ore contains thermal decomposition and topochemical gas reduction. The former reaction can proceed in inert gas, such as argon and nitrogen. After thermal decomposition, haematite forms sub-oxides and releases oxygen. Topochemical gas reduction is the interaction of gas and haematite ore, to reduce haematite and oxidise hydrogen or carbon monoxide in the gas. Usually, thermal decomposition is faster than topochemical gas reduction rate [4,6].
Thermal decomposition
First, to illustrate the importance of involving inert gas in the thermodynamic system, the equilibrium state of the haematite ore without inert gas is estimated within the temperature range of 1500–1900 K. Here, the initial state of the system was assumed to contain only solid ore, and the system in equilibrium state was under standard pressure. Figure 1 shows how the phase composition of the ore varies with increasing temperature. Haematite starts to decompose to oxygen and sub-oxides at 1630 K. At the same time, slag starts to form. The primary slag principally contains silica. The decomposition ratio is elevated rapidly around 1700 K. The formation of both magnetite–containing spinel and FeO containing slag is promoted at the same time. Magnetite is the most stable solid in the inert gas above 1630 K. However, it starts to transfer to molten slag above 1720 K. The slag mainly contains Fe2+ and Fe3+. A fully molten particle forms above 1833 K. The ratio of Fe3+ to Fe2+ in the slag increases with the increasing temperature, when the equilibrium state of ore transforms to fully molten slag. A slightly weight loss of slag at high temperature which is hard to be monitored is due to the release of oxygen to the surroundings. The calculation result suggests that there are also vaporisation of FeO(g) and other volatiles at high temperatures. But oxygen is still the principal component of the gas phase. Principal components of the haematite ore in equilibrium state at different temperatures without inert gas in the system.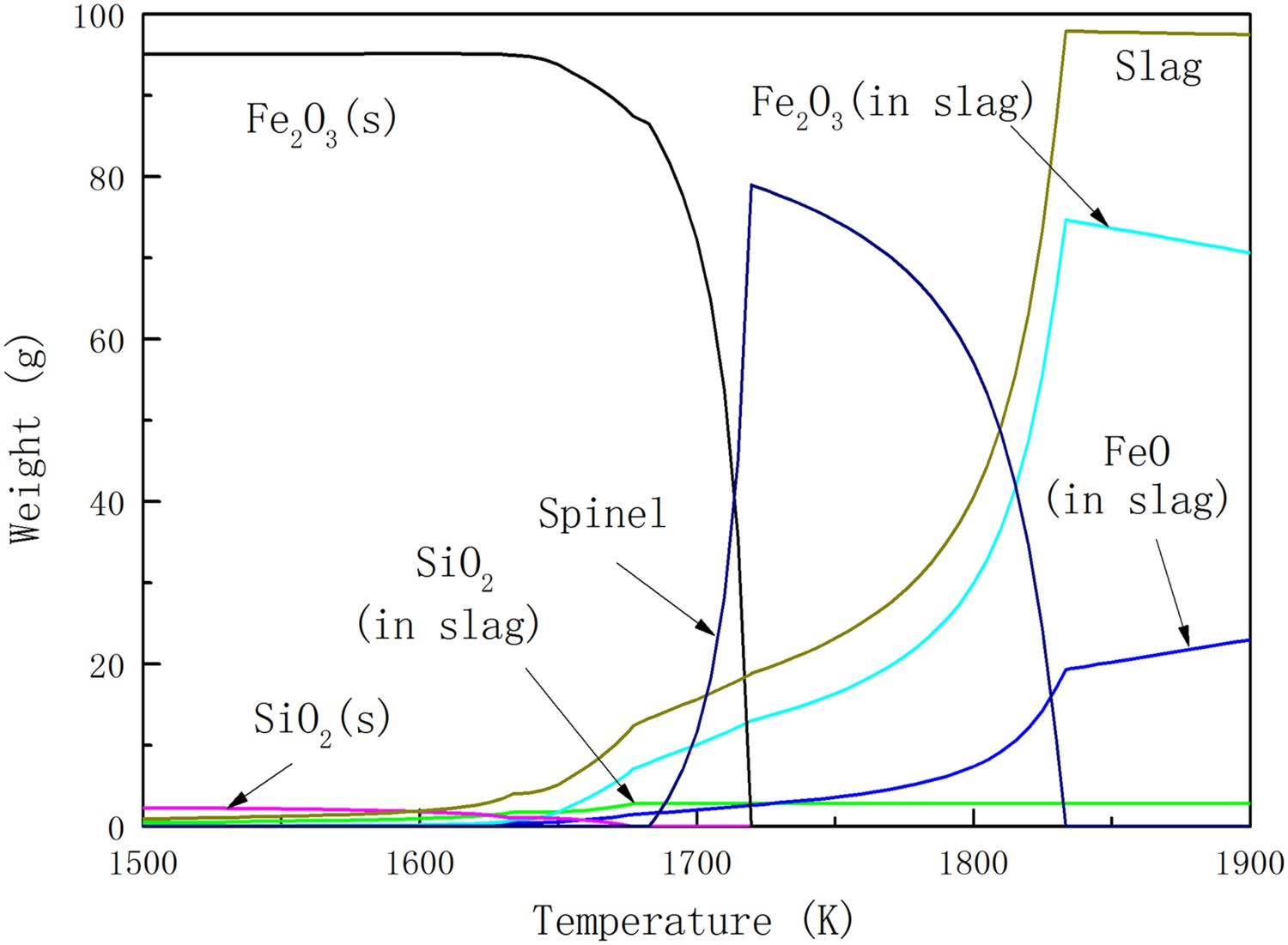
When pure argon with 100 times the weight of the ore is introduced into the system, it turns out a very different calculation result (Figure 2). The reduction degree of haematite ore in this system is deeper than the one without inert gas. In an inert atmosphere, haematite is no longer a stable phase during 1500–1900 K as mentioned above, and the melting of magnetite starts at least from 1630 K. Furthermore, the content of FeO in slag in the argon-containing system is much more compared with the system without inert gas. The reason of this phenomena is that the released oxygen from ore is diluted in argon gas. Lower oxygen pressure results in deeper reduction. It implies high gas/ore ratio could improve the thermal decomposition. Principal components of the haematite ore in equilibrium state at different temperatures in the argon-containing system.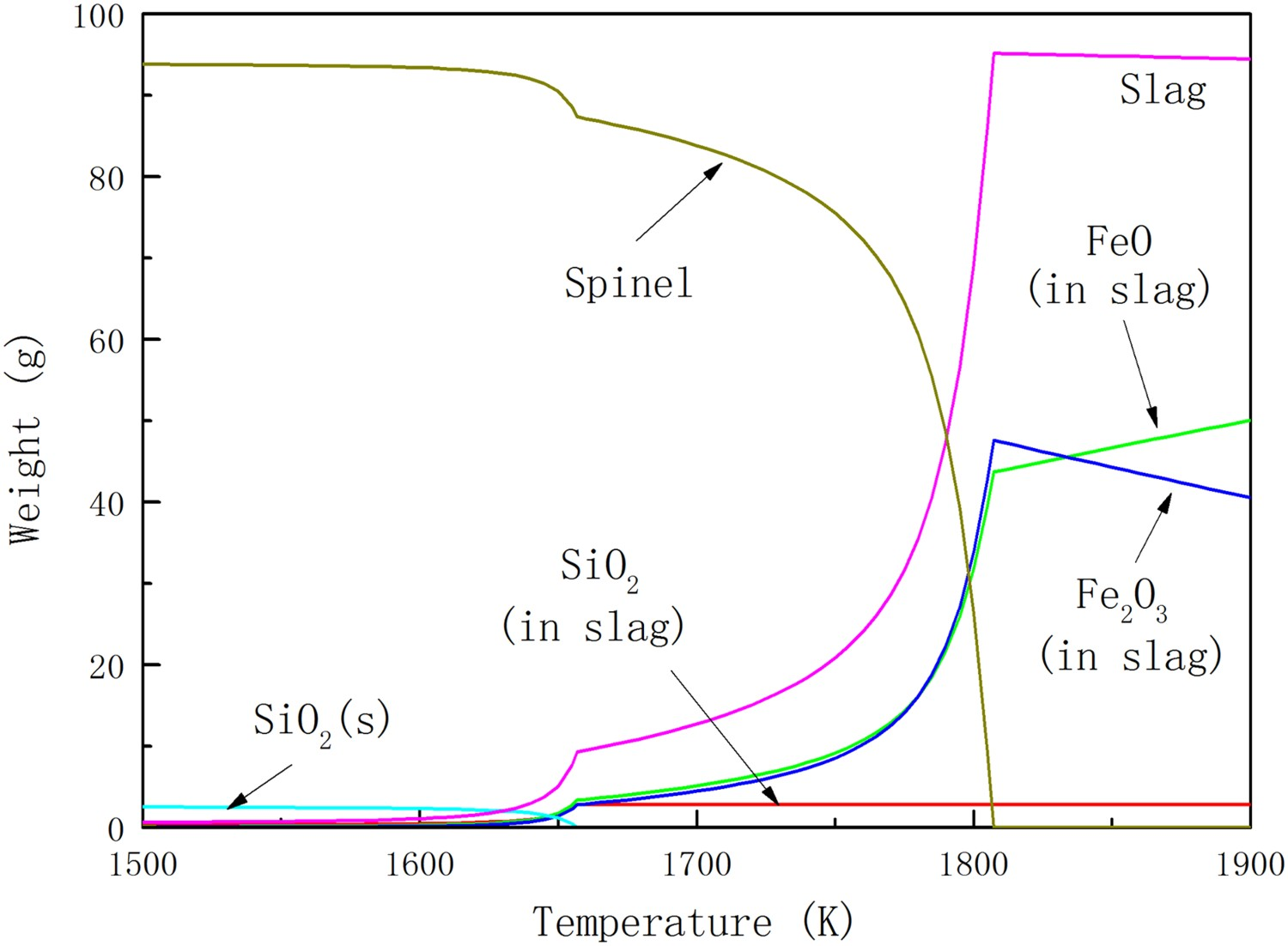
The difference between the weight of reduced ore and raw material (100 g) is the amount of released gas. The amount of released oxygen and its partial pressure in the argon-containing system are plotted in Figure 3. The ratio of the amount of released oxygen is corresponding to the reduction degree of iron ore. The partial pressure of oxygen increases with the increasing temperature. There are two turning points of the partial pressure curve of oxygen, which correspond to the melting of SiO2 in form of tridymite and complete melting of the ore, respectively. Reduction degree and the amount and partial pressure of released oxygen from thermal decomposition of the haematite ore in the argon-containing system.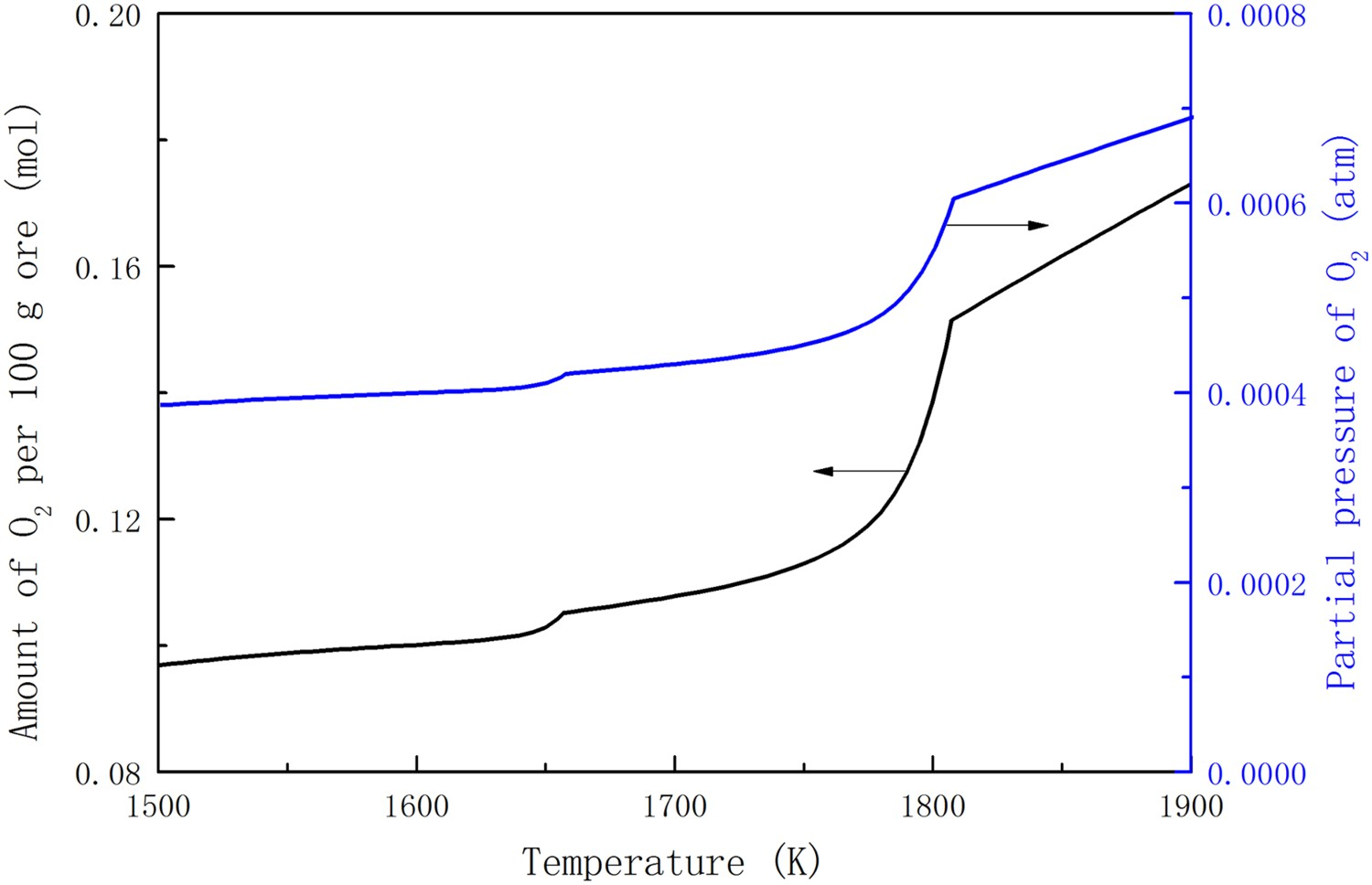
The amount of released oxygen has a linear relationship with the reduction degree of the haematite ore during thermal decomposition. The result in the argon-containing system indicates that the reduction degree of the ore increases with increasing equilibrium temperature. However, the effect of temperature on the reduction degree of haematite ore is not obvious until 1750 K. Even above 1750 K, elevating reaction temperature by 250 K can only raise the reduction degree of the haematite ore from 12.4% to 19.1% in thermal decomposition in the view of thermodynamics.
Topochemical gas reduction
As already noted, the product of thermal decomposition could not be metallic iron. The products that could result from the topochemical gas reduction depend on the composition of the gas, such as the PCR value. With lowest PCR value, the ore particles in PCR1 and PCR1’ gas could reach the highest reduction degree in equilibrium. The equilibrium state of haematite ore in PCR1 gas is shown in Figure 4 as illustration. Equilibrium state of the haematite ore in PCR1 gas atmosphere at different temperatures.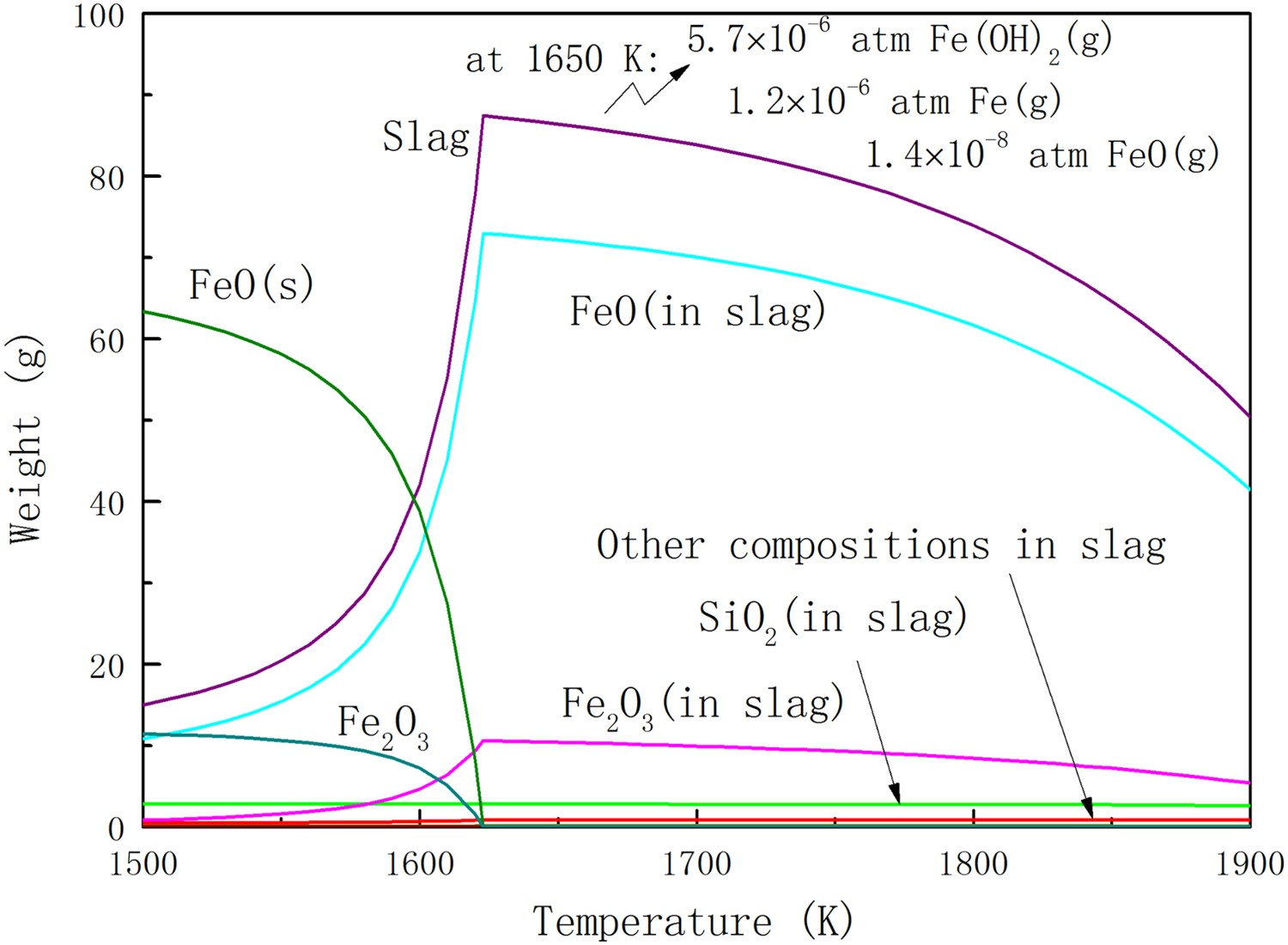
Figure 4 shows a significantly different equilibrium state of iron ore comparing to Figure 2. The most obvious difference is that the spinel phase is not a stable phase in the equilibrium state in PCR1 gas from 1500 to 1900 K. Abundant solid FeO and slag are generated above 1500 K. With the increase of equilibrium temperature, the amount of slag increases exponentially comparing with the mass reduction of solid phases. The molten gangues form a low proportion of the slag. And the ratio of Fe2+ to Fe3+ changes little above 1623 K. Because of a highly reducing atmosphere, Fe2+ maintains a high concentration in slag. It is well known that FeO rich slag could result in refractory erosion [9], which is a challenge of reactor maintenance. On the positive aspect, high FeO concentration can reduce the viscosity of the slag, and thereby accelerate ions diffusion in the slag. A high reduction rate could be obtained due to the high FeO content as a result.
Comparing to low temperature, a serious weight loss of the reduced ore at high temperature could be noticed. The principal volatiles at 1650 K are marked in Figure 4, which are mostly composed by Fe(OH)3. This is due to the high concentration of hydrogen (10 vol.-%) in PCR1 gas. Hydrogen or the produced water vapour could react with oxides, forming hydroxides. Usually, the partial pressure of hydroxides is much higher than the corresponding oxides. Comparably, high vapour pressure of hydroxides has been widely reported in the oxidation of boron nitride [10] and stainless steel [11] in hydrogen bearing atmosphere. It also suggests that high gas blowing rate in HTDF could result in a relatively serious weight loss of the ore.
In order to study the effect of PCR value and hydrogen content on the equilibrium state of haematite ore, the thermodynamic calculation results or the equilibrium state of the haematite ore in PCR3’ gas is shown in Figure 5. PCR3’ gas contains no hydrogen and has a higher PCR value. There are two significant differences between Figures 4 and 5: the liquidus temperature at equilibrium and the weight loss of slag at high temperature. The liquidus temperature of the ore in equilibrium is defined as the highest temperature where any solid phase is present in equilibrium. The weight loss of slag at high temperature is less serious in PCR3’, the hydrogen-free gas, than in PCR1 gas. The partial pressure of Fe(g) and FeO(g) remains almost the same in PCR1 and PCR3’ gases, while the total vapour pressure in PCR1 gas is larger than in PCR3’ gas. This is because the vapour pressure of Fe(OH)2 is 5 times that of Fe(g) and FeO(g). However, the vaporisation in practical system would not be so serious because of the high load of ore in the cyclone. The differences of slag weight loss in these two different situations may not be very obvious. Equilibrium state of the haematite ore in PCR3’ gas atmosphere at different temperatures.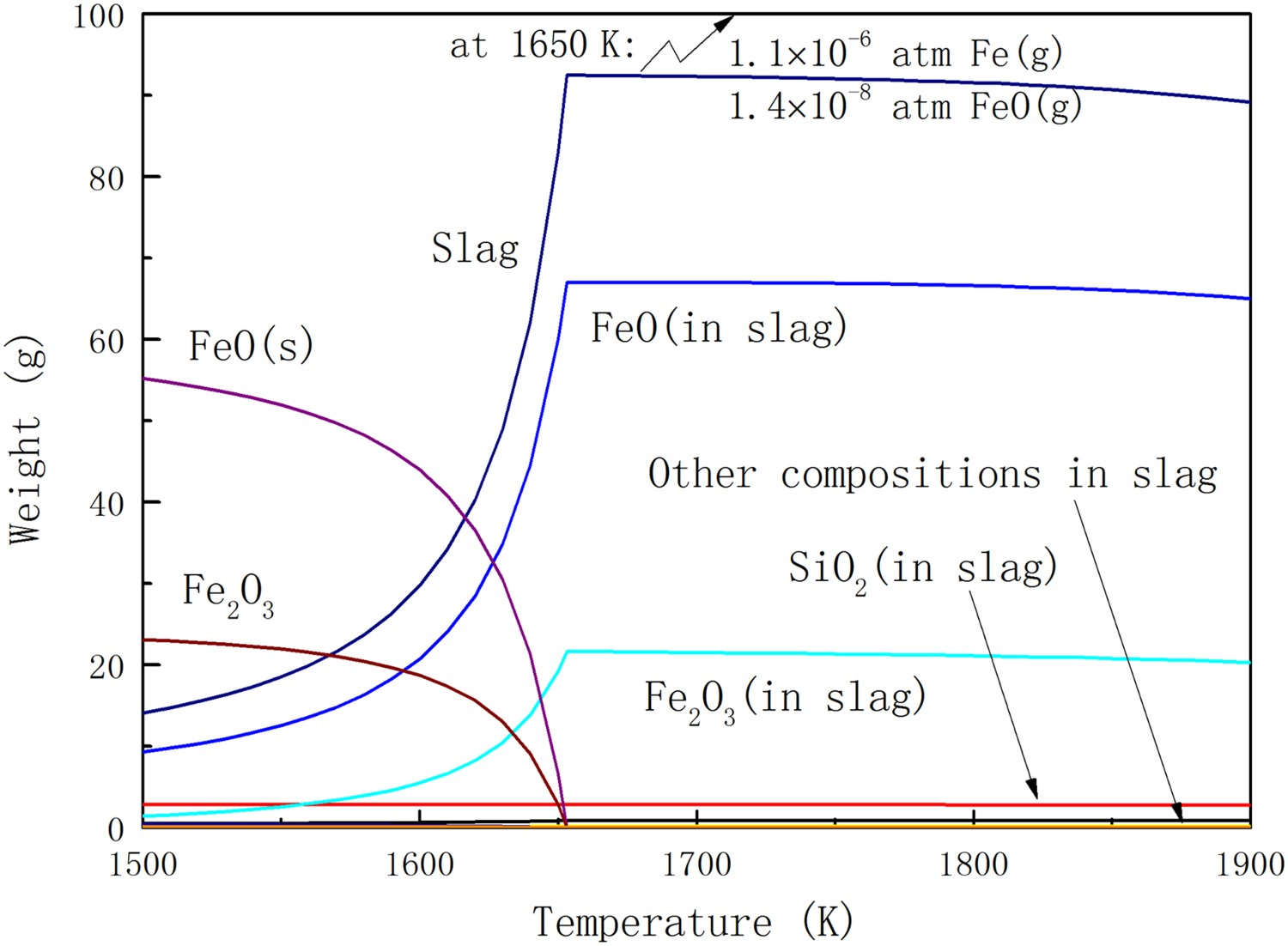
The relationship of the liquidus temperature and the gas composition is drawn in Figure 6. The figure illustrates that the liquidus temperature decreases with the decreasing PCR value of gas. Meanwhile, the liquidus temperature of ore in the hydrogen-free gas is a little higher than hydrogen-containing gas with the same PCR value. It implies that the phase state of ore particles can be adjusted by gas composition without changing PCR value and temperature. Liquidus temperature of solid phase of the reduced haematite ore in equilibrium state.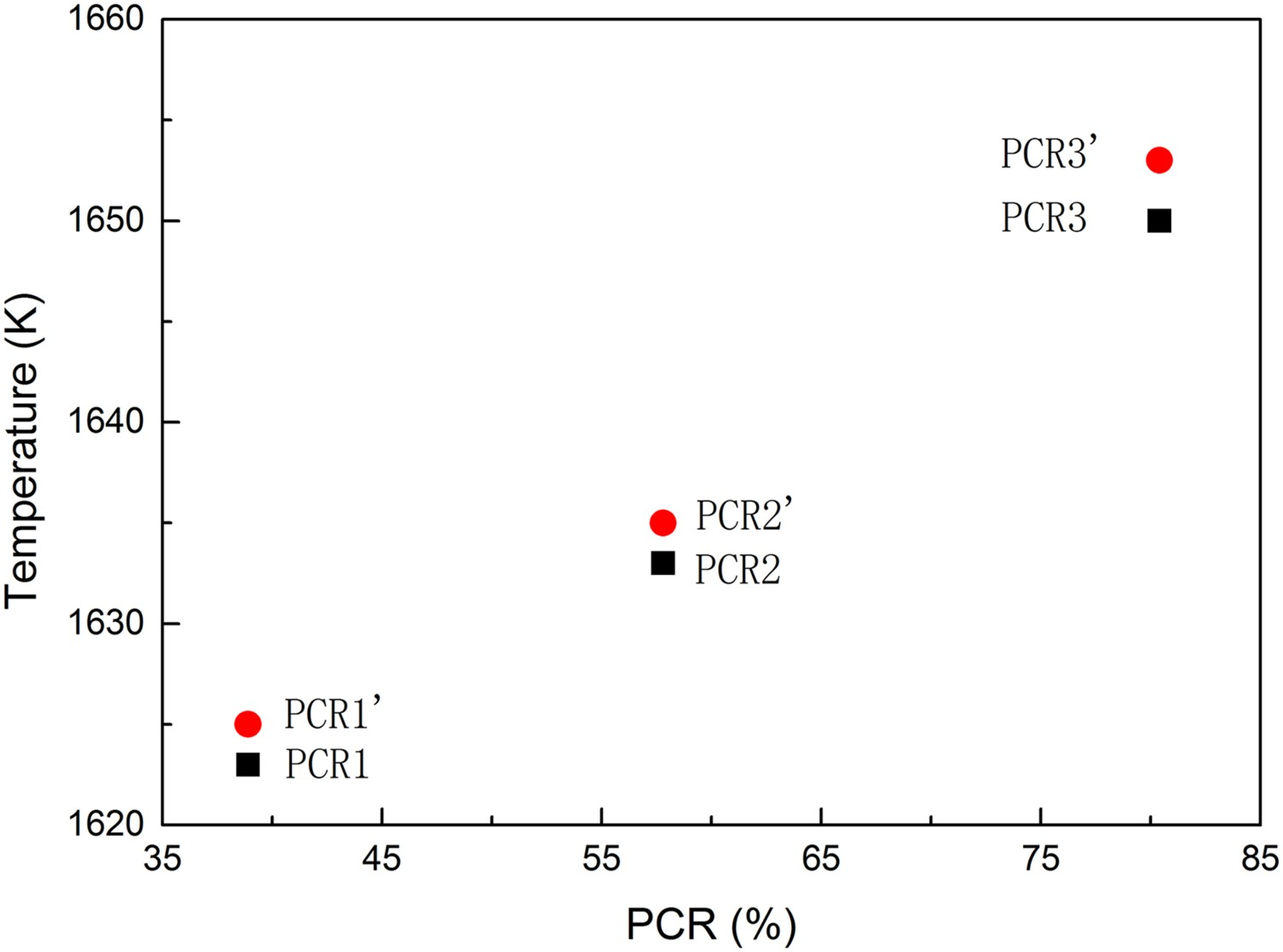
To examine the reason for the variation of liquidus temperature in different atmospheres, the slag compositions of the haematite ore in equilibrium are compared in Figure 7. A comparison of the slag composition is focused at 1650 K, a typical temperature in HTDF experiments. The ratio of Fe2+:Fe3+ decreases with increasing PCR value in reducing gas. It indicates a decreasing reduction degree of the haematite ore. But the principal composition of slag is also FeO in equilibrium in reducing gas. The melting point of FeO, 1644 K, is lower than the set temperature. FeO also has the lowest melting point in the ore components. It is expected that the liquidus temperature is principally decreased by an increase of FeO content in the slag [12]. As a conclusion, we know that iron ore in reducing gas will be reduced to a molten drop in HTDF if the residence time allows, and the lowest valence state of iron would be 2+. Slag compositions of reduced iron oxide ore in equilibrium state at 1650 K.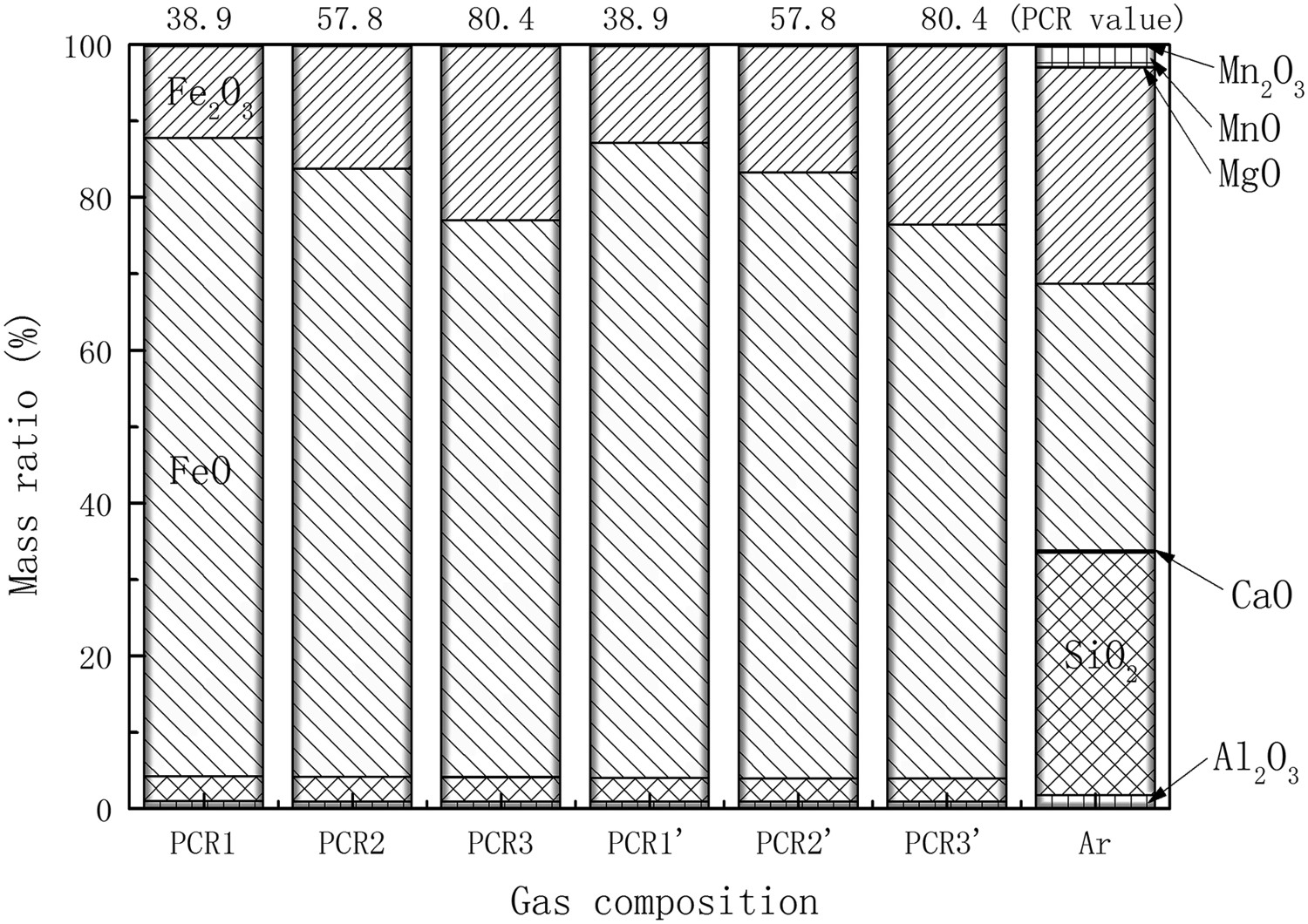
The content of other minerals in the slag at 1650 K are almost the same in different reducing gases. But a very little variation of SiO2 concentration could be noticed from the calculation results. It shows a slight decrease of SiO2 content with the increase of PCR value. The existence of hydrogen can also reduce the SiO2 content slightly. And one could see the difference of the slag composition in reducing gas and inert gas. It is known that the thermal decomposition is faster than the topochemical gas reduction in practice [6]. Therefore, the slag composition in inert gas could be very similar to the primary slag generated in reducing atmosphere in HTDF.
The theoretical content of FeO in the reduced ore and the reduction degree of the ore in PCR2’ gas are plotted in Figure 8 as functions of equilibrium temperature of the ore. It should be noted that the content of FeO is related to the reduction degree because it is the final product in the reduction. And the improvement of reduction degree of the ore is not obvious even though the equilibrium temperature has been elevated by 400 K. The HTDF experimental results of Qu et al. [5] are extracted into Figure 8 for comparison with the equilibrium states. Different from the calculation results, the experimental results show an obviously increasing reduction degree of ore with the elevation of the reaction temperature. And the experimental result is closer to the calculation one at higher temperature. It indicates two issues. First, the reaction rate of ore reduction could be improved significantly by elevating reaction temperature in the view of thermodynamics. It means the effect of temperature on kinetics is much more important than it is on thermodynamics. Second, the reaction time of 2020 ms is not enough for the haematite ore to reach the equilibrium state in the reduction process. This conclusion is consistent with the viewpoint of Qu et al. [4]. Comparison of equilbrium state of the haematite ore and the reported experimental results at 2020 ms in HTDF in PCR2’ gas.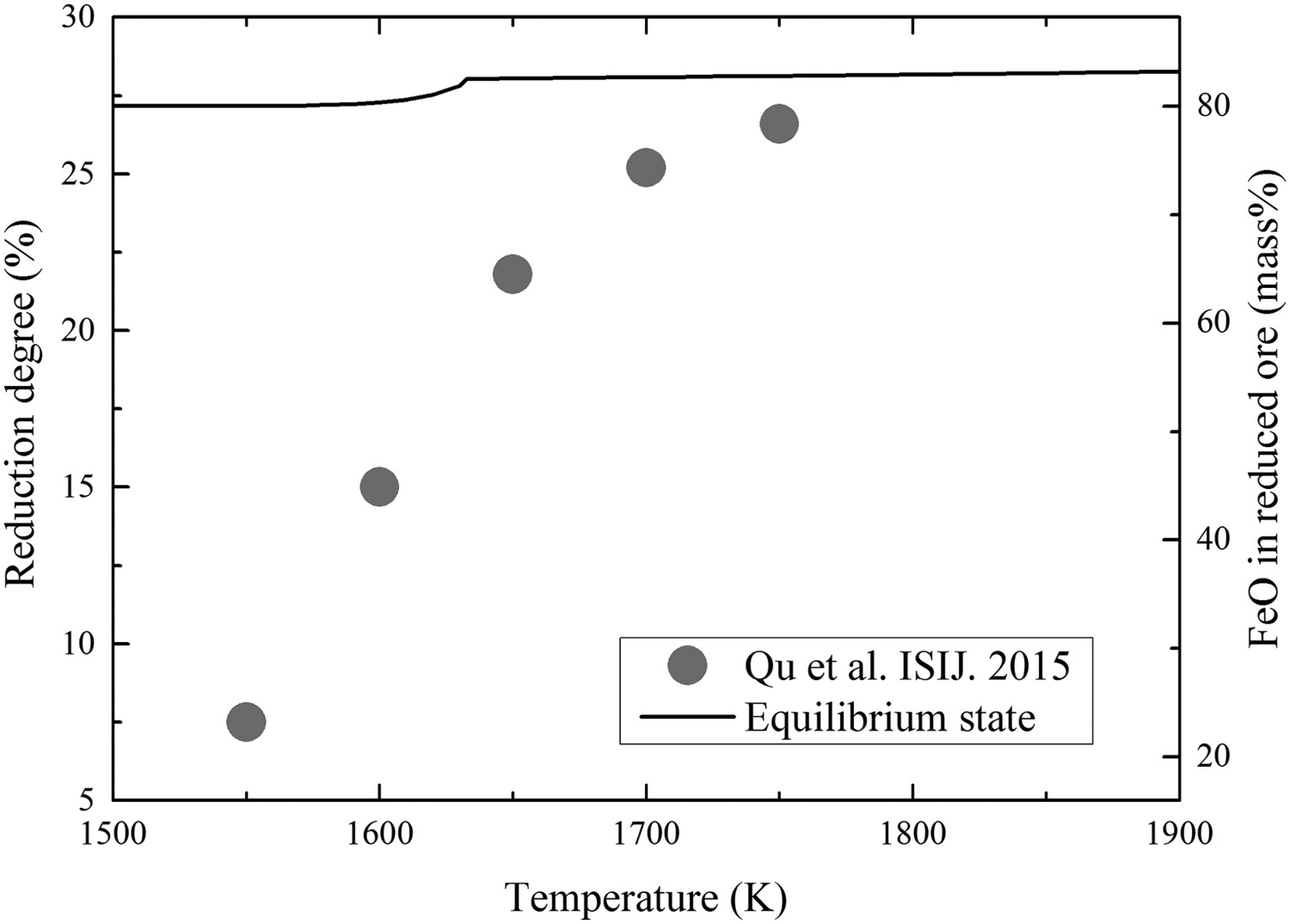
Conclusions
The haematite ore starts thermal decomposition from 1630 K to magnetite and FeO containing slag. The ore melts completely above 1720 K in inert gas. The reduction degree of the ore changes slightly after melting.
At high temperatures (>1660 K) in the reducing gas (PCR = 38.9– 80.4), the equilibrium state of reduced ore consists of FeO-based slag. FeO content in slag is higher in the gas with lower PCR value. It leads to a lower liquidus temperature of the reduced ore.
The reduction degree of the ore does not increase obviously with the increase of temperature according to thermodynamics. Comparing the calculation results with the reported experimental results, one can conclude that the residence time of ore particles should be longer than 2020 ms in the HTDF to reach equilibrium state.
