Abstract
With the advent of alternative smelting-reduction processes of ironmaking like COREX® and FINEX®, the demand for non-coking coals as a major raw material has increased. In these processes, char is formed from non-coking coals inside the melter-gasifier (MG) by pyrolysis. This char, either in lumpy or briquetted form, is the major reductant and fuel of the smelting-reduction process. Just like the coke in blast furnace, the char should also have certain chemical and mechanical properties suitable for operational requirements of the MG. However, there is a lack of systematic works related to the properties of chars from non-coking coals for ironmaking. This work aims to define a standardised testing method to determine the reactive and mechanical properties of lumpy and briquetted chars comparable to existing ISO norms. It also demonstrates the effect of different operational conditions on char properties. The testing method described in this work should be useful to the industry personnel to evaluate the quality of chars from non-coking coals for their usage in the MG.
Introduction
In the past decades, the steel industry has witnessed significant advances in the field of alternative ironmaking processes. Although well-defined and widely standardised raw material testing methods have been established for the blast furnace process extensively, there exists an absence of systematically standardised and widely accepted norms in case of different alternative ironmaking processes. Internal standards of different companies for COREX® coal testing vary significantly and can sometimes cause ambiguity. COREX® and FINEX® have established themselves as commercially successful smelting-reduction processes in the last couple of decades [1–3]. The main reductant for COREX® and FINEX® is non-coking coal. Although non-coking coals have much inferior properties than the traditional blast furnace coke, they must also have certain chemical and mechanical properties under operational conditions [3]. A suitable type of solid reducing agent should not exceed a certain degree of reactivity (when affected by reactive gases) when it descends through the bed of the melter-gasifier (MG) and must also have satisfactory mechanical properties to support the load of the burden in the lower part of the furnace. The present work is related to the evaluation of the behaviour of non-coking coals under different simulated process conditions of the MG. The major aim of this work is to suggest a standard test method to determine the reactivity and strength indices of chars (both lumpy and briquetted) from non-coking coals keeping analogy with internationally accepted test norms. In case of briquettes, influences of different binders and coal blends have been examined additionally. A standard blast furnace coke was chosen for the purpose of comparison.
Coal and char in the MG
The MGs of both of the COREX® and FINEX® processes are principally identical. Any discussion pertaining to the MG will refer to both COREX® and FINEX® process. Non-coking coal is charged directly into the dome of the MG, along with DRI and fluxes [4]. As the temperature of the dome varies between 1000–1100 °C, the coal experiences a thermal shock directly upon entering the MG and the pyrolysis is started at once. Smaller coal lumps are converted to char as it becomes devolatilised on its way through the freeboard zone until it reaches the fixed bed. Coarse lumpy coal is mainly devolatilised in the upper region of the fixed bed. In actual process, the coal undergoes in-situ pyrolysis in the MG while being attacked simultaneously by reactive gases like carbon dioxide (CO2) and small amount of steam. The solid carbon structure derived by the devolatilisation of coal is called char. The char bed provides support to the burden inside the reactor. A solid column of char similar to the Dead Man’s zone of blast furnace is also formed at the lower zone of the MG (Figure 1).
Schematic section of the MG.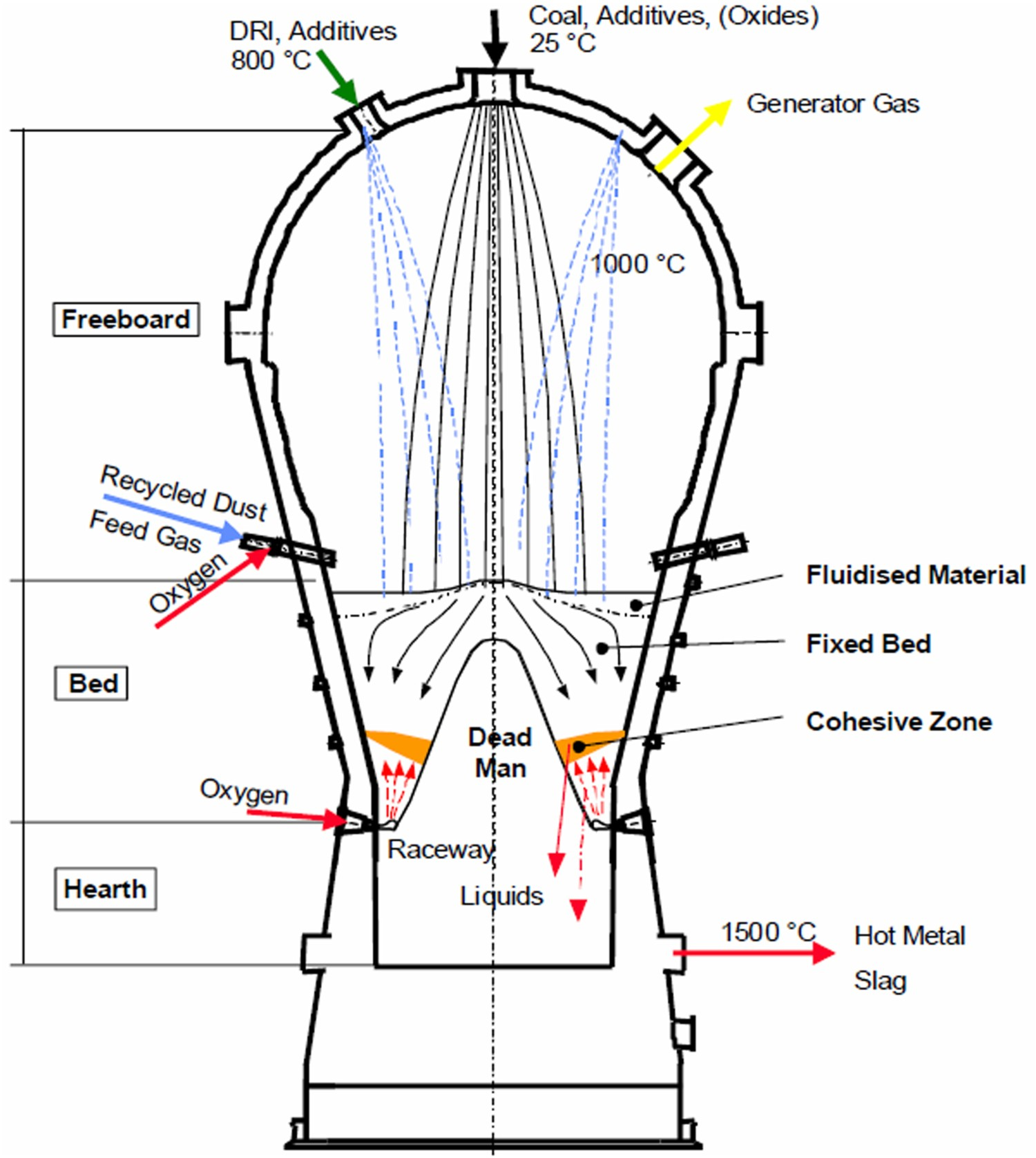
Reactivity and strength tests
Only very few publications have dealt with the experimental procedure for the determination of the reactivity and strength of chars in MG [4,5]. However, the processes have not been explained in detail. The internal norms of various companies (which are confidential by nature) also differ from each other. The authors of the present work communicated an attempt to measure the reactivity and strength of chars at the very basic stage of this work in 2014 [6]. The authors here aim to suggest a detailed standardised test method for the determination of the reactivity and strength of chars, which would be comparable to the internationally available standards. Two slightly different methods are explained, one for lumpy specimens (mentioned hereafter as Standard L) and the other one for briquetted specimens (mentioned hereafter as Standard B).
Standard L
The test would be performed in two steps, namely the following:
Step 1 – Preparation of char from coal samples
A retort furnace for the purpose of pyrolysis is heated at 1000 °C under N2 atmosphere. The coal sample (+16–20 mm size fraction, 300–400 g mass) is directly charged into the heated furnace (at 1000°C). It is kept at this temperature for 60 min. The way of charging the coal sample into the preheated furnace simulates the thermal effect on coal due to the rapid heating condition after entering the MG. After 60 min, the char is pushed out from the furnace into a quench-box and cooled to room temperature under N2 flow.
Step 2 – Reactivity and strength tests
The experimental determination of the reactivity of the char samples is based on the standard technique of the ‘Determination of coke reactivity index (CRI) and coke strength after reaction (CSR)’ (ISO 18894) [7]. As the reaction conditions inside the MG are not very much different from the conditions of the blast furnace, this test procedure has been implemented with a few modifications. The following terms will be used instead of CRI and CSR as the material discussed here is not coke.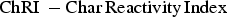
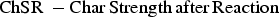
These parameters and the test conditions are analogous but not identical to RI (Reactivity Index) and RSI (Reaction Strength Index) used in the COREX® coal specification by Primetals Technologies. Some deviations from various currently existing methods were required for enhanced compatibility with ISO 18894. The error margins and repetition conditions were kept similar as ISO 18894 [7].
For the ChRI test, the char has to be crushed to a size of +5–10 mm. This is a mentionable deviation from ISO 18894. The charged coal suffers more abrasion and breakage in the MG compared to the blast furnace coke and therefore, the mean particle size [8] is much lesser than the standard blast furnace charge. As a result, the average lump size of coal in MG is lesser than BF cokes. Also, the selected size range represents a broad size variation of the specimen inside the MG. The largest lump in the sample mass is doubly bigger than the smallest lump in this sample size range. This deviation was incorporated aiming for a closer process simulation.
A homogenised sample having a mass of 200 ± 3 g is filled into a steel retort designed as per ISO 18894 and then heated up to 1100°C while the sample is held under an unreactive atmosphere of N2 flowing at 10 l/min. When 1100°C is attained, the temperature is allowed to stabilise for 10 min. The atmosphere is then changed by purging CO2 into the retort with a flow rate of 5 l/min. The char is gasified by forming carbon monoxide (Boudouard Reaction). This reaction is endothermic [9].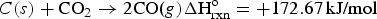
Although the internal standard of Primetals Technologies uses a reaction time of 1 hour, in this work it was modified and standardised to 2 hours for better comparability with ISO 18894. After 2 hours of exposure to CO2, the test is stopped and the retort is cooled down to room temperature under N2 (5 l/min). The experiments are repeated to check the reproducibility as per norm. For detailed experimental setup, the standard ISO 18894 is to be consulted.
The ChRI is calculated as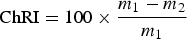
m 1 = mass of char before the reaction
m 2 = mass of char after the reaction
After reaction, the reacted sample mass is taken out from the retort and tumbled in the standard tumbler made according to ISO 18894 at 20 rev min−1 for 30 min. Afterwards, the sample is sieved using 5 mm and 3.15 mm mesh sieves. Therefore, two different ChSR values can be obtained.
ChSR is defined as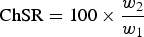
w 1 = mass after reaction
w 2 = mass > x mm (after tumbling)
where, x = 5 mm or 3.15 mm
ChSR is to be expressed as ‘ChSR > x’ while reporting.
Variations in process conditions
Other than the standard test conditions, the following process parameters were varied for all char samples to find their effects on the reactivity and strength. Gas composition Temperature Particle size
Test parameters.
Standard B
This special standard would be more useful industrially as most of the MGs in modern days are charged with briquettes as the coal source. The advantage of this method is to be able to evaluate the properties of whole briquettes eliminating the need of breaking them. Here, an additional step related to the production of briquettes will be mentioned.
Step 1 – Briquette Production
The coal is crushed and screened to produce the feed for making briquettes. The particle size of coal fines used for briquetting is ≤4 mm. Water and certain binders (e.g. molasses) are added to the crushed coal and then mixed thoroughly in an industrial mixer. Coal briquettes are produced from this mixture using a roller briquetting machine. The closest geometric analogy of the produced briquettes could be considered as an approximate ellipsoid. The average dimensions of a single briquette are: 46 mm (major axis) × 34 mm (minor axis) × 25 mm (maximum thickness). The average volume is around 20cm3. The briquettes thus produced are sieved to desired size by removing broken specimens and residual crushed coal.
Step 2 – Preparation of Char Briquettes
In this case, whole coal briquettes are charged into the heated pyrolysis furnace. Otherwise, the charring and cooling process of briquettes is similar as explained before in chapter 3.1.
Step 3 – Reactivity and Strength Tests
For briquettes, certain conditions should be slightly deviated from Standard L. In this case, whole briquettes should be charged in the reaction retort. The mass of each charred briquette varies in the range 9–12 g. Therefore, the mass input in the retort is changed to 200 ± 6 g. Certain semi-coking coals show strong caking properties, resulting cracked and broken briquettes after pyrolysis. In this case, if whole briquettes are not available, broken briquette pieces in the size range of 34–46 mm are to be accepted to remain closer to the actual geometry. The ChRI and ChSR are to be determined in the similar way stated before.
Sample selection
Proximate analysis of coals and chars (all values are in mass- %).
Proximate analysis of coke (all values are in mass-%).
Variations in briquetting process
The briquettes were produced with different types of binders (molasses and carbon based) and coal sorts to examine their effects on briquette properties under certain conditions. The binders used here are molasses and another carbon-based binder (CBB). The master table of briquette types along with the test results is provided in chapter 5.2.
Results and discussions
Standard L tests
The standard tests reveal that C1 has the highest reactivity and lowest strength while, as predicted, the coke sample CK has the lowest CRI value and the highest CSR. C2 has intermediary values. For the sake of comparability, the coke sample was also tested with the same size fraction as chars (5–10 mm). Figure 2 shows the difference between standard properties of all the samples.
Reactivity and strength under standard test conditions.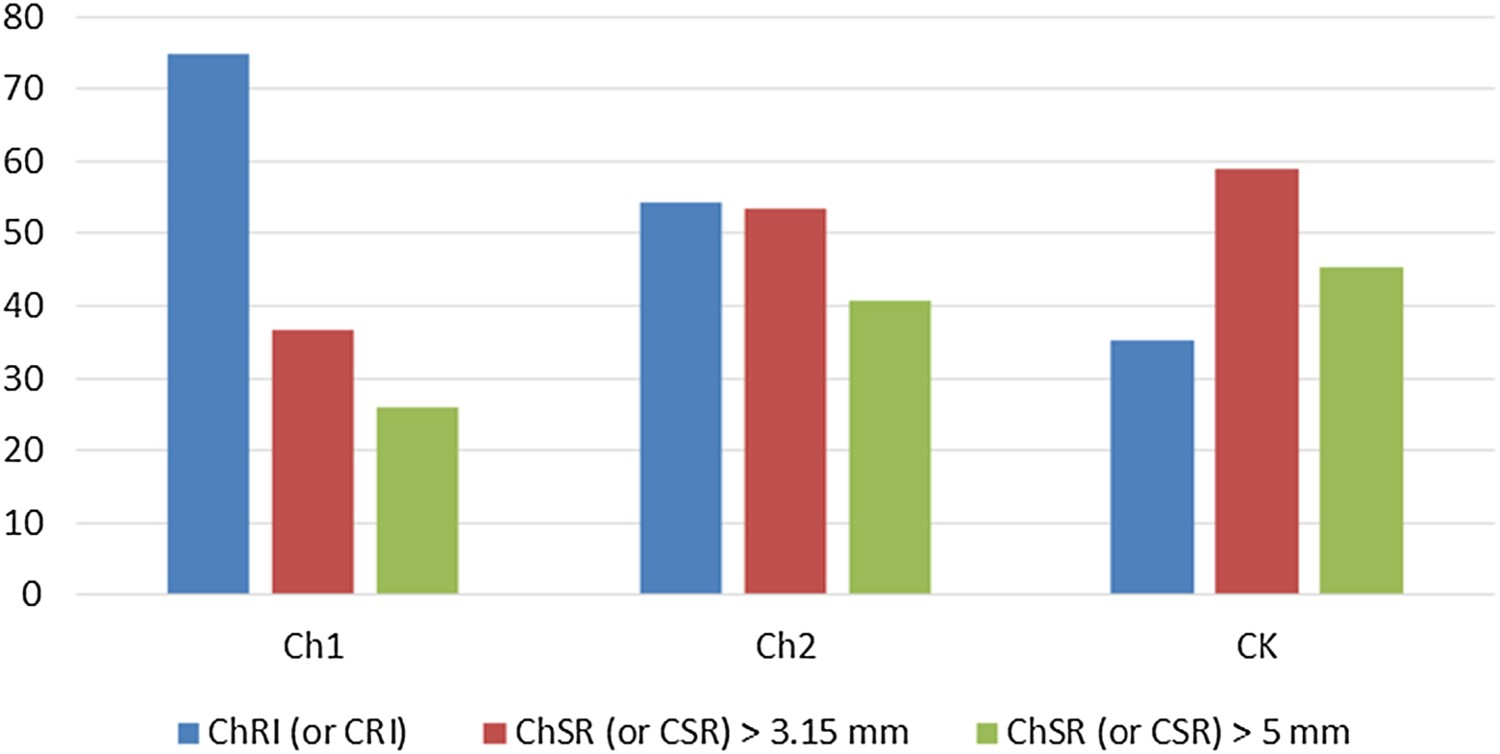
Test results under different conditions for lumpy chars.
* Sieve used after tumbling: 10 mm.
Changing the gas composition to a 1:1 mixture of CO2 and N2 (Test CO2N2) keeping the flow rate constant results in lower reactivity and higher strength in all cases, whereas experiments under steam and CO2 mixture (Test CO2H2O) gives rise to much higher reactivity values and corresponding lower strength indices compared to Standard L. Experiments conducted at different temperatures show significant lowering of reactivity indices at 1000°C and subsequently lower temperatures. ChSR increases as the reactivity decreases. ChSR indices at 1000°C and 900°C interestingly show slightly higher values for Ch1 than Ch2. However, the differences are very small in comparison and the values can be considered almost equal. Change of particle size range to 19–22.4 mm (Test VarS) showed significant lowering in reactivity for Ch2. It was observed that Ch1 is so strongly reactive that it demonstrated much lesser change in ChRI compared to the other samples even when experimented at a larger particle size. When screened using a 10 mm sieve after tumbling as per ISO 18894, it exhibited an almost insignificant ChSR value.
Comparative reactivity and strength indices of char samples under variable process conditions are summarised in Figures 3–5.
Comparison of ChRI values. Comparison of ChSR (>3.15 mm) values. Comparison of ChSR (>5 mm) values.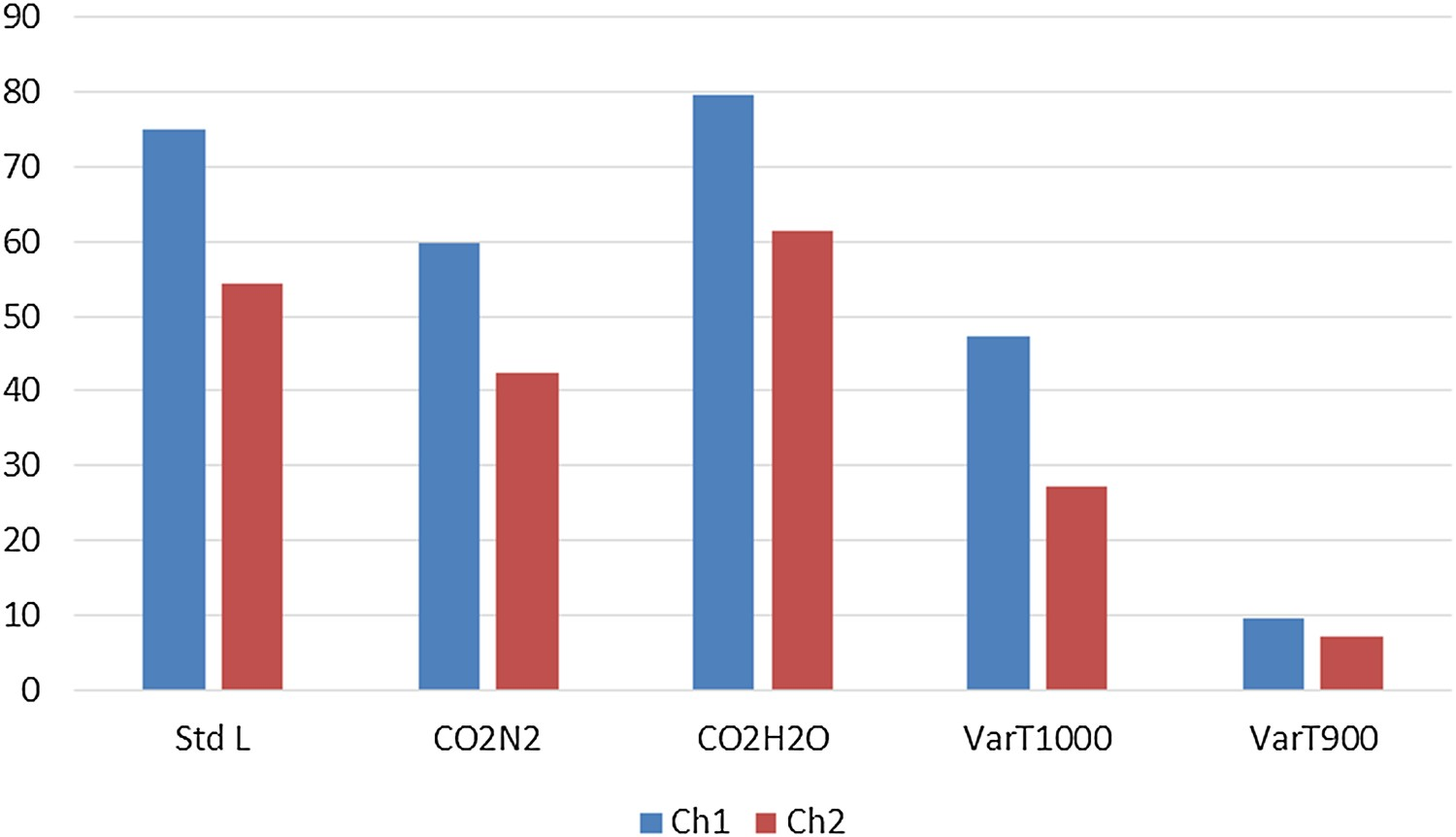
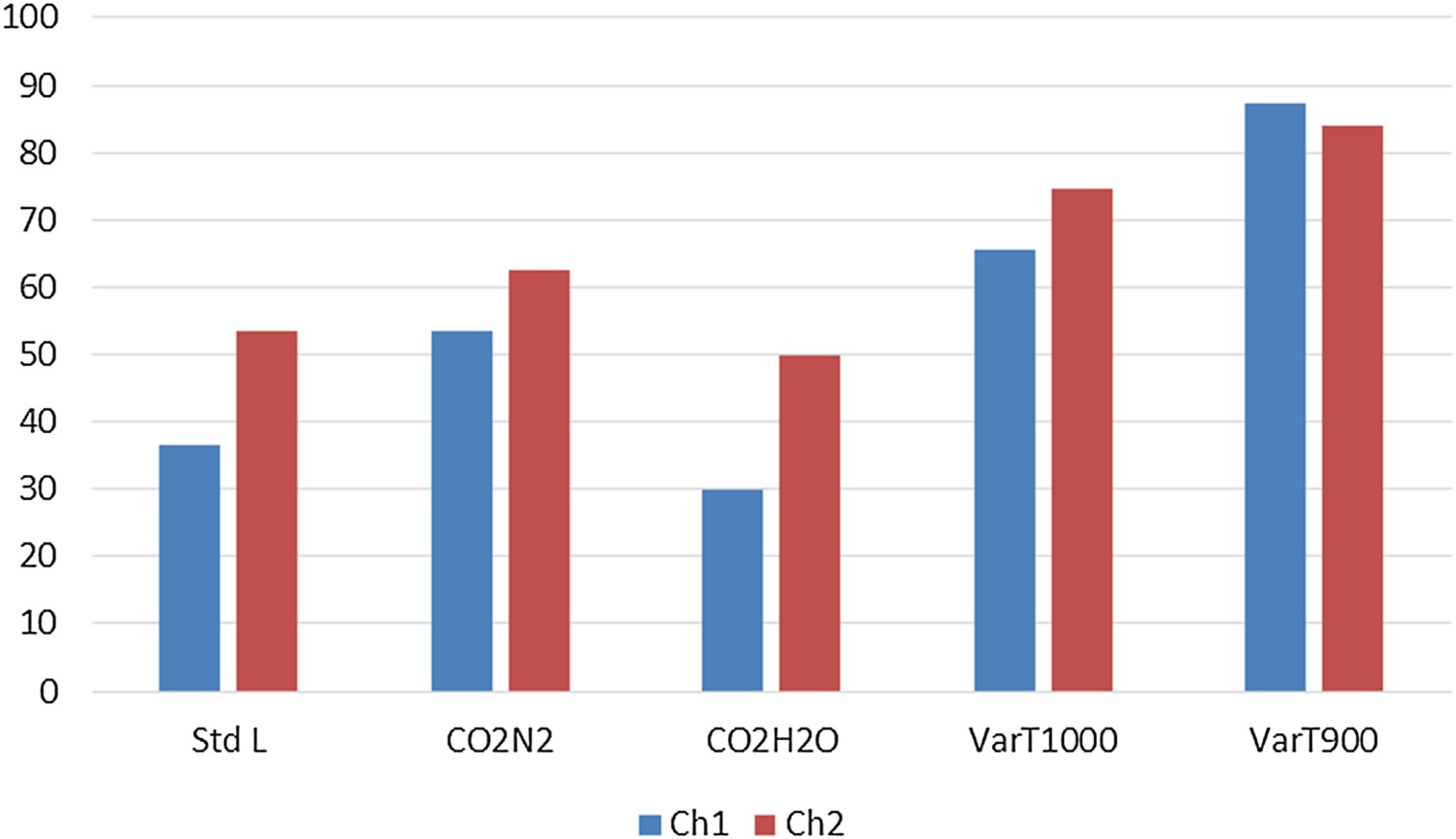
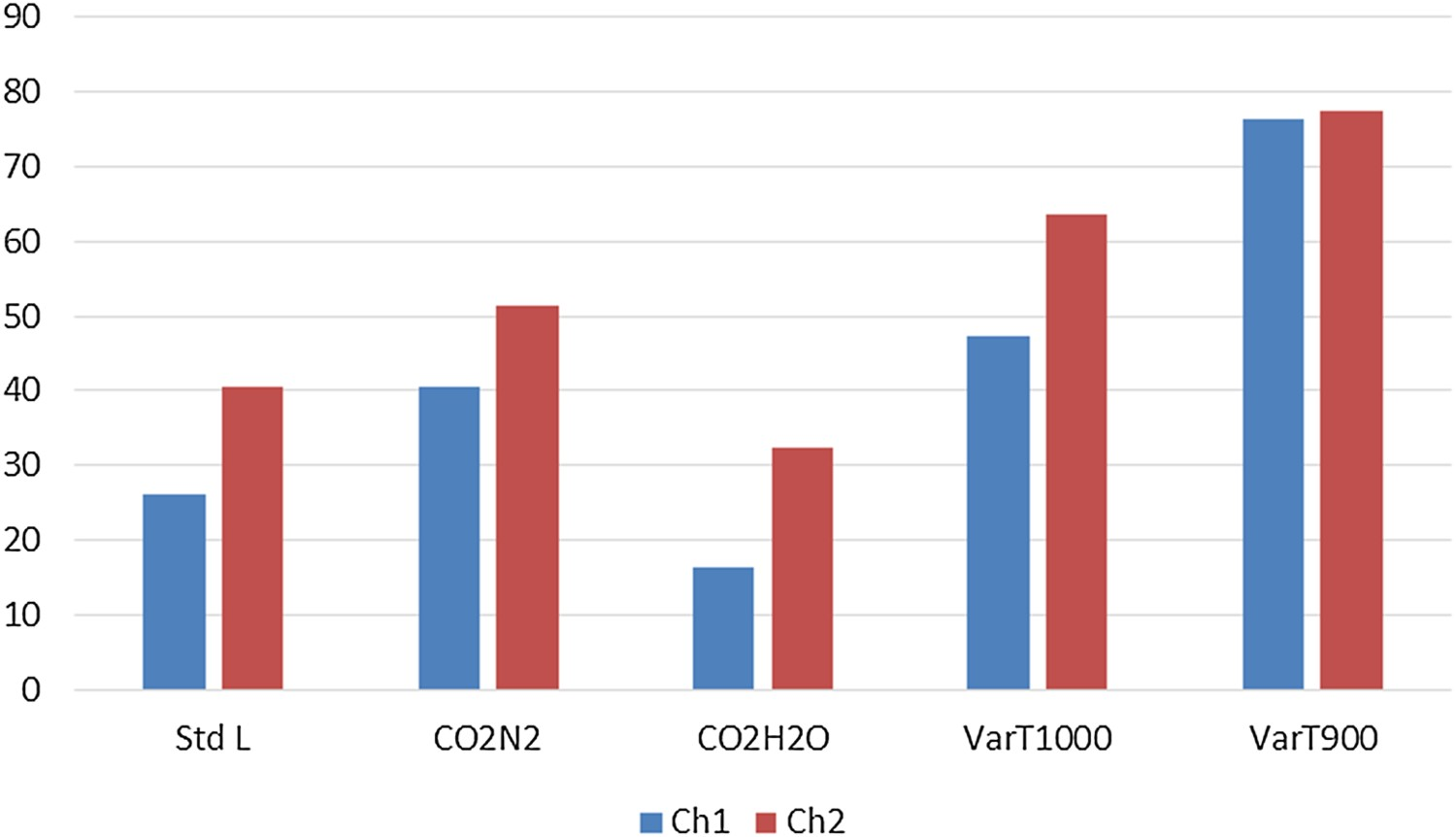
Standard B tests
Test results of briquettes (Standard B).
C2 showed the properties of a semi-coking coal. When devolatilised, the briquettes made of C2 coal showed long cracks and breakage along the cross section. No single C2 briquette was retained in whole shape as it was before charging. Therefore, the size selected for the tests are sieved as mentioned in the description of Standard B.
The following observations could be summarised from the results C1 shows better reactivity in the basic briquetted format (B1) compared to lumpy char (Standard L). However, the strength after reaction decreases substantially. When used as a binder, CBB results in better properties for C1 (B2). C2 shows more or less consistent properties in all cases (B5 and B6). The ChSR values are slightly better when using CBB. The briquettes with best properties are made using a blend of C1 and C3 (B3 and B4). B4 shows the best combination of strength and reactivity. CBB is suggested to be the binder implementing best properties.
Comparison of Standard L and Standard B
A comparison of Standard L and Standard B tests are shown in Figure 6. The basic molasses bonded briquettes have been used for comparison. The betterment of reactive properties (i.e. lower ChRI) is evident. However, for a weaker char like Ch1, the strength index would be significantly lower as the briquette will tend to abrade more during the tumbling process.
Comparison of Standard L and Standard B.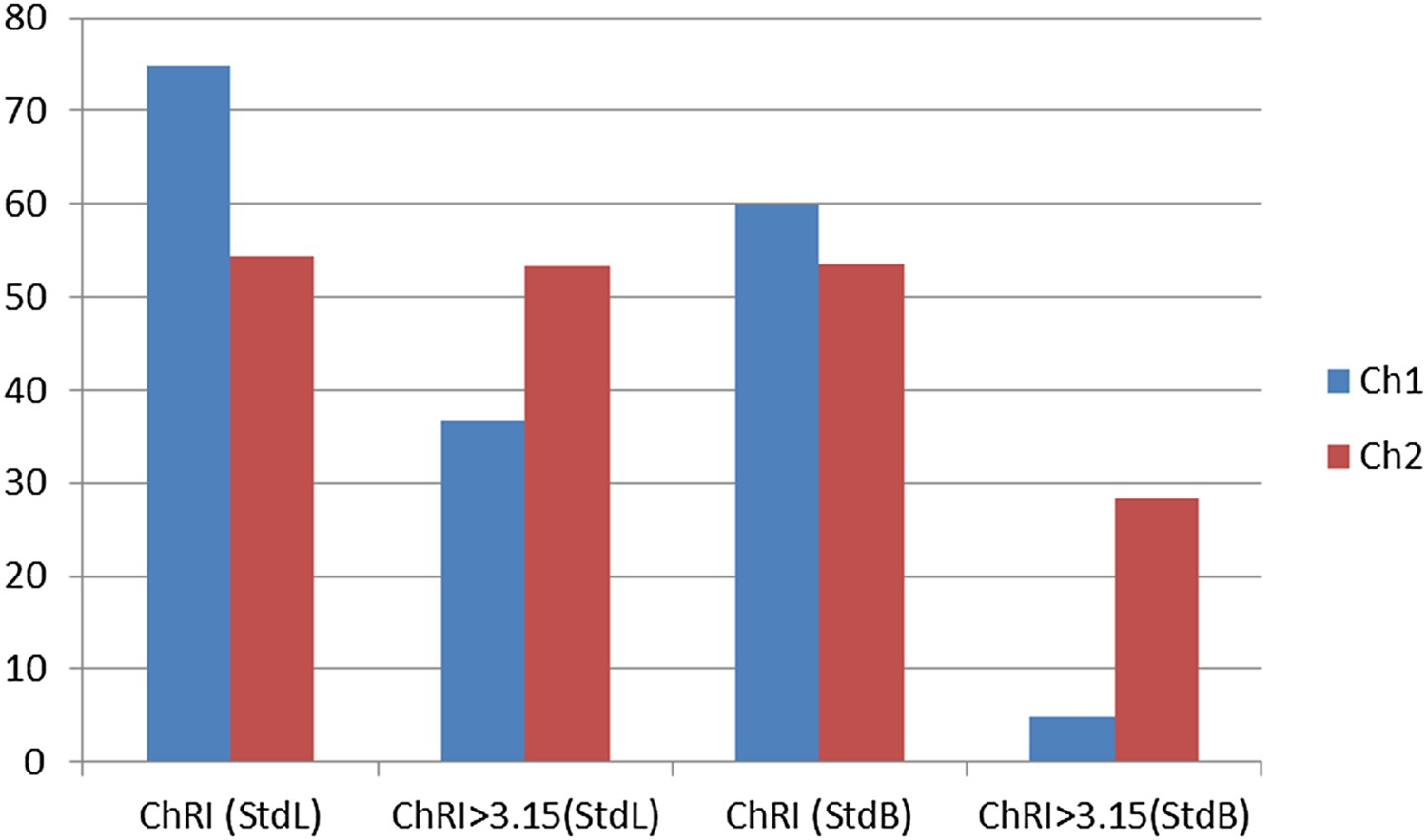
It is also important to note that for all practical industrial purposes, the sieve size 3.15 mm is recommended to report the final ChSR values. However, the screening through 5 mm sieve indicates the mechanical stability of bigger coal lumps in the charge and is also advised to perform when required by the operator.
Conclusion
In the current work, a standardised procedure has been suggested for the experimental determination of the reactivity and strength of chars from non-coking coals under MG conditions. Both lumpy and briquetted chars have been taken into consideration. This test method is also modifiable if there is a mixed charge containing both lumps and briquettes. In that case, the input material could be modified to the actual charge ratio of lumps and briquettes in the real process. The effect of different process conditions on the strength and reactivity of lumpy chars has been studied. The influence of various additives and binders on briquettes has also been investigated. A comparability check with existing standards has also been taken into consideration. As a corollary, the Standard B procedure opens up a greater scope of further research activities in the domain related to the optimisation of briquetting parameters (coal blends, binders, etc.) suitable for MG operations. The authors believe that this testing procedure would be useful as a standard test method for the quality evaluation of chars from non-coking coals under COREX®/FINEX® conditions, providing more insight on the coal/char quality to the industry personnel for a more effective and optimised process operation.
Footnotes
Acknowledgements
Financial support provided by K1-Met is gratefully acknowledged. K1-Met is the member of COMET – Competence Centres for Excellent Technologies and is financially supported by the Austrian ministers BMVIT and, BMVITJ, the provinces of Upper Austria, Styria and Tyrol, SFG and Tiroler Zukunftsstiftung. COMET is managed by FFG (Austrian research promotion agency).
Disclosure statement
No potential conflict of interest was reported by the authors.
