Abstract
This paper concerns the mechanism of iron oxide reduction and rare earth compound transformation in carbon monoxide circumstance. Rare earth Bayan Obo complex iron ore (REBOCIO) was reduced by CO–N2 gas mixtures in a laboratory fixed bed reactor in the temperature range from 1073 to 1273 K. The influences of reductive conditions on the reduction rate including reduction temperature, reduction time and carbon monoxide content were studied. And the characteristics of reduced samples were analysed by XRD, BES and EDS. It is shown that the reduction degree increased with the increasing of temperature and monoxide content. Above 1123 K, the reduction is controlled by interfacial chemical reaction at early stage of the reaction and then turns to the internal diffusion controlling with reaction processing. The rare earth mineral transformation path at temperatures above 1123 K in the process of reduction is suggested as follow: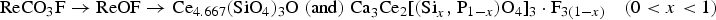
Get full access to this article
View all access options for this article.
