Abstract
This investigation highlights on surface modifications and adhesive bonding of aluminium. The surface of the aluminium substrate is modified by magnetron sputtering and nanoparticle reinforcements are employed into the adhesive and their effects on adhesive strength are examined. Owing to the concern of durability of the aluminium surface, adhesive joints are still questionable for their widespread use in structural applications. Various environmental conditions affect properties of adhesives and their performance may degrade over time when adhesive joints are exposed to varying moisture and temperature conditions. Modified aluminium surfaces are examined by morphological techniques to understand the result of deposition on the morphology of substrate. Surface modified samples are subjected to harsh environments and analysed by energy dispersive spectra and surface durability is quantified by corrosion penetration rate. The effect of nano-reinforcements in adhesives on bond durability is examined when the bonded joints are subjected to the harsh environment.
Introduction
Aluminium has been established as the most acceptable structural material for aerospace industry for quite a long time [1,2]. The eminence of aluminium over other metals in aerospace specific applications is due to its superior qualities like relative abundance, lightweight and low cost [3]. The widely employed conventional methods of joining aluminium parts like welding and riveting have some shortcomings as they induce thermal and mechanical stress in the parts. In order to overcome these disadvantages, extensive research is being done to develop adhesive bonding as an effective alternative and reliable method of aluminium joining [4]. Adhesive bonding provides uniform stress distribution between the aluminium joints, relatively less weight and easy fabrication process when compared with welding and riveting.
The process of adhesion between the polymer adhesive and metal substrate can be explained by mechanisms like interlocking, diffusion or adsorption [5-7]. The bond strength mainly depends on the interactions between the adhesive and the substrate. Therefore, the condition of the surface of substrate is crucial to ensure a strong joint [8]. In the case of aluminium, when the surface is exposed to oxygen, it forms brittle aluminium oxides which are not favourable for good joint strength [9]. Further, quality of the adhesive bond primarily depends on its durability in the service environment. Adhesive joints are considerably sensitive to environmental conditions especially humidity and high temperature [10]. This investigation deals with the study of effects of surface modification and adhesive reinforcement on strength and durability of adhesive bonded joints.
Titanium nitride is a hard ceramic material with high chemical stability thermal and low electrical and thermal resistivity [11]. In this study, titanium nitride is deposited over the substrate particularly by sputter deposition as it can be implemented irrespective of the substrate.
With the emergence of nanocomposites, the enhancement of mechanical properties due to unique interactions of polymer with fillers has been established [12]. The usage of polymeric materials and adhesives in major components of aerospace is still narrow due to high temperatures experienced by aerodynamic heating. This drawback can be overcome by the thermally stable nanofillers reinforced high-temperature resistant adhesives [5]. Nanosilica is being used as nanofillers due to their ease of functionalisation, thermal stability and low toxicity.
Therefore, in this investigation, the effect of surface modification by titanium nitride deposition on aluminium alloy and the effect of nanosilica reinforcement in adhesive on strength and durability of adhesive bond are studied. Further, the surface morphology characterisation and surface durability test is carried out for titanium nitride deposited aluminium sample. The study is based on the lap shear tensile testing of joints prepared with substrate and adhesive and this work is strongly recommended by the aerospace industry.
Experimental details
Materials
Aluminium alloy 2024 as adherends were used in this investigation. Duralco 4703, an epoxy-based adhesive is used as the adhesive. It is manufactured by Cotronics Corporation. The adhesive was mixed with hardener over the substrate surface in a liquid state. Its maximum use temperature is 350°C. The mixing weight ratio of adhesive with hardener is 100:22 while the curing takes place at 120°C for 4 h. Nanosilica powder is dispersed (5% by weight) in the adhesive at room temperature.
Surface modification
The samples were cleaned with acetone before surface modification of aluminium. Plasma-based sputtering system was used for the surface modification process. Plasma direct current (DC) magnetron sputtering system consists of a process chamber where the substrate and target are positioned and this chamber is completely sealable. The argon environment creates the plasma in the chamber due the high voltage applied between the electrodes. Titanium nitride was sputtered using high purity (99.995%) titanium target on the aluminium substrate. The approximate time required was 3 h in respect of preconditioning, plasma etching on the substrate and the subsequent coatings.
Surface characterisation by SEM
The surface morphology of titanium deposited aluminium and raw aluminium sample was characterised by SEM analysis. A sample of dimension 25 mm × 25 mm was used for the analysis.
Adhesive joint preparation
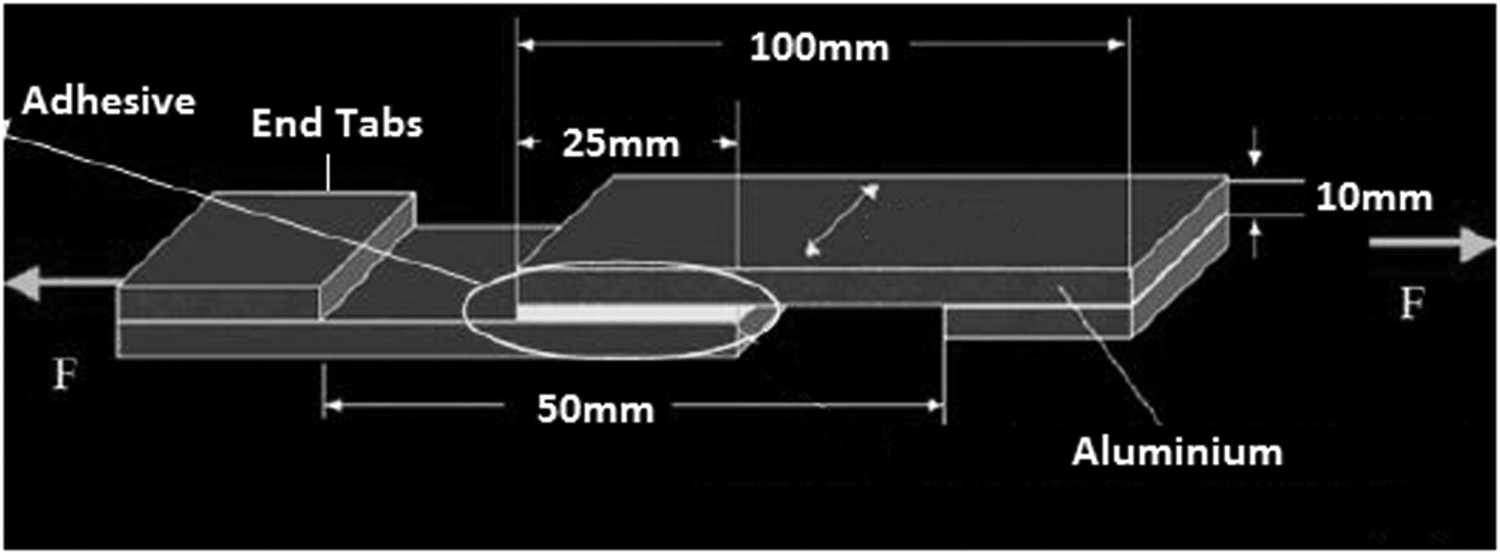
Adhesive joints were prepared with substrates of dimension 100 mm × 25 mm × 5 mm by applying adhesive over an overlap of length 25 mm. Schematic diagram of adhesive joint is revealed in Figure 1. This Figure reveals a standardised method of adhesive bonding of aluminium which is prepared before the lap-shear tensile testing. The rationale which is mentioned in the figure is based on ASTM standard (ASTM D 3164). Zwick/Roell Z010 was used to conduct the testing according to ASTM D 3164, the rate of extension was set as 5 mm min−1. The following types of adhesive joints were prepared and tested under various conditions. These are
Neat adhesive with raw and surface modified aluminium substrate Nanosilica reinforced adhesive with raw and surface modified aluminium substrate. Schematic diagram of adhesive bonded aluminium.
Surface durability
The surface modified sample of substrate of size 25 mm × 25 mm is exposed to harsh environments by immersing it in acidic and saline conditions for 30 days and the effects are studied by energy dispersive spectroscopy (EDS). Acidic condition corresponds to 0.1 N H2SO4 solution (pH-1.2) and saline condition corresponds to 10% NaCl solution.
Corrosion penetration rate (CPR) can be defined as the speed at which the surface in a specific environment deteriorates due to the chemical reaction when it is exposed to a corrosive environment. Thus the rate of decomposition of deposited layer can be quantified by calculating the CPR. CPR is calculated by the following relation,
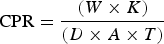
Bond durability
Durability studies of adhesive joints are generally carried out by accelerated aging test under extreme environmental conditions. The joints are tested to determine their strength and to the examine degree of degradation [14].
Adhesive joints with modified and as received aluminium with neat and reinforced adhesives are exposed to high temperature of 250°C with 6 and 18 h at room temperature to study the effects of surface modification and adhesive reinforcement on thermal degradation of bonds.
Results and discussion
Surface characterisation
From the SEM images shown in Figure 2(a,b), it can be observed that the surface of titanium nitride deposited specimen is uniform compared to that of as received aluminium. Aluminium surface in general is susceptible to oxidation and therefore, it undergoes corrosion. Deposition of titanium nitride on aluminium essentially prevents the formation of oxide groups on the aluminium surface. It can be noted from the images that micro-roughness created due to titanium nitride deposition is more continuous compared to that of basic aluminium. Thus surface modification resulted in a uniform substrate with enhanced micro-roughness which would enhance the adhesion process due to efficient mechanical interlocking. Surface modification significantly changes the surface morphology which can be observed with the help of surface imaging techniques. In this study, titanium nitride deposition was employed to modify the surface of the specimen. It was observed from the SEM images that there was a uniform distribution of micro roughness along the surface of the surface-modified specimen which enhances the mechanical interlocking by providing more a effective surface area for the adhesive to fill out and thereby increasing the adhesive bond strength.
SEM image of (a) unmodified aluminium and (b) titanium nitride deposited aluminium.
Lap shear tensile testing
The results from testing of adhesive joints of aluminium are shown in Figure 3. Neat adhesive joints with titanium nitride deposited aluminium showed 20% increase in joint strength in comparison with adhesive joint of unmodified aluminium. In the case of unmodified aluminium as substrate, nanosilica reinforcement enhanced strength by 50% and with titanium nitride deposited aluminium as substrate, nanosilica reinforcement enhanced strength by 90%. Thus it can be inferred from these results that surface modification and adhesive reinforcement have an enhancement effect on the adhesive bond strength. Karjalainen et al. [11] reveal that surface modification enhances the surface energy of aluminium substrate. The enhancement in the polar component of the surface energy results in an overall increase of surface energy of the substrate as to which the molecules of adhesive get attracted to suitable sites on the substrate surface. This enabled a better interaction between the adhesive and substrate thus enhancing the bond strength. The increased interaction led to a cohesive failure i.e. the fracture happened within the adhesive. While the failure in unmodified aluminium joint was due to apparent interfacial failure i.e. the fracture happened at the interface of adhesive and substrate. The fracture surfaces of aluminium are shown in Figure 4. The zones A and B indicate fracture surface of unmodified aluminium and titanium nitride deposited aluminium. This figure clearly reveals that in the case of unmodified aluminium the oxide layer increases brittle characteristics and consequently, adhesive is removed from the substrate. This is because the oxide layer essentially forms hydrogen bond with adhesive and ionic bond with aluminium. Bond energy of ionic bond is significantly higher than hydrogen bonding. Therefore, the aluminium strongly attracts oxygen groups and pulls towards itself and breakage of hydrogen bonds result in poor adhesive bond strength. But in the case of TiN-coated aluminium, the Ti–N–Al bond is formed. In this case, nitrogen interacts with titanium as well as with aluminium and adhesive. Therefore, due to the presence of titanium, aluminium cannot pull out nitrogen towards itself and consequently, there are no breakages of the bond. Therefore, there is enhancement of interfacial adhesion resulting in failure of adhesive bonding of TiN-coated aluminium appears to be cohesive.
Maximum share force of titanium deposited aluminium, copper deposited aluminium and unmodified aluminium. Fractured surfaces of unmodified aluminium indicated by A and titanium nitride deposited aluminium indicated by B.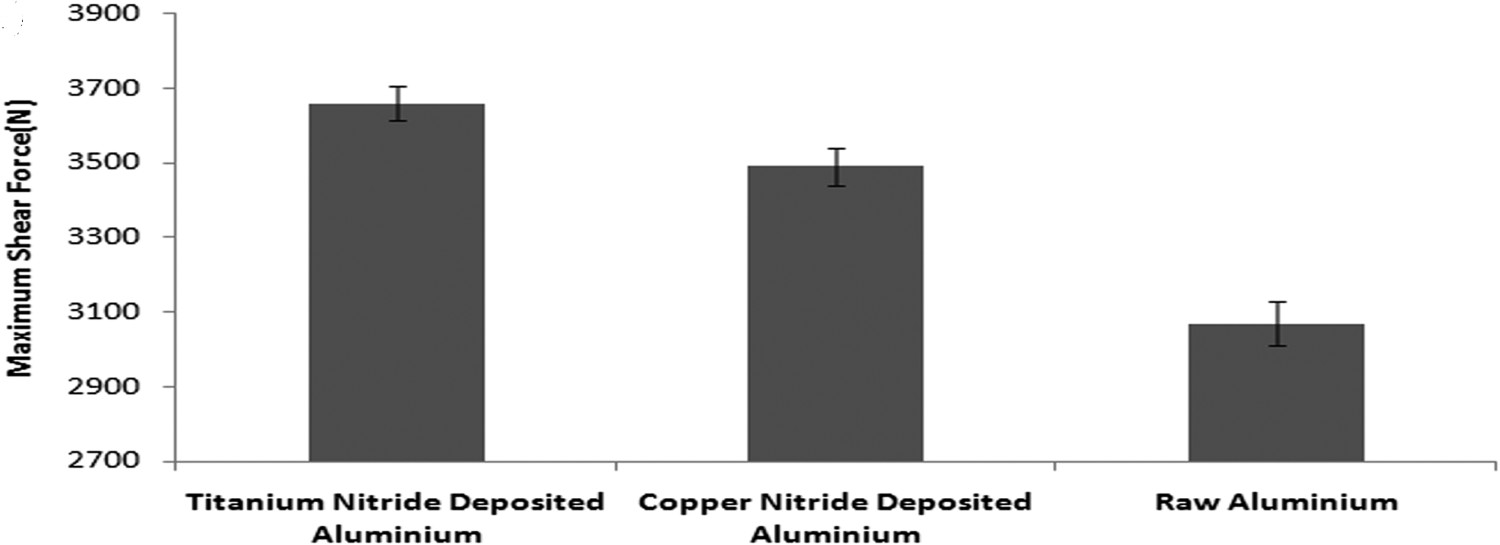

Surface durability
The samples subjected to harsh environments after deposition of titanium nitride over aluminium were examined by EDS. The results obtained from EDS analysis are shown in Figure 5(a–c). The SEM micrograph in the figure indicates that the chemical compounds are exhibited into this specific area of the surfaces of aluminium.
EDS results with SEM micrograph of (a) titanium nitride deposited aluminium, (b) titanium nitride deposited aluminium immersed in NaCl solution and (c) titanium nitride deposited aluminium after immersion in H2SO4 solution.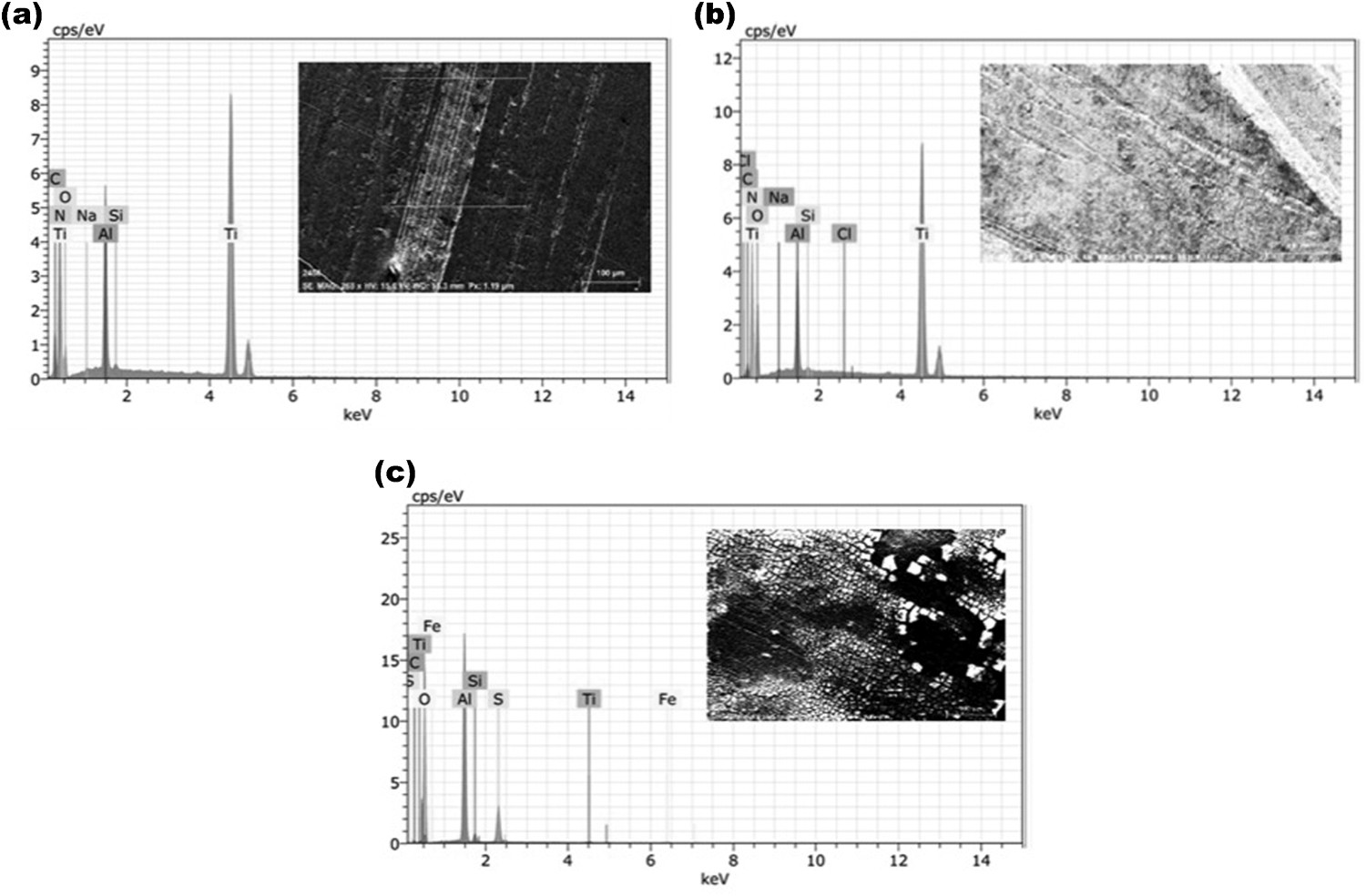
It is clear from the figures that the major element present on the surface by weight was titanium (51.06%) followed by nitrogen (23.14%) and further followed by aluminium (8.32%) thus indicating the presence of titanium nitride coating. When titanium nitride is deposited on aluminium surface essentially prevents the formation of oxide groups resulting in less susceptible to corrosion.
The specimen examined after immersion in NaCl for 30 days through EDS showed a decrease in weight of titanium present on surface. The normalised weight of titanium decreased to 49.11%.
The sample is exposed to the acidic environment (pH-1.2) showed rapid degradation of titanium nitride layer resulting in a normalised weight percentage of only 0.72% titanium in the area analysed. Oxygen, aluminium and sulphur were the most prominent elements present on the surface by weight. This is due to erosion of the titanium nitride surface layer and oxidation of aluminium surface. Corrosion penetration rate (CPR) due to immersion in harsh environments can be quantified using the normalised percentage of titanium and the results obtained showed a CPR of 5.06E−4 mm/year in NaCl while the same for H2SO4 was 0.01306 mm/year. Reinforcement of the adhesive enhances the mechanical and thermal properties of the bond when compared with conventional matrices [15]. The high interface surface area of the nanofillers improved the cohesive properties of adhesives due to the proper stress transfer from adhesive to filler when the loads are applied. Also, the extensive cross linking of epoxies is further enhanced by the silica nanofiller which leads to an improved bond strength compared to neat adhesives.
Aluminium when exposed to air forms oxides which are brittle in nature. Adhesive joints for metallic adherends show complex adhesive/adherend interface, which is composed of thin layers of oxide on substrate and adhesive which affects the joint durability. Thus to ensure joint durability, the surface needs to be modified to exhibit resistance to corrosion and formation of oxides to prevent the formation of brittle oxide interface which is prone to interfacial fracture. From the surface durability analysis conducted, titanium nitride deposition over the sample showed considerable resistance to corrosion even when subjected to harsh environments. This is because of the nitride ion made ionic bond (Ti–N–Al) with both the titanium (Ti) and aluminium (Al). Therefore, it increases the force of attraction in between the surface of aluminium and the adhesive and enhanced the adhesive bond strength under harsh environments.
Bond durability
The effect of adhesive reinforcement on bond degradation due to the simulated environment is given is Figure 6.
Results of lap shear tensile testing of joints subjected to harsh environments.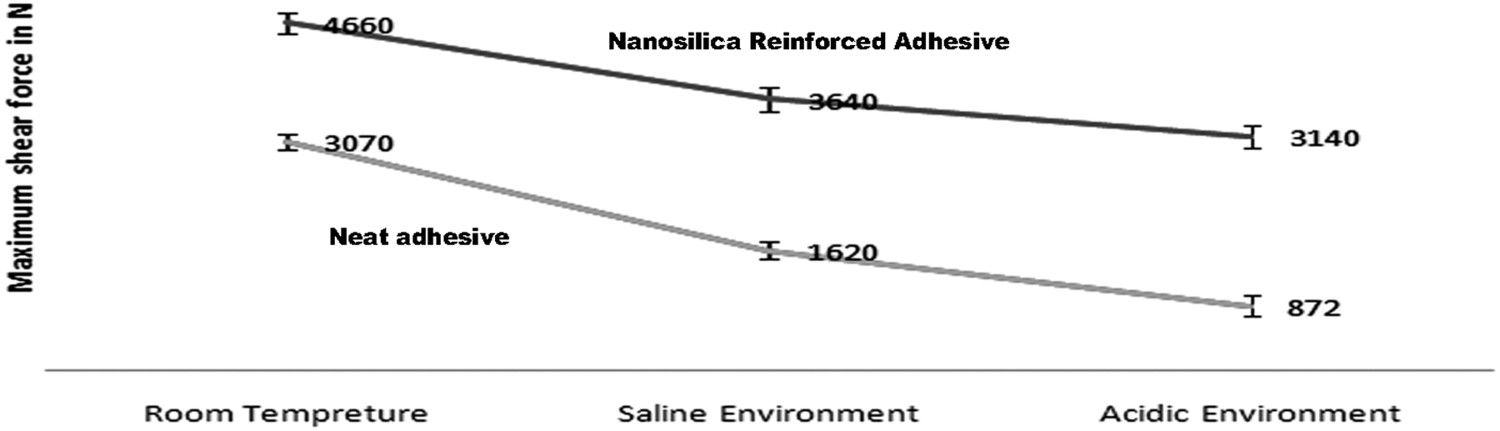
In the case of adhesive joints of unmodified aluminium, the saline environment decreased the strength by 47% while acidic environment contributed to 70% decrease in strength. On comparison, joints with nanosilica reinforced adhesive, experienced 20% decrease in strength in saline environment and 30% decrease in strength in acidic solution.
On comparing the strength of joint with neat adhesive and nanosilica reinforced adhesive after the immersion tests, it can be observed that the reinforced adhesive show better strength. The results reveal that reinforced adhesive joint shows more than two times the strength of neat adhesive joint after exposure of joints to the saline environment and almost four times after exposure of joints to acidic environment. This is because, the nano-reinforced materials works as building block materials into the adhesive. Owing to these nano-materials, the activation energy needs to be very high in order to create damages on the adhesive. Therefore, the reinforcement adhesive shows high bond strength under harsh environments.
Results from lap shear tensile testing after subjecting the prepared joints to thermal cycle test is shown in Figure 7.
Results of lap shear tensile testing of joints subjected to thermal cycles.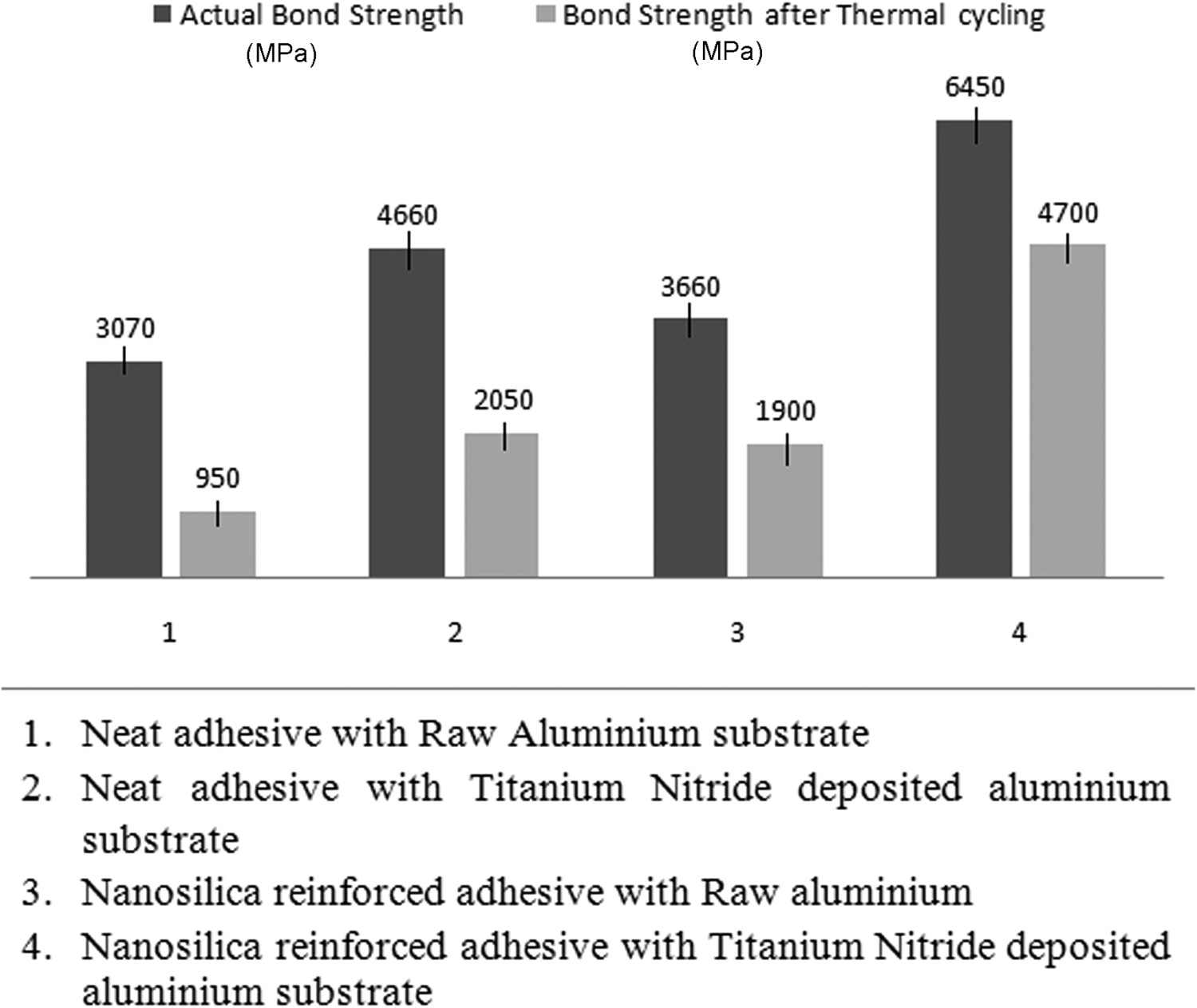
Analysis of the obtained results reveals that joints with raw aluminium as substrate showed a decrease of 65 and 45% strength with neat adhesive and nanosilica reinforced adhesive respectively after thermal cycle test. Similar observation revealed that titanium nitride deposited joints’ strength decreased by 45 and 25% with neat adhesive and nanosilica reinforced adhesive respectively after thermal cycle test. Studies have shown that structural adhesives especially epoxy-based adhesive possess natural tendency to absorb water as the epoxy-based adhesives due to the presence of hydroxyl (OH) groups in their molecular structure, that essentially attract water molecules resulting in the formation of hydrogen bonds [16]. Diffusion of moisture in the polymer matrix is affected by numerous factors, such as environmental humidity, conditioning temperature, and additives in polymers. Nanosilica reinforced adhesive increases the surface area between filler and resin which reduces the moisture ingression by reducing the diffusion coefficient through the adhesive. Also, the rise in the diffusion coefficient has been observed with the increase in temperature [17], which degrades the strength of adhesive. The addition of nanosilica in the adhesive helps in reducing the rate of degradation of the polymer chains due to temperature changes as the inorganic phase acts as a radical sink to absorb heat.
Conclusion
From the above-mentioned result and discussion, it can be concluded that
Physical vapour deposition of titanium nitride on aluminium surfaces essentially increases adhesion characteristics of aluminium. Nano-silica reinforced adhesive increases the adhesion force between nano-silica and resin and also increases the adhesive bond strength. TiN-coated aluminium and nano-silica reinforced adhesive enhances the adhesive bond strength and helps to retention of adhesive bond strength under aggressive environmental conditions.
Therefore, the present work is enlightened to aviation and space industry about the upcoming requirement of component of structural materials and their performance under different environmental conditions.
Footnotes
Disclosure statement
No potential conflict of interest was reported by the authors.
