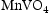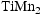Abstract
The success of fusion power requires alloys with good mechanical properties and the ability to withstand extreme thermal and irradiation conditions without prohibitive levels of activation or structural degradation. Body-centred cubic multi-principal component alloys, V–Cr–Mn and Ti–V–Cr–Mn have shown promising high-temperature stability at  . For the alloys to be suitable for use in nuclear fusion reactors, they must be stable across a wide range of temperatures relevant to fusion applications. Here, we assess alloy microstructural stability in these systems following long-term heat treatments at various temperatures. Encouragingly, most of the alloys showed no significant change in microstructure across all temperatures. This contrasts with many other multi-principal component alloys, which develop complex microstructures after exposure to intermediate temperatures.
. For the alloys to be suitable for use in nuclear fusion reactors, they must be stable across a wide range of temperatures relevant to fusion applications. Here, we assess alloy microstructural stability in these systems following long-term heat treatments at various temperatures. Encouragingly, most of the alloys showed no significant change in microstructure across all temperatures. This contrasts with many other multi-principal component alloys, which develop complex microstructures after exposure to intermediate temperatures.
Keywords
Introduction
As global energy demand continues to grow, the world needs to expand its power output through the construction of new energy sources. However, as the UN's Paris Agreement to reduce greenhouse gas emissions came into effect in late 2016, there is now additional pressure to ensure that these new energy sources are not only able to cope with global demand, but also minimise carbon emissions [1]. Renewable sources such as solar and wind have had significant technological improvements in recent years and are more cost-effective than ever, but they are still beset by problems with supply reliability, which can leave grids short of power if there is less sunshine or wind than expected [2].
Nuclear fusion offers an alternative low-carbon energy source with potentially abundant fuel. However, fusion reactors must minimise the amount of harmful radioactive waste they produce in order to be an attractive part of the energy supply ecosystem. Even if designing a reactor which produces no waste requiring deep geological disposal may not be feasible in the short-term [3,4], it is still desirable that elements used in reactor materials should produce only intermediate-level waste that is as short-lived as possible. Such a scenario may allow for fusion waste to be recategorised as separate from fission waste (see Nicholas et al. [5] for further discussion). This criteria necessitates the use of low-activation alloys, made from elements that will not remain radioactive for extended periods of time after exposure to fusion neutrons.
Figure 1 shows the time taken for elements to reach specific radioactivity levels that can be classified as low-level waste according to UK legislation if the element was used in the divertor of a DEMO (the planned full-power demonstration fusion reactor) reactor design for 14 years of pulsed operation [3]. In order to minimise the long-lived waste produced, it is desirable that any new structural materials for fusion reactors only use elements that generate short-lived waste. Certain elements that are commonly used for nuclear structural applications (e.g. Ni or Zr) may be unacceptable from a waste-management perspective.
Time taken to reach low-level waste after 14 years of pulsed operation in the DEMO divertor. Taken from [3].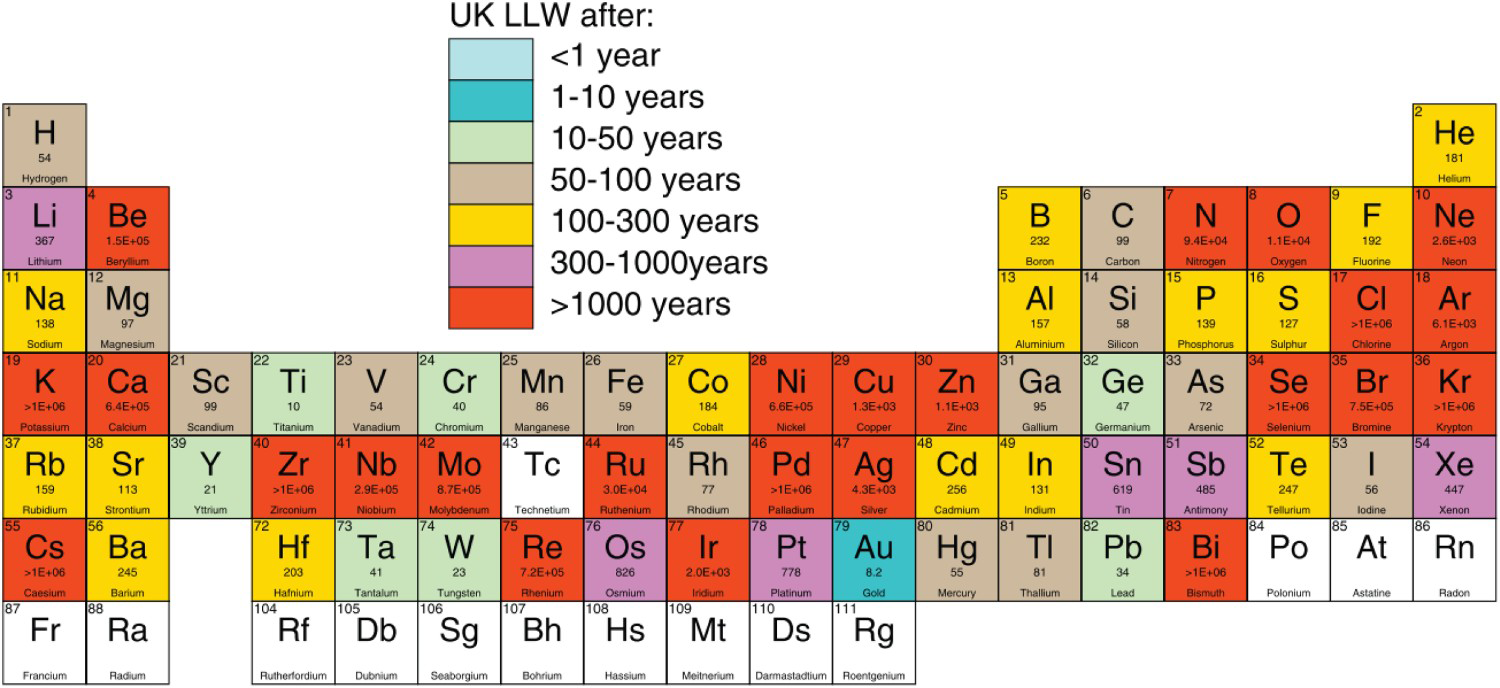
Previous work on the V–Cr–Mn and Ti–V–Cr–Mn alloy systems by the authors has confirmed that a range of compositions are capable of forming a single body-centred cubic (BCC) solid solution phase at  [6]. A single phase is desirable for irradiation resistance (as opposed to a microstructure comprising significant fractions of two or more phases, since differential irradiation damage across them is likely to cause issues), so any alloy used for fusion applications should exist as a stable single phase across the operating temperature range of a fusion reactor. It is thought that alloys with BCC crystal structures are more resistant to irradiation swelling than FCC and HCP alloys, as discussed in Ref. [7]. The operating temperature will be limited by the materials used in the structural components. It is also desirable that the operating temperature is as high as possible to improve thermodynamic efficiency [8]. The microstructure must also be at thermodynamic equilibrium at the desired operating temperature. Many compositionally complex alloys appear stable at a given temperature after short-term exposures, but longer-term treatments often result in decomposition into multi-phase microstructures [9,10]. Many components inside a fusion reactor will remain at elevated temperatures for extended periods of time (although the exact length will depend on reactor design and operation), so it is important that any materials used remain microstructurally stable across these timescales.
[6]. A single phase is desirable for irradiation resistance (as opposed to a microstructure comprising significant fractions of two or more phases, since differential irradiation damage across them is likely to cause issues), so any alloy used for fusion applications should exist as a stable single phase across the operating temperature range of a fusion reactor. It is thought that alloys with BCC crystal structures are more resistant to irradiation swelling than FCC and HCP alloys, as discussed in Ref. [7]. The operating temperature will be limited by the materials used in the structural components. It is also desirable that the operating temperature is as high as possible to improve thermodynamic efficiency [8]. The microstructure must also be at thermodynamic equilibrium at the desired operating temperature. Many compositionally complex alloys appear stable at a given temperature after short-term exposures, but longer-term treatments often result in decomposition into multi-phase microstructures [9,10]. Many components inside a fusion reactor will remain at elevated temperatures for extended periods of time (although the exact length will depend on reactor design and operation), so it is important that any materials used remain microstructurally stable across these timescales.
This study aims to investigate the long-term phase stability of the alloys first presented in the previous work [6] by heat treating the alloys for periods of time up to several weeks at fusion-relevant temperatures: 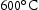 ,
, 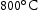 and
and 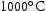 . The resulting microstructures were then characterised using scanning electron microscopy (SEM), transmission electron microscopy (TEM), and X-ray diffraction (XRD) to assess thermodynamic stability. Microhardness was also assessed and correlated with microstructure. The relationship between interstitial content and alloy properties was investigated. Conclusions are drawn regarding the relationship between alloy composition and properties.
. The resulting microstructures were then characterised using scanning electron microscopy (SEM), transmission electron microscopy (TEM), and X-ray diffraction (XRD) to assess thermodynamic stability. Microhardness was also assessed and correlated with microstructure. The relationship between interstitial content and alloy properties was investigated. Conclusions are drawn regarding the relationship between alloy composition and properties.
Method
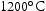 for 100 h, before quenching in water. From these homogenised samples, further sections of the ternary and quaternary alloys were cut for the purpose of heat treatment at
for 100 h, before quenching in water. From these homogenised samples, further sections of the ternary and quaternary alloys were cut for the purpose of heat treatment at 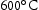 ,
, 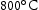 and
and 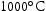 . Three samples were held at each temperature for 1000 h. A V–4Cr–4Ti reference sample was also heat treated using the same procedure at
. Three samples were held at each temperature for 1000 h. A V–4Cr–4Ti reference sample was also heat treated using the same procedure at 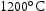 for 100 h.
for 100 h.
Measured alloy compositions (determined using wavelength dispersive spectroscopy) compared with their nominal values.
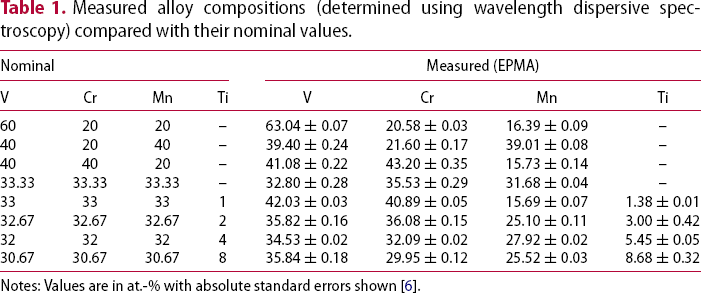
Notes: Values are in at.-% with absolute standard errors shown [6].
CALPHAD results were obtained by using Thermocalc 2021a with a high-entropy alloy database (TCHEA4) and a ferrous alloy database (TCFE10). The ferrous database, although not strictly appropriate for performing calculations on alloys not containing Fe, was used as it was capable of assessing O and S interstitial impurities, which are not included in TCHEA4. All ternary and quaternary alloys were evaluated using both databases between temperatures of 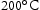 and
and 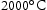 . Measured compositions were assessed, with and without interstitial impurities (C, N, O and S in the TCFE10 database; C and N in the TCHEA4 database) as the presence of interstitial elements is known to be the cause of certain microstructural features in this suite of alloys [6]. Interstitial impurity contents were known for some alloys (see below). For the alloys where interstitial content was not known, an average of the measured values was used as an estimate.
. Measured compositions were assessed, with and without interstitial impurities (C, N, O and S in the TCFE10 database; C and N in the TCHEA4 database) as the presence of interstitial elements is known to be the cause of certain microstructural features in this suite of alloys [6]. Interstitial impurity contents were known for some alloys (see below). For the alloys where interstitial content was not known, an average of the measured values was used as an estimate.
SEM specimens were prepared using standard metallographic techniques. The final polish was performed with a 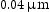 oxide polishing suspension. Back scattered electron (BSE) images and energy-dispersive X-ray (EDX) spectra were obtained using a Zeiss Ultra55 microscope at 10 kV with an Oxford Instruments EDX detector. SEM-EDX quantification was performed using the Oxford Instruments AZTEC software. Analysis of the SEM images was performed using ImageJ software with manual thresholding. Due to the isotropy of the microstructure, area fraction of features was assumed to be equal to the volume fraction. As the thresholding was performed by eye, the errors in the measurements were assumed to be
oxide polishing suspension. Back scattered electron (BSE) images and energy-dispersive X-ray (EDX) spectra were obtained using a Zeiss Ultra55 microscope at 10 kV with an Oxford Instruments EDX detector. SEM-EDX quantification was performed using the Oxford Instruments AZTEC software. Analysis of the SEM images was performed using ImageJ software with manual thresholding. Due to the isotropy of the microstructure, area fraction of features was assumed to be equal to the volume fraction. As the thresholding was performed by eye, the errors in the measurements were assumed to be 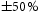 of the stated value. Vickers hardness measurements were taken with a load of 9.8 N and dwell time of 10 s using a Matsuzawa MMT-X indenter. Nine microhardness measurements were made spaced 0.5 mm apart in a three-by-three grid formation. Microindents typically spanned multiple microstructural features and sampled a representative volume of material. Thin foils were prepared via the focused ion beam (FIB) in-situ lift-out method [11] using an FEI Nova Nanolab 600 dual beam SEM/FIB, operated at 30 kV and with a
of the stated value. Vickers hardness measurements were taken with a load of 9.8 N and dwell time of 10 s using a Matsuzawa MMT-X indenter. Nine microhardness measurements were made spaced 0.5 mm apart in a three-by-three grid formation. Microindents typically spanned multiple microstructural features and sampled a representative volume of material. Thin foils were prepared via the focused ion beam (FIB) in-situ lift-out method [11] using an FEI Nova Nanolab 600 dual beam SEM/FIB, operated at 30 kV and with a 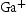 beam current of 10 pA–9 nA. An FEI Talos F200 TEM with a 200 kV X-FEG was used to produce high angle annular dark field (HAADF) images, EDX maps (using Super X EDX with four silicon drift detectors), and selected area electron diffraction (SAED) patterns. TEM-EDX quantification was performed using the Velox software. XRD measurements were performed using a Rigaku Miniflex diffractometer with a Cu source and Ni-foil to filter Cu Kβ radiation. The slit size was 1 mm and scans were performed with 2θ angles between
beam current of 10 pA–9 nA. An FEI Talos F200 TEM with a 200 kV X-FEG was used to produce high angle annular dark field (HAADF) images, EDX maps (using Super X EDX with four silicon drift detectors), and selected area electron diffraction (SAED) patterns. TEM-EDX quantification was performed using the Velox software. XRD measurements were performed using a Rigaku Miniflex diffractometer with a Cu source and Ni-foil to filter Cu Kβ radiation. The slit size was 1 mm and scans were performed with 2θ angles between 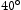 and
and 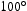 .
.
A selection of samples in the homogenised state were sent for external analysis to determine C, N, O and S content using LECO infrared combustion analysers. The surface of the samples was ground away to ensure no oxide film was present before analysis. Two reference samples of V–4Cr–4Ti have also been included for comparison. One was a highly purified sample in its as-received state from the Culham Centre for Fusion Energy, having undergone refinement processes described in Ref. [12]. The other sample was the as-received V–4Cr–4Ti which was subject to the same homogenisation process as the other alloys.
Results
Impurity analysis
Results of the elemental analysis for carbon, nitrogen, oxygen and sulphur impurities in the homogenised alloys are shown in Table 2. Oxygen is by far the most prevalent impurity element in all of the alloys and appears to be correlated with alloy vanadium content (Figure 2) in the homogenised alloys. Vanadium has a high solubility of oxygen relative to other metals [13,14], which may explain the relationship.
Variation of oxygen concentration with vanadium content. Blue markers are measured in homogenised condition. Orange marker is as-received. Samples at Impurity content of a subset of the alloys measured by LECO analysis.
 -% are V–4Cr–4Ti reference.
-% are V–4Cr–4Ti reference.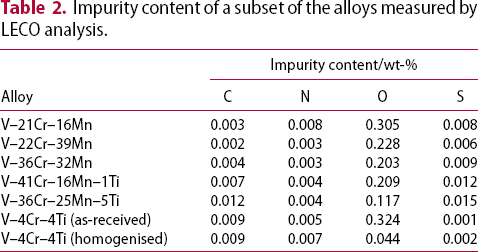
CALPHAD
Using the TCHEA4 database, CALPHAD results predicted the formation of a single BCC phase at high temperatures for all the ternary alloys, as seen in Figure 3. Some sigma phase is predicted to form below Phase proportions of alloys with no interstitial impurities, determined by CALPHAD using the TCHEA4 database. Temperatures of the study are marked with dashed lines. for the higher Mn ternary alloys. In the quaternary alloys, some Laves and sigma phases are predicted for temperatures as high as around
for the higher Mn ternary alloys. In the quaternary alloys, some Laves and sigma phases are predicted for temperatures as high as around  for V–30Cr–26Mn–9Ti. None of the alloys, in their interstitial-free states, are predicted to form anything other than a BCC phase in the range of temperatures studied in this work, with the exception of V–30Cr–26Mn–9Ti.
for V–30Cr–26Mn–9Ti. None of the alloys, in their interstitial-free states, are predicted to form anything other than a BCC phase in the range of temperatures studied in this work, with the exception of V–30Cr–26Mn–9Ti.
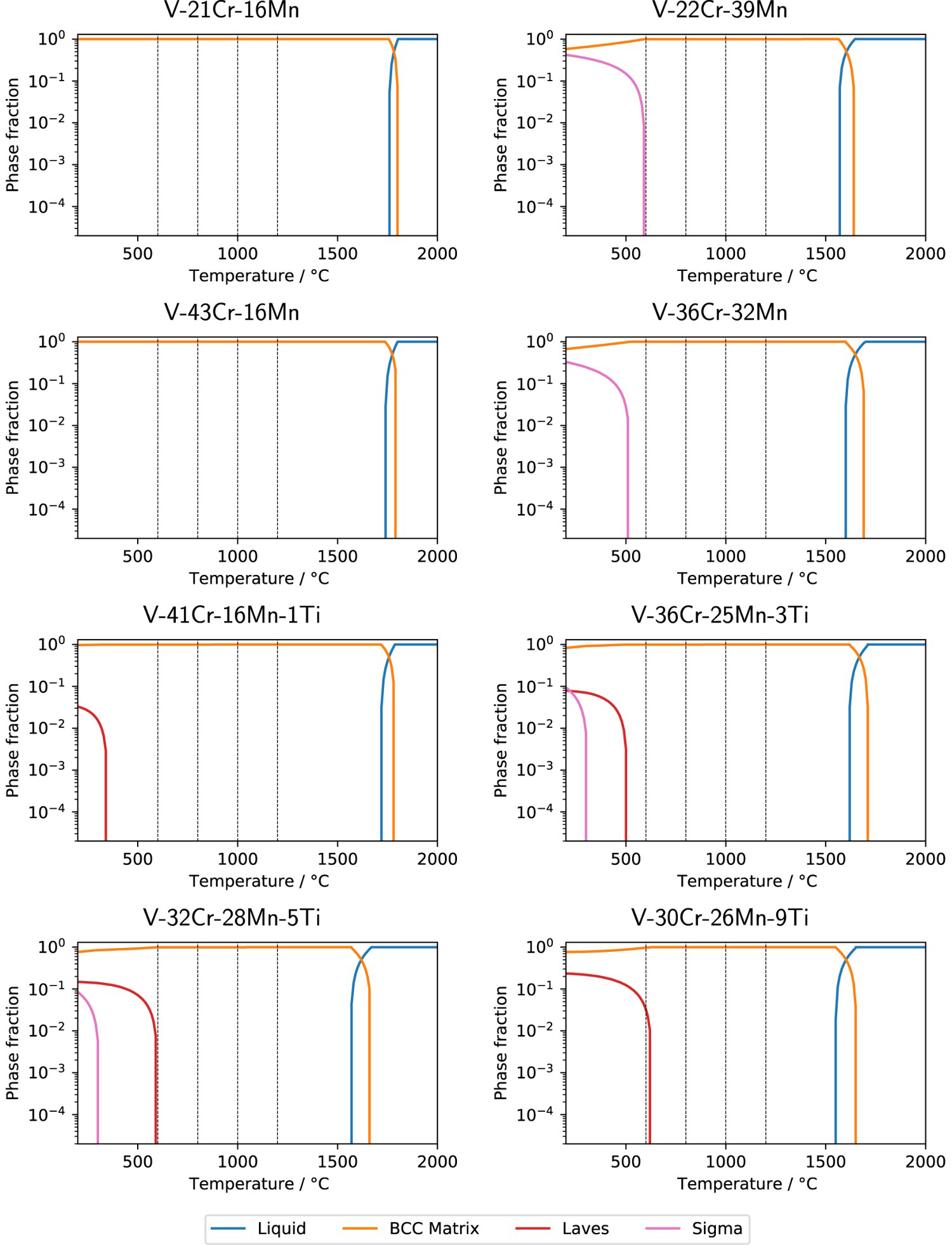
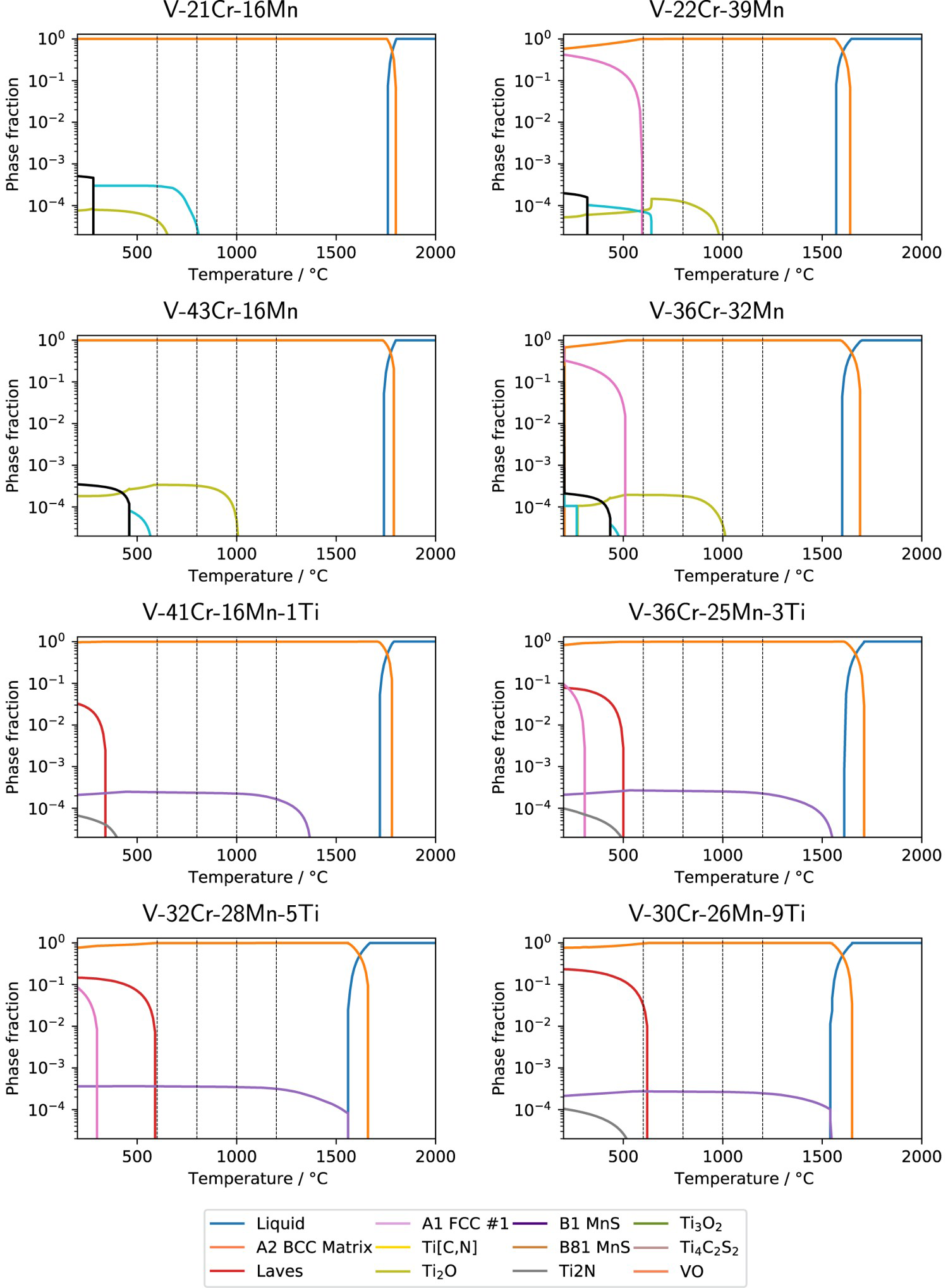
Figure 4 shows how the predicted phases change when including C and N interstitial impurity atoms (O was not included in the database) in the calculation performed using TCHEA4. A wide variety of new phases are predicted in both the ternary and quaternary alloys containing the impurities. When C and N impurities (the concentrations of which were determined by LECO analysis) are included in the calculation, the phases present in the ternary alloys increase in number to include vanadium carbides and nitrides, which were not observed in the samples. A zeta phase is also predicted at lower temperatures, but again outside the range of study. For the quaternary alloys, titanium carbonitrides are predicted in lieu of their vanadium counterparts. Carbides and carbonitrides are predicted in all alloys between Phase proportions of alloys containing both C and N impurities, determined by CALPHAD using the TCHEA4 database. Temperatures of the study are marked with dashed lines. and
and  in sub-percent quantities. Sigma and Laves phases are expected in the same fractions and temperature ranges as their interstitial-free equivalents.
in sub-percent quantities. Sigma and Laves phases are expected in the same fractions and temperature ranges as their interstitial-free equivalents.
The same calculations were also run using TCFE10, a ferrous alloys database (shown in Supp. Figures 1 and 2). The Laves and sigma phases were still predicted to form at roughly the same temperatures and fractions for all alloys, with the only difference in predictions caused by O and S being added to the assessed elements. Including O led to the prediction of VO in the ternary alloys, whereas including S led to very small proportions of MnS and 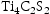 phases being predicted.
phases being predicted.
Microstructure
Figures 5–8 show BSE images of the four ternary V–Cr–Mn alloys after heat treatment. Most of the alloys showed limited microstructural changes from the homogenised state (Figures 5, 7, and 8(a)), which was previously identified to be a single-phase BCC matrix with BSE images of V–21Cr–16Mn (a) homogenised at BSE images of V–22Cr–39Mn (a) homogenised at BSE images of V–43Cr–16Mn (a) homogenised at BSE images of V–36Cr–32Mn (a) homogenised at 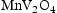 precipitates formed from oxygen impurities [6]. Figure 6 indicated some slight microstructural changes in V–22Cr–39Mn with long, thin precipitates appearing in the heat-treated samples (b)–(d). EDX indicated that these morphologically distinct precipitates had the same chemistry as the precipitates in the other ternary alloys.
precipitates formed from oxygen impurities [6]. Figure 6 indicated some slight microstructural changes in V–22Cr–39Mn with long, thin precipitates appearing in the heat-treated samples (b)–(d). EDX indicated that these morphologically distinct precipitates had the same chemistry as the precipitates in the other ternary alloys.
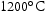
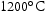 for 100 h (b) held at
for 100 h (b) held at 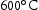 for 1000 h (c) held at
for 1000 h (c) held at 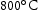 for 1000 h (d) held at
for 1000 h (d) held at 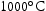 for 1000 h.
for 1000 h.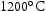
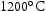 for 100 h (b) held at
for 100 h (b) held at 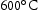 for 1000 h (c) held at
for 1000 h (c) held at 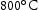 for 1000 h (d) held at
for 1000 h (d) held at 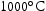 for 1000 h.
for 1000 h.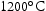
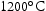 for 100 h (b) held at
for 100 h (b) held at 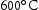 for 1000 h (c) held at
for 1000 h (c) held at 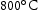 for 1000 h (d) held at
for 1000 h (d) held at 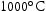 for 1000 h.
for 1000 h.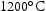
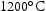 for 100 h (b) held at
for 100 h (b) held at 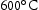 for 1000 h (c) held at
for 1000 h (c) held at 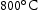 for 1000 h (d) held at
for 1000 h (d) held at 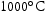 for 1000 h.
for 1000 h.
Figures 9–12 show BSE images of the quaternary alloys heat treated in the same manner as the ternary alloys. Alloys V–41Cr–16Mn–1Ti and V–36Cr–25Mn–3Ti again show a single-phase matrix with small precipitates in all four heat treatments. The only change in microstructure appears to be a slight coarsening of the precipitates in V–41Cr–16Mn–1Ti, with an average particle area of BSE images of V–41Cr–16Mn–1Ti (a) homogenised at BSE images of V–36Cr–25Mn–3Ti (a) homogenised at BSE images of V–32Cr–28Mn–5Ti (a) homogenised at BSE images of V–30Cr–26Mn–9Ti (a) homogenised at 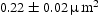 in the homogenised state, versus an average area of
in the homogenised state, versus an average area of 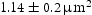 after heat treating. EDX indicated that these precipitates were rich in Ti and presumably the same Ti[C,O,N] precipitates observed in the homogenised state [6].
after heat treating. EDX indicated that these precipitates were rich in Ti and presumably the same Ti[C,O,N] precipitates observed in the homogenised state [6].
 for 100 h (b) held at
for 100 h (b) held at  for 1000 h (c) held at
for 1000 h (c) held at 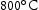 for 1000 h (d) held at
for 1000 h (d) held at 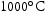 for 1000 h.
for 1000 h.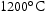
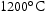 for 100 h (b) held at
for 100 h (b) held at 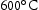 for 1000 h (c) held at
for 1000 h (c) held at 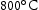 for 1000 h (d) held at
for 1000 h (d) held at 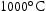 for 1000 h.
for 1000 h.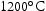
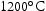 for 100 h (b) held at
for 100 h (b) held at 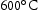 for 1000 h (c) held at
for 1000 h (c) held at 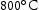 for 1000 h (d) held at
for 1000 h (d) held at 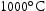 for 1000 h.
for 1000 h.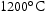
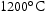 for 100 h (b) held at
for 100 h (b) held at 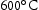 for 1000 h (c) held at
for 1000 h (c) held at 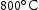 for 1000 h (d) held at
for 1000 h (d) held at 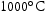 for 1000 h.
for 1000 h.
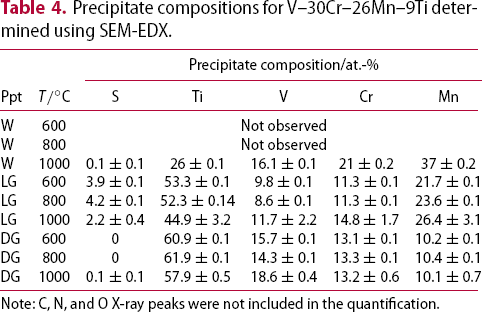
Alloys V–32Cr–28Mn–5Ti and V–30Cr–26Mn–9Ti showed more significant microstructural changes with respect to heat treatment temperature. Three different precipitates are observed: one denoted white (W), which only appears in the TEM-EDX map and diffraction patterns from V–30Cr–26Mn–9Ti held at Precipitate compositions for V–32Cr–28Mn–5Ti determined using SEM-EDX. C, N, and O X-ray peaks were not included in the quantification. Precipitate compositions for V–30Cr–26Mn–9Ti determined using SEM-EDX. Note: C, N, and O X-ray peaks were not included in the quantification.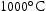 heat treatments, and two other types, denoted dark grey (DG) and light grey (LG), which appear at all three ageing temperatures (Figures 11 and 12). Tables 3 and 4 show the results of SEM-EDX quantification of the precipitates in alloys V–32Cr–28Mn–5Ti and V–30Cr–26Mn–9Ti respectively. SEM- and STEM-EDX (shown in Figure 13) indicated the white precipitates were enriched in Ti and Mn relative to the matrix. The dark grey precipitates were chemically similar to those observed in the lower at.%Ti alloys. The light grey precipitates were also enriched in Ti, but also showed an increase in N and S relative to the matrix.
heat treatments, and two other types, denoted dark grey (DG) and light grey (LG), which appear at all three ageing temperatures (Figures 11 and 12). Tables 3 and 4 show the results of SEM-EDX quantification of the precipitates in alloys V–32Cr–28Mn–5Ti and V–30Cr–26Mn–9Ti respectively. SEM- and STEM-EDX (shown in Figure 13) indicated the white precipitates were enriched in Ti and Mn relative to the matrix. The dark grey precipitates were chemically similar to those observed in the lower at.%Ti alloys. The light grey precipitates were also enriched in Ti, but also showed an increase in N and S relative to the matrix.
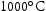
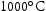 for 1000 h. (a) Bright field image, (b) Ti map, (c) V map, (d) Cr map, (e) Mn map, (f) C map, (g) N map, (h) O map, (i) S map, (j) diffraction pattern of matrix, (k) diffraction pattern of region DG and (l) diffraction pattern of region W.
for 1000 h. (a) Bright field image, (b) Ti map, (c) V map, (d) Cr map, (e) Mn map, (f) C map, (g) N map, (h) O map, (i) S map, (j) diffraction pattern of matrix, (k) diffraction pattern of region DG and (l) diffraction pattern of region W.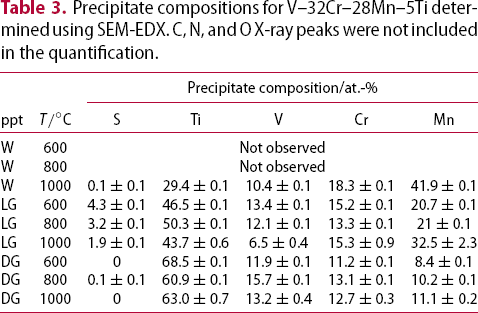
SAED was utilised to identify these new precipitates. Figure 13 shows TEM-EDX maps of a FIB foil taken from a region of alloy V–30Cr–26Mn–9Ti treated for 1000 h at 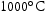 that contained all three precipitates. TEM-EDX was used to match regions of the foil to their respective precipitates (Figure 13(a–f)). Electron diffraction patterns for W and DG precipitates and the matrix are shown in Figure 13. The matrix was BCC (
that contained all three precipitates. TEM-EDX was used to match regions of the foil to their respective precipitates (Figure 13(a–f)). Electron diffraction patterns for W and DG precipitates and the matrix are shown in Figure 13. The matrix was BCC (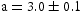 Å), dark grey was FCC (
Å), dark grey was FCC (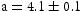 Å), and white was hexagonal (
Å), and white was hexagonal (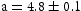 Åand
Åand 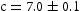 Å). The diffraction pattern for the light grey precipitate could not be indexed.
Å). The diffraction pattern for the light grey precipitate could not be indexed.
A FIB foil taken from V–32Cr–28Mn–5Ti heat treated for 1000 h at 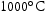 used the same approach to identify the precipitates and found similar chemistries and lattice parameters. Furthermore, the diffraction pattern of a light grey precipitate was indexed as FCC with a lattice parameter of approximately
used the same approach to identify the precipitates and found similar chemistries and lattice parameters. Furthermore, the diffraction pattern of a light grey precipitate was indexed as FCC with a lattice parameter of approximately 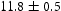 Å. Difficulty obtaining good diffraction patterns contributed to the low precision of this measurement.
Å. Difficulty obtaining good diffraction patterns contributed to the low precision of this measurement.
FIB foils of alloys V–32Cr–28Mn–5Ti and V–30Cr–26Mn–9Ti heat treated at 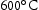 and
and 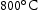 were also investigated. Both light grey and dark grey precipitates had similar compositions and identical lattice parameters to their counterparts in the
were also investigated. Both light grey and dark grey precipitates had similar compositions and identical lattice parameters to their counterparts in the 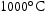 samples. Interestingly, the white phase was observed in the V–30Cr–26Mn–9Ti samples in the form of sub-micron precipitates that were found adjacent to light grey particles, despite not being observed under SEM.
samples. Interestingly, the white phase was observed in the V–30Cr–26Mn–9Ti samples in the form of sub-micron precipitates that were found adjacent to light grey particles, despite not being observed under SEM.
Overall, the lattice parameters of the white precipitates suggested a C14-Laves phase, and the chemistries implied it was 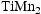 with significant deviations from ideal stoichiometry (which is observed for this phase in the binary Ti–Mn system [15]). The dark grey phase comprised mostly of Ti and also showed high quantities of N and O which were not included in the quantification due to large errors in fitting the X-ray peak data (see Supp. Figure 3), and had a lattice parameter consistent with Ti–[C,O,N] precipitates observed in the homogenised alloys [6]. The light grey phase contained high amounts of Ti, N (not quantified) and S, with an FCC lattice (
with significant deviations from ideal stoichiometry (which is observed for this phase in the binary Ti–Mn system [15]). The dark grey phase comprised mostly of Ti and also showed high quantities of N and O which were not included in the quantification due to large errors in fitting the X-ray peak data (see Supp. Figure 3), and had a lattice parameter consistent with Ti–[C,O,N] precipitates observed in the homogenised alloys [6]. The light grey phase contained high amounts of Ti, N (not quantified) and S, with an FCC lattice ( Å). The relatively high proportions of Ti and N and the lattice parameter were similar to nitrides found in the Ti–Ni–Al–N system [16]. The XRD patterns of the ternary alloys showed only bcc reflections for all alloys and heat treatments (see Supp. Figure 4).
Å). The relatively high proportions of Ti and N and the lattice parameter were similar to nitrides found in the Ti–Ni–Al–N system [16]. The XRD patterns of the ternary alloys showed only bcc reflections for all alloys and heat treatments (see Supp. Figure 4).
Microhardness
Microhardness data are presented in Figure 14, which shows no apparent relation between heat treatment conditions and hardness. There are significant variations between the various heat treatments of a single alloy, which may be explained by differences in interstitial content, which is known to have a noticeable effect on the mechanical properties of vanadium alloys [17].
Variation in Vickers hardness after heat treatment for 1000 h at the temperatures shown. Error bars show the standard deviation in the measurements taken.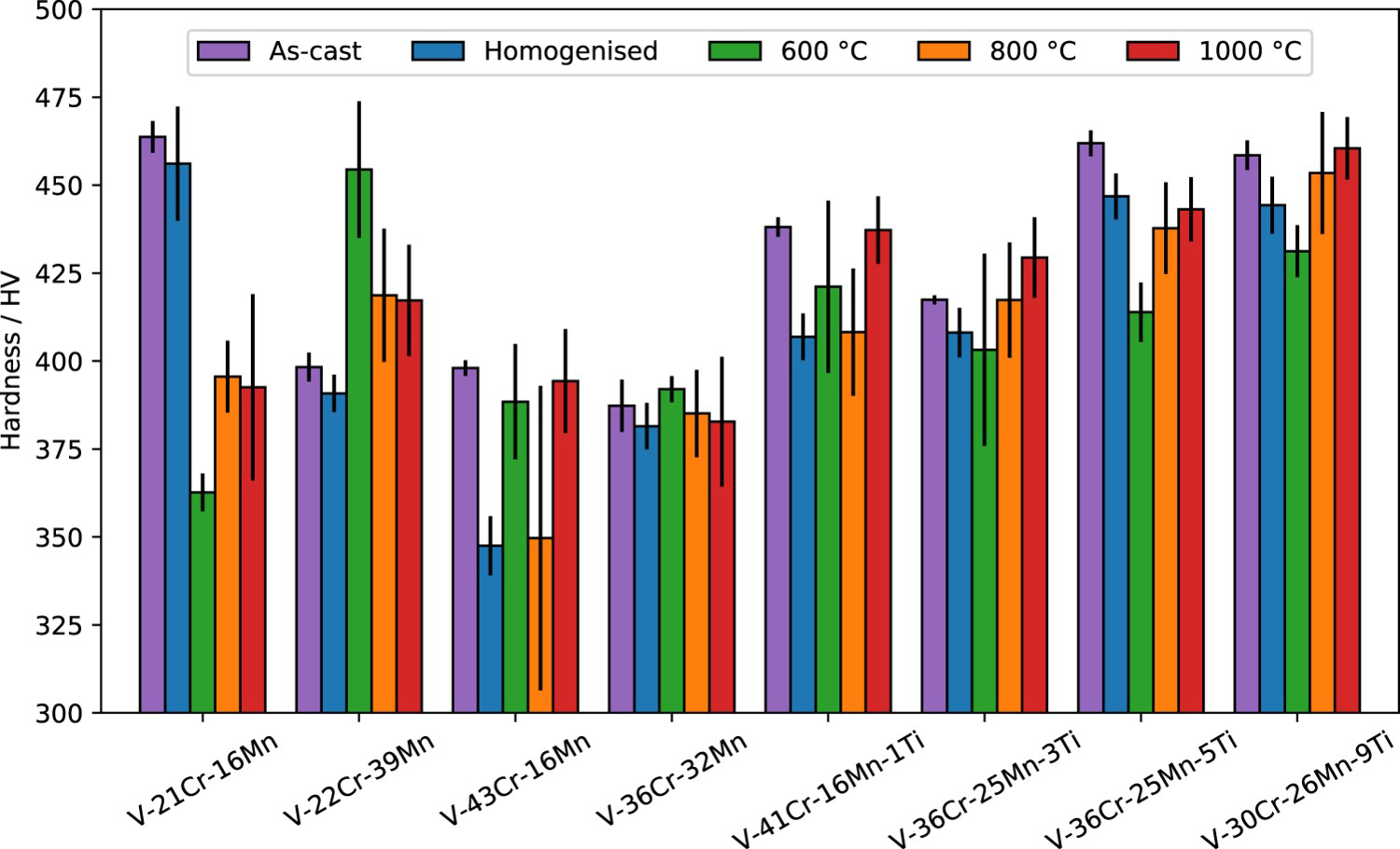
Discussion
Microstructure and CALPHAD predictions
Ternary alloys
The results indicate that no significant microstructural evolution of the alloys occurred during heat treatment at the temperatures studied for times of up to 1000 h in five of the eight compositions studied. Out of the ternary alloys, only V–22Cr–39Mn showed a noticeable change in microstructure. In this alloy, round particles appear at grain boundaries, whereas the long, thin precipitates appear inside grains. However, these morphologically distinct precipitates do not change in character between heat treatments and are suggested to be in the same phase as the round precipitates in the same alloy and the other ternary alloys. Any precipitate evolution might be problematic from a fusion perspective, rendering V–22Cr–39Mn unsuitable for such applications. However, as the precipitates are caused by interstitial impurities, control of these elements may prevent the observed microstructural changes from occurring.
In the case of interstitial-free ternary alloys, the TCHEA4 database successfully predicted that a single BCC phase will be present across all temperature ranges (Figure 3). In TCHEA4, the sigma phase is predicted to precipitate at lower temperatures (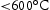 ) in alloys with higher Mn content, these temperatures are outside the range of this study. The main discrepancy between the TCHEA4 predictions and observed microstructures are the
) in alloys with higher Mn content, these temperatures are outside the range of this study. The main discrepancy between the TCHEA4 predictions and observed microstructures are the 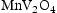 precipitates. As the TCHEA4 database does not include oxygen, future studies employing a modelling approach capable of reproducing these oxide phases would be valuable. The TCFE10 database which did include O in the calculations was also unable to predict the existence of this phase.
precipitates. As the TCHEA4 database does not include oxygen, future studies employing a modelling approach capable of reproducing these oxide phases would be valuable. The TCFE10 database which did include O in the calculations was also unable to predict the existence of this phase.
Quaternary alloys
The two alloys with lower Ti content (V–41Cr–16Mn–1Ti and V–36Cr–25Mn–3Ti) displayed good thermal stability. The only microstructural change observed was a coarsening of the Ti–[C,O,N] precipitates in V–41Cr–16Mn–1Ti. The fact that these precipitates are still present at 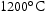 and
and 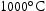 in all Ti-containing alloys is somewhat notable as equivalent precipitates in V–4Cr–4Ti alloys will dissolve at these temperatures [18]. TCHEA4 predictions indicate this may be due to Mn acting as a stabiliser for these particles, as dissolution temperature seems to increase with Mn content (Figure 4). TCHEA4 results for V–4Cr–4Ti predicted a much lower dissolution temperature of around
in all Ti-containing alloys is somewhat notable as equivalent precipitates in V–4Cr–4Ti alloys will dissolve at these temperatures [18]. TCHEA4 predictions indicate this may be due to Mn acting as a stabiliser for these particles, as dissolution temperature seems to increase with Mn content (Figure 4). TCHEA4 results for V–4Cr–4Ti predicted a much lower dissolution temperature of around 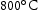 for the Ti–[C,N] precipitates (see Supp. Figure 5) relative to the alloys studied here.
for the Ti–[C,N] precipitates (see Supp. Figure 5) relative to the alloys studied here.
The two alloys with the higher Ti content (V–32Cr–28Mn–5Ti and V–30Cr–26Mn–9Ti) did exhibit evidence of new phases, such as the 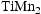 Laves phase and possibly a nitride, forming after heat treatment at fusion-relevant temperatures. Another study examining the TiVCrMnFe system found that increased Ti content promoted the formation of the Laves phase, with 20 at.-% Ti leading to a Laves phase fraction of greater than 50% [19]. The appearance of these phases only at higher Ti concentrations is not necessarily an issue from an alloy design perspective. Low Ti content (
Laves phase and possibly a nitride, forming after heat treatment at fusion-relevant temperatures. Another study examining the TiVCrMnFe system found that increased Ti content promoted the formation of the Laves phase, with 20 at.-% Ti leading to a Laves phase fraction of greater than 50% [19]. The appearance of these phases only at higher Ti concentrations is not necessarily an issue from an alloy design perspective. Low Ti content (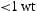 - %) has been reported to be sufficient for suppressing void swelling in vanadium fusion alloys [20]. Due to variations in the alloy composition from the arc-melting process, the Mn content of the Laves-forming quaternaries is higher than those that don't form Laves phases. As the Laves phase appears to be
- %) has been reported to be sufficient for suppressing void swelling in vanadium fusion alloys [20]. Due to variations in the alloy composition from the arc-melting process, the Mn content of the Laves-forming quaternaries is higher than those that don't form Laves phases. As the Laves phase appears to be 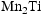 , it is likely that this increased Mn content is an additional driver for the formation of the Laves phase. For V–30Cr–26Mn–9Ti aged at
, it is likely that this increased Mn content is an additional driver for the formation of the Laves phase. For V–30Cr–26Mn–9Ti aged at 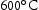 and
and 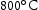 , a probable reason for the observation of the Laves phase in the TEM and not in the SEM is that the particles that were detected in the FIB foil were less than a micron in size and were either adjacent to or partially surrounded by larger light grey type precipitates. There appears to be a connection between the Laves phase and the unknown nitrides. They are compositionally similar (showing high Ti and N content relative to the matrix) and often appear adjacent to each other if present in the same alloy.
, a probable reason for the observation of the Laves phase in the TEM and not in the SEM is that the particles that were detected in the FIB foil were less than a micron in size and were either adjacent to or partially surrounded by larger light grey type precipitates. There appears to be a connection between the Laves phase and the unknown nitrides. They are compositionally similar (showing high Ti and N content relative to the matrix) and often appear adjacent to each other if present in the same alloy.
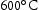 in V–30Cr–26Mn–9Ti). However, in practice, the Laves phase was present at
in V–30Cr–26Mn–9Ti). However, in practice, the Laves phase was present at 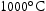 for both V–32Cr–28Mn–5Ti and V–30Cr–26Mn–9Ti. The calculations also overestimate the fraction of Laves phase present in both alloys where it is found. It also forms in very small quantities at
for both V–32Cr–28Mn–5Ti and V–30Cr–26Mn–9Ti. The calculations also overestimate the fraction of Laves phase present in both alloys where it is found. It also forms in very small quantities at 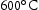 and
and 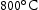 in V–30Cr–26Mn–9Ti. Given that O is not included in TCHEA4, it is fair to say that the appearance of precipitates is correctly predicted in the CALPHAD calculations with interstitials (Figure 4) if the predicted Ti[C,N] phase is treated analogously to the Ti[C,O,N] phase seen in practice. Quantitatively, the volume fraction of precipitates measured from SEM micrographs was around 1%. This is divergent from both the HEA4 database and TCFE10 predictions, which estimate fractions of Ti-rich precipitates to be below 0.1%, but this is to be expected given that neither database predicted the precipitate observed in reality. Table 5 summarises the phases present in each of the alloy heat treatments and their approximate volume fractions.
in V–30Cr–26Mn–9Ti. Given that O is not included in TCHEA4, it is fair to say that the appearance of precipitates is correctly predicted in the CALPHAD calculations with interstitials (Figure 4) if the predicted Ti[C,N] phase is treated analogously to the Ti[C,O,N] phase seen in practice. Quantitatively, the volume fraction of precipitates measured from SEM micrographs was around 1%. This is divergent from both the HEA4 database and TCFE10 predictions, which estimate fractions of Ti-rich precipitates to be below 0.1%, but this is to be expected given that neither database predicted the precipitate observed in reality. Table 5 summarises the phases present in each of the alloy heat treatments and their approximate volume fractions.
Alloy heat treatments and the volume fraction present within them.

 denotes phases where quantification was difficult due to small differences in BSE contrast. Error in measurements is assumed to be
denotes phases where quantification was difficult due to small differences in BSE contrast. Error in measurements is assumed to be 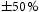 of the stated value.
of the stated value.
Microhardness and impurities
As discussed in a previous work [6], in the quaternary, Ti-containing alloys, the solid solution strengthening from Ti may outweigh the softening effect of gettering interstitials, leading to an overall hardening of these alloys relative to the ternary system. V–21Cr–16Mn is an exception, as this alloy was harder than all of the quaternary alloys in the as-cast state. It also has the highest concentration of interstitials, shown in Table 2. Interstitial impurities are known to harden vanadium alloys [17], so this is a probable source of the anomalous hardness. The presence of new phases in alloys V–32Cr–28Mn–5Ti and V–30Cr–26Mn–9Ti does not appear to have had any systematic effect on the microhardness properties of these alloys.
The source of these interstitial impurities present in these alloys was investigated using the V–4Cr–4Ti reference sample. Comparing the highly purified V–4Cr–4Ti sample to one that had been homogenised under the same conditions as the alloys studied here reveals an order of magnitude increase in the oxygen content. Therefore, as might be expected, the homogenisation procedure and subsequent heat treatments (which used the same method of encapsulation followed by quenching) are likely to have introduced additional impurities to the alloys, potentially altering their properties despite efforts to mitigate against this.
Fusion applications
As seen in Section ‘Microstructure & CALPHAD predictions’, the alloy systems seem to possess good thermal stability at a range of fusion-relevant temperatures. Only when Ti is present above a few at.-% or Mn is present above equiatomic proportions is any sort of microstructural evolution observed. Nevertheless, many of the changes seen occur in phases that arise as a consequence of the alloys interstitial content rather than their principal constituents. Of course, phases that are produced by interstitials are not inconsequential and are utilised in most candidate fusion structural materials, but their presence is well controlled through processing routes.
In order to verify if these materials have real potential as structural materials for fusion applications, there are a range of other factors that need consideration. Foremost of which will be the mechanical properties of these alloys. Although not quantitatively determined, the brittleness observed when handling and preparing these alloys would limit their usefulness in structural applications. It is not yet known if this brittleness is inherent to such a highly alloyed refractory BCC material, or if interstitials play a significant role. Furthermore, the effects of radiation on these alloys must be assessed. Some V-based alloys have exhibited significant and undesirable irradiation swelling, but this seems to be mitigated by the addition of Ti [21,22].
As previously mentioned, the neutronic properties of the majority elements of these alloys render them acceptable from a long-term recycling and disposal standpoint. However, there are other safety issues pertaining to element choice in fusion reactors that shall be touched upon here. Analyses by Piet et al. [23,24] gave an overall score for each element in the periodic table based on structural and biological safety considerations in the event of an accident. According to those studies, Mn is the element contained in these alloys that is of greatest concern. This is due to its particularly high short-term decay properties, which could pose a radiological hazard from the dose released if an accident were to occur. Furthermore, the large temperature rises accompanying the decay heat could lead to further exposure to harmful materials. However, the same analysis reveals that elements such as W (already used extensively in fusion components) score equally as poorly, so such findings do not necessarily preclude the use of an element in a reactor.
Conclusions
V–Cr–Mn alloys were found to possess thermal phase stability across a range of fusion-relevant temperatures, from CALPHAD predictions were generally accurate for most compositions, predicting a predominantly BCC microstructure across the range of temperatures investigated. It also correctly predicted the higher temperature of dissolution for Ti–[C,N] precipitates in these alloys relative to V–4Cr–4Ti. Laves phase appears in some Ti–V–Cr–Mn alloys after ageing at Another phase appeared at all three ageing temperatures studied in the higher Ti concentration alloys that is enriched in N, S, and Ti. No consistent changes in hardness measurements were observed, with many alloys displaying little change at all Interstitial impurity content varied considerably between samples. This is thought to be introduced in the homogenisation and heat treat processes, and may have an effect on alloy properties. Most alloys were stable in fusion-relevant temperature regimes, with no microstructural evolution observed.
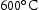 to
to 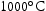 .
.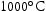 , which is not predicted by Thermocalc CALPHAD databases. This behaviour is suppressed in alloys with reduced Ti content.
, which is not predicted by Thermocalc CALPHAD databases. This behaviour is suppressed in alloys with reduced Ti content.
Footnotes
Acknowledgments
The authors acknowledge the use of the Department of Materials X-ray Diffraction Suite at the University of Manchester and for the technical support, advice and assistance provided by Dr John E. Warren and Gary Harrison. The authors would also like to Dr Mark Gilbert for fruitful discussions regarding the activation properties of elements.
Disclosure statement
No potential conflict of interest was reported by the author(s).