Abstract
In this work, we report on the controlled change of the wetting behaviour of silica thin films deposited by a reactive e-beam evaporation. The as-deposited coated surfaces were treated with acetone solvent using an ultra-sonic bath followed by characterising their contact angles. As confirmed by a repeated contact angle measurement, the silica thin films changed their wetting behaviour from superhydrophilic to hydrophilic which we attributed to the creation of OH groups and hydroxylation process. X-ray photoelectron spectroscopy analysis suggested a SiO x stoichiometry of SiO1.85 for the non-treated samples and SiO1.91 for the solvent-treated ones. Such promising results pave the way to develop a cost-effective and efficient anti-soiling and self-cleaning coating for a large-scale application.
Keywords
Introduction
It is well known that hydrophilic thin films with a contact angle of above 60° can be used in anti-adhesion, antibacterial, and anti-soiling technologies [1-6]. On the other hand, hydrophilic materials are essential in developing water membranes and shifting the boiling heat point for miscellaneous applications [7,8]. It is highly desirable to engineer surface properties with a cost-effective method to develop hydrophilic anti-dust and anti-reflection coatings for the geographical area with extensive rainfall. Mostly, hydrophilicity can be formulated using high surface energy materials to resist dust accumulation and due to soluble properties, such layers become dominant environmental friendly materials. The wetting behaviour of such layers resulted in reducing surface adhesion energy. Due to the adapted cost-effective and controlled tuning of surface through solvent treatment, it is believed that such hydrophilic surfaces will be the best competitors for developing anti-soiling coatings, for example in the space industry, where it is substantially required to adapt the repelling technology. Moreover, flexible along with cheap solvent treatment methods will be highly beneficial for large-scale photovoltaic plants.
Generally, wettability strongly depends on surface chemical bonding and patterning such as the surface roughness of the material. Until now, many works have been experimentally validated in terms of exploring the super-hydrophobicity or super-hydrophilicity nature by tuning surface roughness [9-13]. In some previous works, 2D materials like carbon nanotubes were studied to understand such a changing behaviour. However, no works have been reported on acetone treated silica thin films in detail, which is a crucial and a very promising material to develop anti-soiling coating. Basically, silica (SiO2) is bonded covalently with four oxygen atoms in a tetrahedral coordination in a 3D network. Though silica layers show primitive crystallite structure, local structure in between Si and O are always the same [14,15]. The effects of acetone sonication of silica layers were immediate since contact angle measurements confirm that high hydrophobicity occurs which can be attributed to the OH groups. It is mainly due to the absorption of organic molecules on such metal oxide surfaces [16-18]. Certainly, another root cause could be the direct hydroxylation process resulting in altering surface roughness. It is well studied and the theories of Wenzel and Cassie–Baxter are applied to understand such a mechanism [19,20]. In our work, we have used acetone which is a solvent substance to alter the wettability properties. Acetone is an organic material with the formula of (CH3)2CO [21]. It is a widely used solvent, as acetone cyanohydrin, and a precursor to methyl methacrylate [21]. E-beam evaporated silica layers have shown roughened properties with increasing oxygen flow during the deposition and without any solvent treatment. In this work, we demonstrate the change of wettability using acetone solvent with ultra-sonication for one minute only and without any structural change as confirmed by X-ray photoelectron spectroscopy. In this work, the main goal is to demonstrate that solvent treatment confirms the wettability tuning regardless of the substrates types. Since the main target is to develop anti-soiling coating, glass substrates are the best material for practical and large-scale application. Tailoring silica coating with controllable wetting properties provides a cost-effective way to modify the surface wettability.
Methodology
Soda-lime glass (1″ × 3″), FTO coated soda-lime glass (SLG) (1″ × 1″), and Si wafer (4″) were used as substrates for the growth of the silica thin films. All substrates were first cleaned in an ultra-sonic bath with acetone, isopropanol, and deionised water for 10 minutes each. Inert nitrogen gas was used to dry the samples. The thin film deposition was performed using a Denton E-Beam Evaporation Tool™ with deposition pressure of 2e − 5, 7e − 5, 9e − 5, and 2e − 4 Torr pressures under oxygen pressure of 5–20 sccm with substrate rotation. The purity of oxygen gas was 99.995%. SiO2 pellets (Kurt J Lesker) with a purity of 99.995% were used. All films were grown at a stable process temperature of 100°C with the same deposition rate of 1 Å s−1. Oxygen flow rate was varied accordingly to adjust the deposition pressure. Thickness and 3D roughness measurements were measured with a Dektak stylus profilometer. The contact angle measurements were performed by the Kruss DSA25™ tool to assess the wettability (hydrophilicity/hydrophobicity) of the silica films. The optical properties such as transmission (%) were investigated using the UV-Vis spectroscopy (Perkin Elmer™ Lambda 1050 UV/VIS/NIR) in the range of 250–1000 nm. Field emission scanning electron microscopy (FESEM) was employed to investigate the morphology of the oxides using JEOL 7610. Structural properties were investigated by XPS Escalab 250Xi (Thermo Fisher Scientific™). X-ray photoelectron spectroscopy (XPS) spectra analysis and fitting were conducted using Avantage software.
Results and discussions
Hydrophilic properties of the silica thin films
The Drop Shape Analyzer (Kruss DSA25) contact angle tool has been used by varying the syringe liquid volume between 1 and 2 µL and speed from 1.5 to 2.66 µL s−1. In addition, for each sample, we measured from three different spots to make sure that there is no large deviation in terms of angle measurements [22,23]. Figure 1 shows the contact angle measurement of silica layers on three different substrates, namely soda-lime glass, FTO coated glass, and Si-substrates. First, the measurement was carried out on bare reference substrates. The as-deposited samples were measured using the contact angle tool as shown in Figure 1. The samples grown at 2e − 4 Torr (O2: 20 sccm) under oxygen-rich process have shown super-hydrophilic behaviour where the angles varied from 0° to 1.5° for all three different substrates. The samples were sonicated for 5 minutes with acetone in an ultra-sonic bath after 1 day. Interestingly, the surface of the silica layers turns to be hydrophilic with the increment of water droplet and surface interface to 78° (glass), 26° (FTO), and 60° (Si) (Figure 1). The highest hydrophobicity was measured with 78° for the samples grown on soda-lime glass and the lowest was on the FTO coated glass substrate. Generally, silica-based oxide layers are highly hydrophilic surfaces due to the non-polarity of the surface energy [13]. Treatment using acetone solvent results in hydroxylation. In general, a super-hydrophilic surface can be probed to be hydrophilic through the texturing surface and such morphology strongly depends on background gas pressure [24].
Contact angle measurements of the non-treated and treated 100 nm silica films on glass, FTO, and Si substrates.
In the below equations, for uniform surface
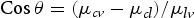
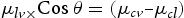
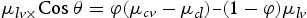
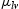 is the tension between liquid and vapour,
is the tension between liquid and vapour, 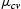 is the tension between surface and vapour,
is the tension between surface and vapour, 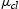 is the tension between surface and liquid. This mechanism is well established and known as the theory of Wenzel and Cassie–Baxter [19,20]. In our case, adsorbing behaviour of the organic group (acetone) is the key mechanism [25]. The previous study confirmed that alkylic groups of all organic molecules results in hydrophilic behaviour because of their intrinsic non-polar character [25]. Also, morphological study by SEM confirms that porosity changes after acetone treatment and samples grown at a high oxygen flow rate. The results correlate a clear dependence of wettability on the sonication with solvent where the degree of super-hydrophilicity changes as the contact angles increase owing to the hydrophilic character induced by solvent treatment.
is the tension between surface and liquid. This mechanism is well established and known as the theory of Wenzel and Cassie–Baxter [19,20]. In our case, adsorbing behaviour of the organic group (acetone) is the key mechanism [25]. The previous study confirmed that alkylic groups of all organic molecules results in hydrophilic behaviour because of their intrinsic non-polar character [25]. Also, morphological study by SEM confirms that porosity changes after acetone treatment and samples grown at a high oxygen flow rate. The results correlate a clear dependence of wettability on the sonication with solvent where the degree of super-hydrophilicity changes as the contact angles increase owing to the hydrophilic character induced by solvent treatment.
Figure 2 shows the contact angle measurements of silica films grown at four different pressures and treated later with acetone in the ultra-sonic bath. As shown in Figure 2, hydrophilicity decreases with the increasing oxygen flowrate (15 sccm). The deposition pressure (2e − 4 Torr) related to higher oxygen flowrate (15 sccm) can roughen the surface significantly and the surface turns to be super-hydrophilic where porosity increases due to lower packing density [24]. Hence, hydrophilic behaviour can be the result of two added factors namely oxygen partial pressure during the reactive growth and sonication using acetone. Increasing surface roughness lowers the interface tension to decrease surface hydrophilicity.
Contact angle measurements of the treated silica layers on glass, FTO, and Si substrates grown at 2e − 5 Torr (5 sccm), 9e − 5 Torr (10 sccm), 7e − 5 Torr (15 sccm), and 2e − 4 Torr (20 sccm) deposition pressures.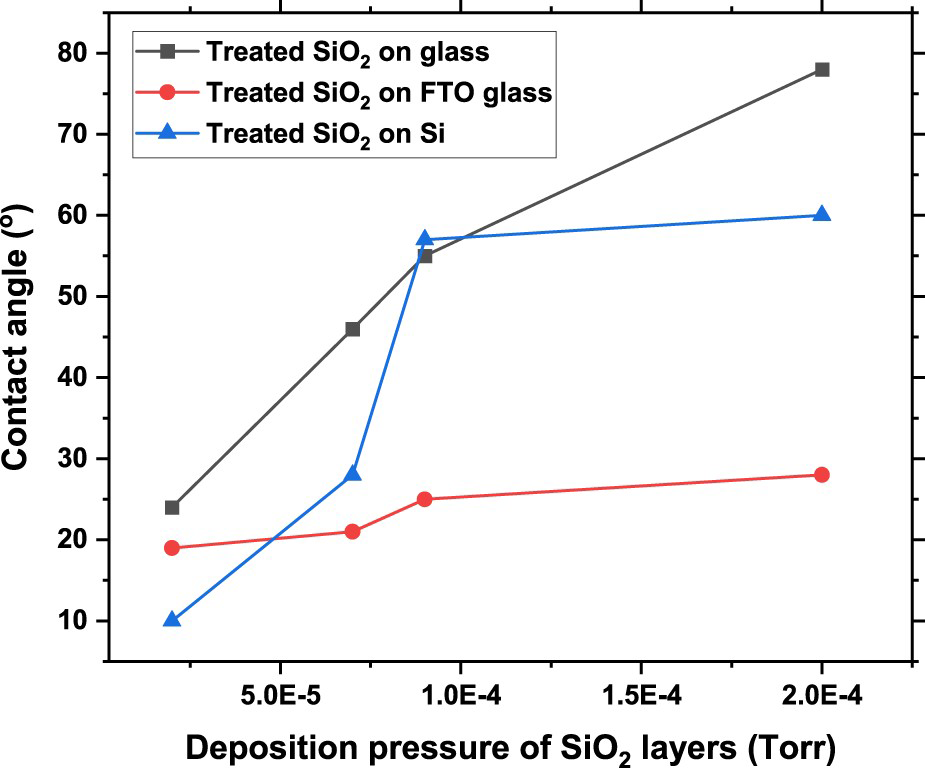
X-ray photoelectron spectroscopy (XPS) analysis the silica thin films
Figure 3 shows the X-ray photoelectron spectroscopy (XPS) survey of samples grown at 2e − 4 Torr deposition pressure on glass (Figure 3(a,b)) and FTO coated glass substrates (Figure 3(c,d)). The results confirm that treated samples were not etched or altered in terms of stoichiometry. As measured, all samples with monatomic etching confirm silicon (Si 2p) and oxygen (O 1s) spectra of the thin films grown reactively under an oxygen atmosphere. Regarding the oxygen chemical state analysis, carbon spectra fitting has been used to identify the amount of oxygen related to carbon species (i.e. C−O and C=O), which can be inferred from the total oxygen to conclude the carbon-related oxygen (C-metal bond). Reactively grown silica samples show Si and O signals with clear SiO
x
chemical states, with Si 2p located at 103 eV. The stoichiometry of the SiO
x
samples was found to be SiO1.85 and SiO1.91 on glass substrates for non-treated and treated samples, respectively, whereas it was SiO1.86 and SiO1.89 on FTO coated glass substrates for non-treated and treated samples, respectively. A clear stoichiometry was derived for both cluster etching and monatomic etching.
XPS survey, along with Si2p and O1s spectra of films grown at 2e − 4 Torr shown for both treated and non-treated samples on glass (a,b) and FTO coated glass substrates (c,d).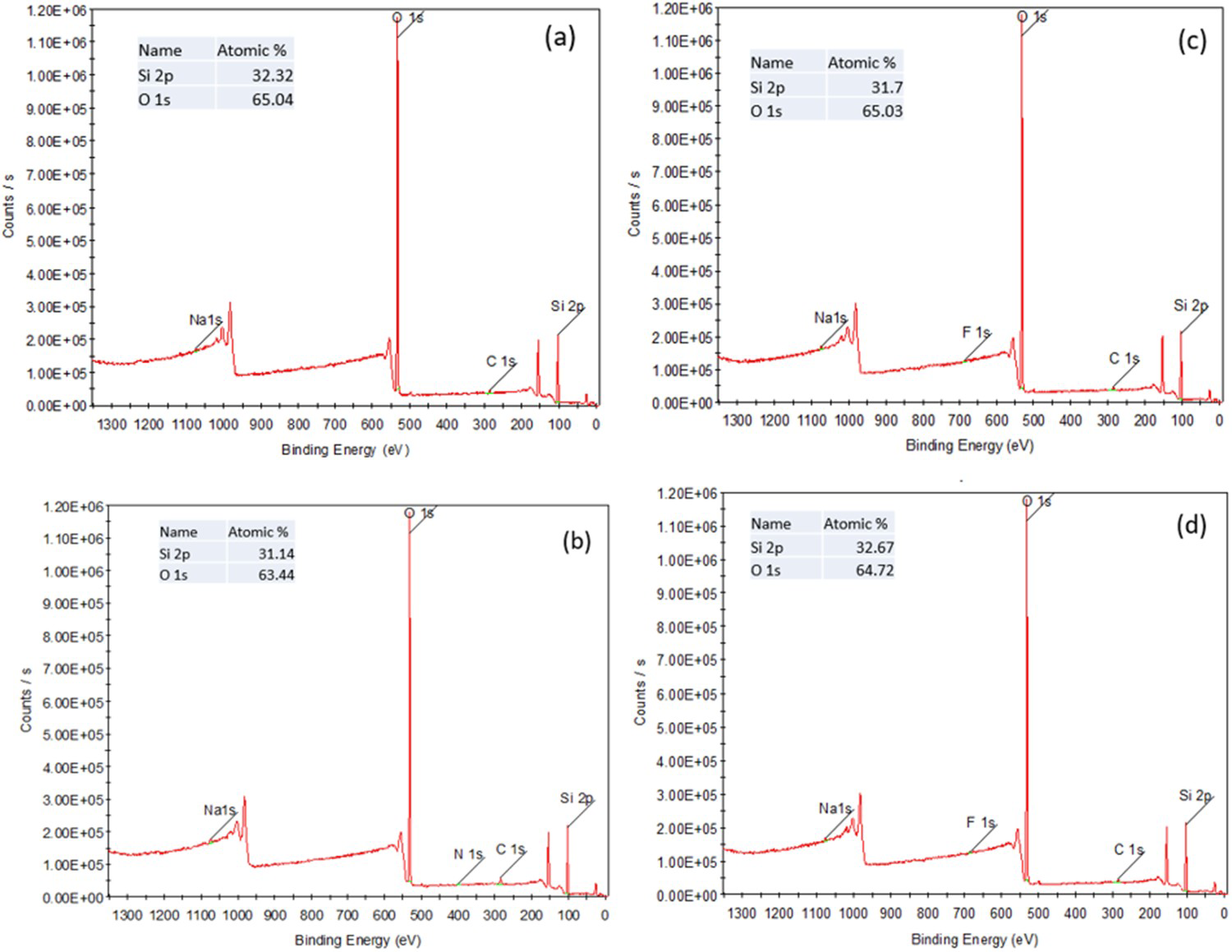
Morphological study of the silica thin films
Figure 4(a–f) shows SEM morphological study of silica films on glass, FTO coated glass, and Si substrates. It has been found that sample roughness increases with partial process gas oxygen pressure due to collided gas atoms on surface. As shown in the FESEM morphological mapping of the thin films of typically 100 nm thickness, grown under at two different extremes presume deposition pressure 2e − 5 and 2e − 4 Torr on three different substrates. Films grown with higher oxygen flow rates result in higher surface roughness. As shown, the hydrophilic behaviour of such silica layers is related to both acetone treatment and oxygen partial pressure, hence the tension reduction between water droplet and surface results in hydrophilicity.
SEM images of e-beam evaporated treated 100 nm silica layers on glass (a,b), FTO (c,d), and Si (e,f) substrates grown at 2e − 5 and 2e − 4 Torr deposition pressures.
Optical study of the silica thin films
Figure 5(a,b) displays the optical transmittance and absorptance of the silica films with varying the deposition pressures. The optical transmission was measured above 90% in the visible range for all silica samples on glass substrates, confirming thus the as-deposited films are highly transparent. All the films on glass substrates show a lower range of absorptance, thus, making such sample ideal candidate for anti-reflection coating as well. The Silica films associated with the deposition pressure of 2e − 4 Torr show the higher absorptance in the visible range. In addition, the layers grown on FTO coated samples show high transparency (>400 nm) where parasitic absorbance phenomenon has been observed below 1000 nm due to the FTO bandgap of 3.86 eV. As confirmed, oxygen content does not affect the optical properties of the films adversely, whereas other features, for example, acetone treatment and morphological properties become critical parameters to our study [22,26,27].
UV-Vis-NIR measurement of the (a) transmittance and absorptance spectra on glass substrates (b) transmittance and absorptance spectra on FTO glass substrates for various deposition pressures.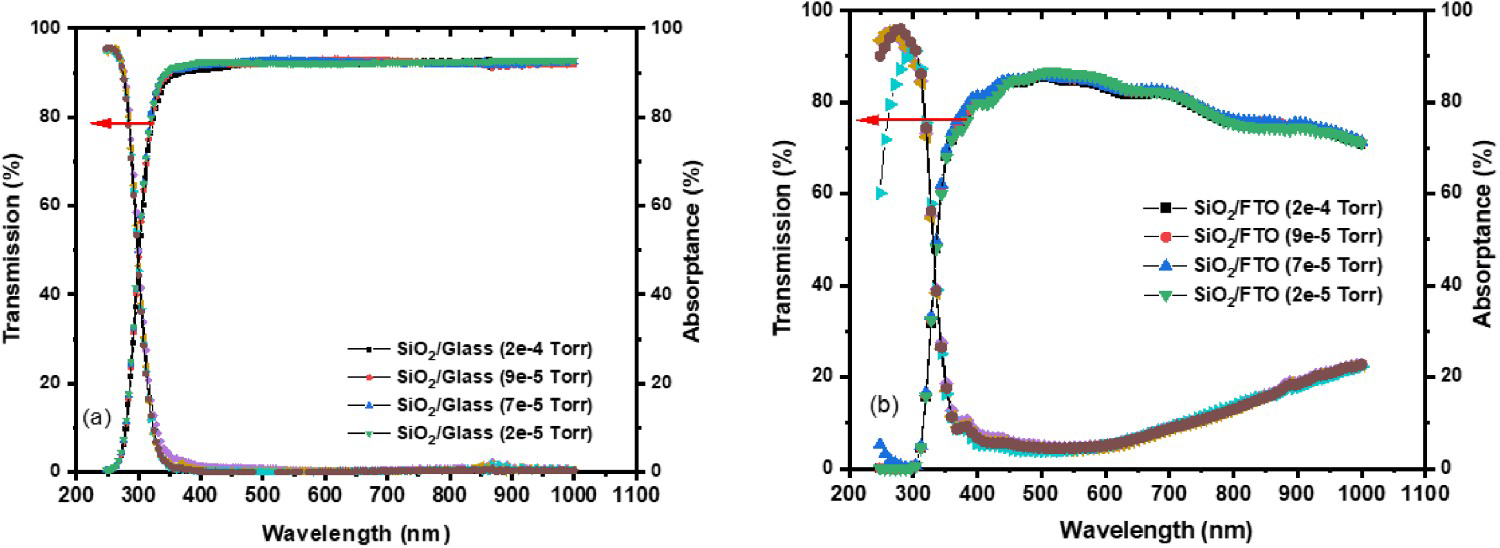
Thickness study of the silica thin films
Measured thickness of the silica films.
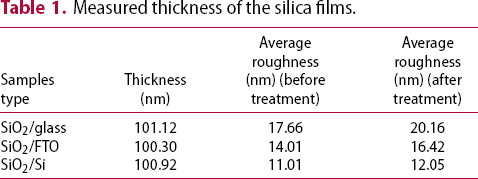
Finally, the treated samples were put in outdoor conditions for 21 days to assess their anti-soiling capability under real-world conditions. Figure 6 shows the representative SEM micrographs of a bare glass and a treated SiO
x
coated samples after 3 weeks of exposure under outdoor conditions. The preliminary results show a clear difference when a hydrophilic coating is sued, and work is currently in progress to quantify this anti-dust property and correlate it with the degree of the surface wettability.
Typical SEM micrographs of the (a) bare glass, and (b) treated samples after 3 weeks of exposure in outdoor conditions.
Summary
In this work, as-deposited and treated e-beam evaporated silica thin films grown at 2e − 5 and 2e − 4 Torr on different substrates like glass, FTO coated glass, and Si were characterised using contact angle measurement and X-ray photoelectron spectroscopy analysis. The morphology of the resulting surfaces was characterised by a field emission scanning electron microscope (FESEM). The stoichiometry of the SiO x samples was found to be SiO1.85 and SiO1.91 on glass substrates for non-treated and treated samples, respectively, whereas it was SiO1.86 and SiO1.89 on FTO coated glass substrates for non-treated and treated samples, respectively. Acetone as a solvent acts in changing the wetting behaviour of such silica layers. Such solvent with OH− group decreases the hydrophilicity level by reacting with the silica surface. Interestingly, the surface of the silica layers turns to be hydrophilic with the increment of water droplet and surface interface to 78° (glass), 26° (FTO), and 60° (Si). The optical transmission was measured above 90% in the visible range for all silica samples on glass substrates, confirming thus the as-deposited films are highly transparent. Finally, outdoor assessment of these samples demonstrated clearly the anti-soiling capacity of the treated samples. This study contributes in tuning the wetting behaviour of thin films surface through an organic agent to develop a cost-effective method for large-scale anti-soling coating. Further studies should be carried out to investigate the effect of outdoor conditions on these coatings along with degradation study.
Footnotes
Acknowledgements
The authors are grateful for the support from QEERI Core Labs and Thin Films Lab for material deposition and characterisation. In addition, we would like to thank Yahya Zakaria for the X-ray photoelectron spectroscopy (XPS) analysis. Open Access funding provided by the Qatar National Library.
Disclosure statement
No potential conflict of interest was reported by the author(s).
