Abstract
Hägg carbide (χ) is, during the tempering of carbon-rich martensite, referred to as a transition carbide which eventually gives way to cementite. However, there are Fe–C binary phase diagrams estimated using thermodynamic data, that define a low-temperature phase field where a mixture of Hägg carbide and ferrite is more stable than that of cementite and ferrite. In this scenario, it may be cementite which is the transition carbide. Evidence is presented here that the predominance of Hägg carbide over cementite in the circumstances described is unlikely to be correct based on historical and new observations. Literature data are also interpreted to show that η and ϵ-carbides are best regarded as transition-phases relative to mixtures of cementite and ferrite.
Keywords
Introduction
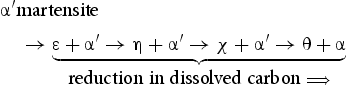
 refers to martensite and α to ferrite. This implies that the first three carbides in this equation form only because there is some kinetic advantage over the more stable cementite.
refers to martensite and α to ferrite. This implies that the first three carbides in this equation form only because there is some kinetic advantage over the more stable cementite.
Iron carbides.

Note: The actual composition of ϵ-carbide can affect its crystal structure, hence the two space groups.
Chipman [3,4] first contradicted this picture, by suggesting that there is a phase field in the Fe–C equilibrium diagram, where mixtures of χ-carbide and ferrite may be more stable than those of cementite and ferrite, even at the smallest of carbon concentrations. The data utilised to reach this conclusion were based on experiments in which iron was reacted with butane [5]. Based on a single point from these experiments, Chipman derived an admittedly approximate equation for the free energy of formation of χ from α-Fe and graphite, in order to define the χ+α phase field on the Fe–C diagram. Given the uncertainties in the data, he emphasised the need to distinguish thermodynamic and kinetic effects, i.e., whether the experiments on the relative stabilities of χ and θ are sufficiently long to achieve equilibrium.
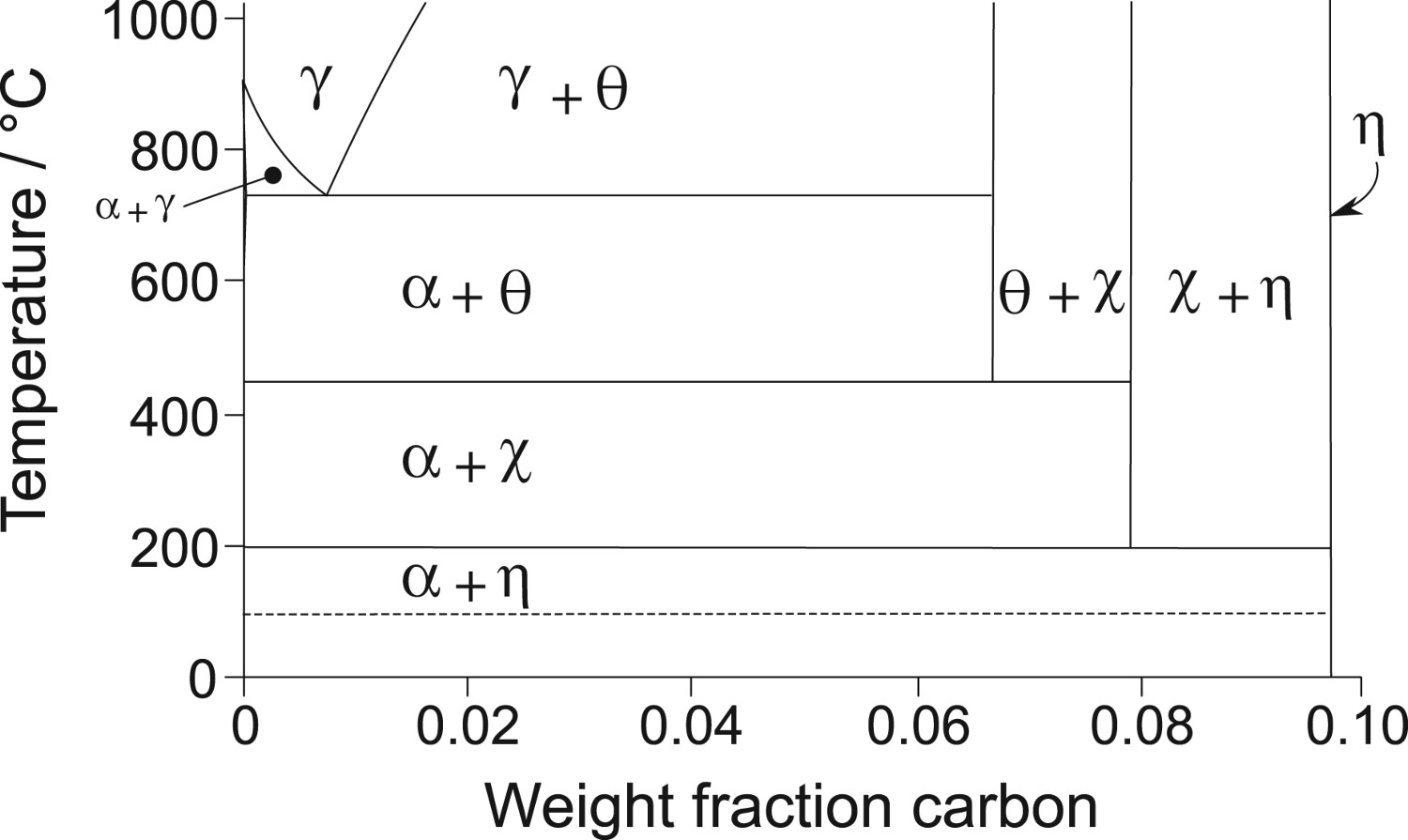
Given the dearth of data, recent work by Naraghi et al. [6] represented the thermodynamic data for χ and η using data for cementite, but weighted by the composition, for example, by writing the heat capacity at constant pressure as 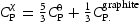 They did not publish the Fe-C phase diagram for temperatures below 900 K, but provided us with the necessary assessed data, resulting in the diagram illustrated in Figure 1. The clear implication is that that χ and η are not always
They did not publish the Fe-C phase diagram for temperatures below 900 K, but provided us with the necessary assessed data, resulting in the diagram illustrated in Figure 1. The clear implication is that that χ and η are not always
Part of the iron-carbon equilibrium phase diagram calculated using data provided by Naraghi et al. [6]. The dashed line represents a temperature below which a solid solution of iron and carbon may tend to undergo the clustering of carbon atoms as a precursor to a conditional spinodal. First principles calculations of the change in internal energy Note: These are calculations that consider the formation of the carbide as an isolated phase, from the constituent atoms. corresponds to a lower stability. The data reported by Faraoun et al. [15] are abnormally large; the space group quoted for cementite by Faraoun et al. is inconsistent with the lattice parameters assumed, and the η-carbide unit cell seems to have a
corresponds to a lower stability. The data reported by Faraoun et al. [15] are abnormally large; the space group quoted for cementite by Faraoun et al. is inconsistent with the lattice parameters assumed, and the η-carbide unit cell seems to have a  [17].
[17].
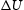 at 0 K and zero pressure for the reaction
at 0 K and zero pressure for the reaction 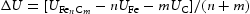 .
.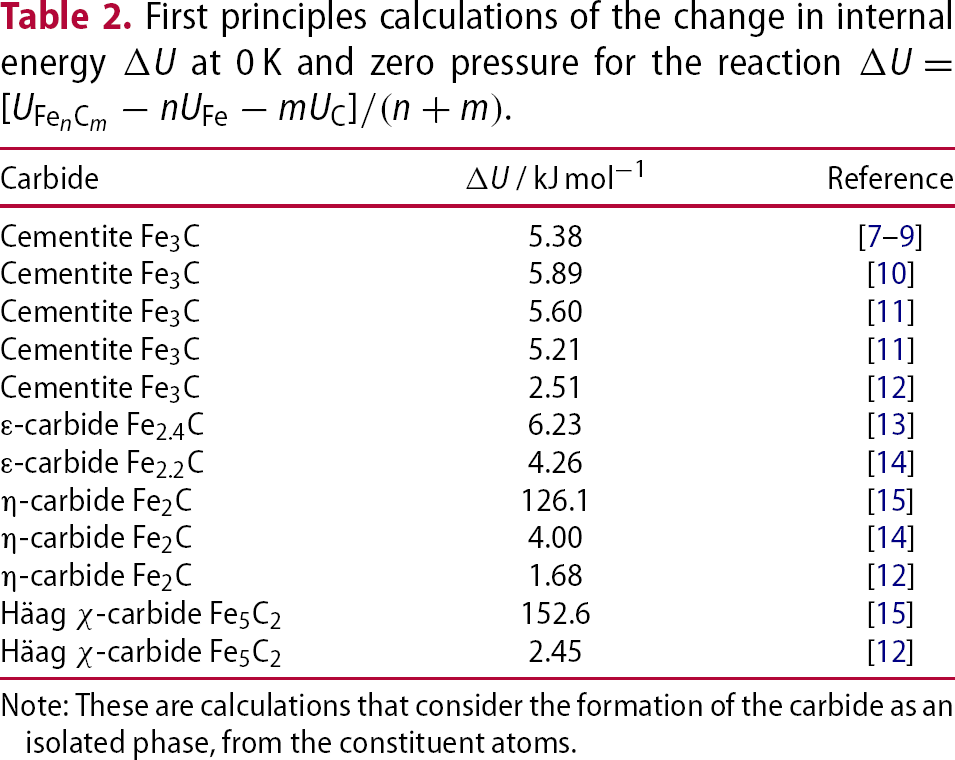
Table 3 summarises the data assembled from a search of the literature, for plain carbon steels (Fe–C) which have been tempered from a supersaturated-ferritic or martensitic condition. The number of observations is small, but not surprising given that Fe–C binary steels are not commercially viable. The data include the tempering temperatures and times. With one exception, the tempering times involved are probably not sufficient for the system to equilibrate and permit χ, η and ϵ to transform into θ. Figure 3 represents a plot of these data, using a Larson–Miller parameter [28] to represent the The formation energy Fe-C steels (neglecting trace impurities) in which carbides have precipitated during tempering of supersaturated ferrite or martensite. Note: The very low carbon data represent cases where allotriomorphic ferrite was quenched and then tempered.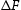
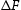 of cementite for the reaction
of cementite for the reaction 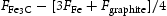 . Data from CALPHAD assessment by Hallstedt et al. [10]. A negative value implies that cementite becomes stable relative to the mixture of α and graphite.
. Data from CALPHAD assessment by Hallstedt et al. [10]. A negative value implies that cementite becomes stable relative to the mixture of α and graphite.
Two conclusions can be drawn from Figure 3. First, that cementite seems to be the most stable phase over a wide temperature range when the strength of the heat treatment is greatest within the scope of the dataset. Second, that cementite is the only precipitate when the excess carbon concentration is very small. This would be expected for a phase of high stability because a small carbon concentration in solution corresponds to a small driving force for precipitation. In such circumstances, transition phases (which by definition lead to a smaller reduction in free energy) would not be able to precipitate.
Plot of the data presented in Table 3, against a Larson–Miller parameter 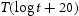
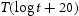 , where
, where
An arc melted 60 g sample made from pure electrolytic-iron and carbon was made and chemically analysed to have the composition Fe-1.60 wt-%. Two samples were sealed in quartz tubes for austenitisation at 1120 C for 1 h before quenching into water. Because of the large carbon concentration, to promote further martensitic transformation, the samples were cooled into liquid nitrogen. Sample A was kept there for 27 h, and sample B for 36 h; the difference is simply a consequence of practicalities.
C for 1 h before quenching into water. Because of the large carbon concentration, to promote further martensitic transformation, the samples were cooled into liquid nitrogen. Sample A was kept there for 27 h, and sample B for 36 h; the difference is simply a consequence of practicalities.
Both were then subjected to X-ray diffraction covering 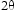 = 25–70
= 25–70 with a step size of 0.03
with a step size of 0.03 and a dwell time of 15 s. The measurements used nickel-filtered CuKα radiation and the data were subjected to Reitveld refinement. Sample A was then sealed in a glass tube and tempered at 400
and a dwell time of 15 s. The measurements used nickel-filtered CuKα radiation and the data were subjected to Reitveld refinement. Sample A was then sealed in a glass tube and tempered at 400 C for 406 h (Larson–Miller parameter 15216) and Sample B at 300 for 295 h (Larson–Miller parameter 12875), after which they were once again subjected to X-ray analysis.
C for 406 h (Larson–Miller parameter 15216) and Sample B at 300 for 295 h (Larson–Miller parameter 12875), after which they were once again subjected to X-ray analysis.
The quenched samples exhibited only martensite and retained austenite, Figure 4. In contrast, the patterns presented in Figure 5(a,b) show that for both tempering temperatures, the austenite has vanished and peaks that can with clarity be indexed to cementite are present. The volume fractions of cementite were measured to be 0.237 X-ray diffraction data from quenched and cryogenically treated samples of Fe-1.60C wt-%. (a) Sample A, containing 0.11 X-ray diffraction data from the tempered samples of Fe-1.60C wt-%. (a) Sample A, tempered at 400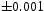 and 0.24
and 0.24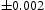 in Samples A and B, respectively. These compare very well with the equilibrium fractions estimated using phase diagram calculations (allowing only cementite and ferrite to exist), for both tempering temperatures, at 0.239. No evidence of χ carbide could be found in either case; Figure 5(c,d) shows the same patterns separately, but with the positions where the strongest of χ peaks are expected. It is evident that there is no χ-carbide present. The two crosses marked onFigure 3 therefore represent cementite.
in Samples A and B, respectively. These compare very well with the equilibrium fractions estimated using phase diagram calculations (allowing only cementite and ferrite to exist), for both tempering temperatures, at 0.239. No evidence of χ carbide could be found in either case; Figure 5(c,d) shows the same patterns separately, but with the positions where the strongest of χ peaks are expected. It is evident that there is no χ-carbide present. The two crosses marked onFigure 3 therefore represent cementite.
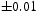
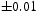 volume fraction of retained austenite. (b) Sample B also containing 0.11
volume fraction of retained austenite. (b) Sample B also containing 0.11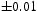 volume fraction of retained austenite.
volume fraction of retained austenite.
 C. The small peak at 62.5
C. The small peak at 62.5 could not be identified with any of the carbide or matrix phases (b) Sample B, tempered at 300
could not be identified with any of the carbide or matrix phases (b) Sample B, tempered at 300 C. The same data were then used to check against χ-carbide: (c) Sample A, tempered at 400
C. The same data were then used to check against χ-carbide: (c) Sample A, tempered at 400 C. (d) Sample B, tempered at 300
C. (d) Sample B, tempered at 300 C.
C.
An investigation of literature data and our own experiments establish that it is not correct to identify a χ+α phase field on the iron-carbon equilibrium phase diagram (Figure 1), one which is supposed to be more stable than θ+α. Furthermore, the analysis of published data indicates that the η+α phase field on Figure 1 is also unlikely to be correct because only cementite is observed to form in very low carbon steels at low tempering temperatures. The conclusion is that the thermodynamic data on which the phase diagram calculations are based need further probing.
Footnotes
Acknowledgments
Disclosure statement
No potential conflict of interest was reported by the authors.
References
 C-Fe-CH
C-Fe-CH -H
-H and Fe
and Fe C-Fe-CH
C-Fe-CH -H
-H
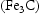
 C and (Fe,Cr)
C and (Fe,Cr) C
C
 C, χ-Fe
C, χ-Fe C
C , and η-Fe
, and η-Fe C from first principle calculation
C from first principle calculation
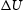 / kJ mol−1
/ kJ mol−1