Abstract
Documentation of Chinese materia medica (CMM) dates back to around 1,100 BC when only dozens of drugs were first described. By the end of the 16th century, the number of drugs documented had reached close to 1,900. And by the end of the last century, published records of CMM have reached 12,800 drugs. The recorded traditional information includes toxicities, incompatibilities, cautions, precautions and contraindications. Thus, contrary to a general misconception, toxicity data on Chinese herbs exist and are not meager. If consulted properly before embarking on a study of CMM, it will save researchers much time and efforts and the frustration of not obtaining consistent or expected results. The history of documentation is reviewed with examples of major classic and contemporary works highlighting and emphasizing the manner in which CMM toxicities have traditionally been documented through clinical experience as opposed to most modern toxicity data that are based on animal experimentation.
Keywords
Introduction
Traditional Chinese medicine (TCM) is a well-established system of healthcare with over 3,000 years of continuous documentation. No other healthcare systems in the world (modern or ancient) can claim such a distinction. Among its various aspects that include diagnostics, acupuncture, moxibustion, qigong
 , exercises, tuina
, exercises, tuina
 massage, and materia medica, the last is probably the most important. Chinese materia medica (CMM), often referred to simply as traditional Chinese herbs or traditional Chinese medicines, is important not just to TCM in modern practice, it is also important to modern American healthcare, as an increasing number of Americans is using it as an alternative therapeutic modality to conventional drugs as well as in dietary supplements. Although to date no systematic investigations have been conducted on the efficacy and safety of CMM that reflect its true toxicity and/or value as they relate to its long-documented literature, it is simply a matter of time before such studies need to be undertaken to protect the health of the American public and to provide them with truly alternative choices in their healthcare. For these reasons, it is imperative that the general facts regarding CMM documentation (hitherto still little known to the modern scientific and medical communities outside of China) be presented to interested researchers, providing them with some basic information regarding the traditional properties of these materials and their major differences from conventional drugs or botanicals intended for use solely as raw materials for modern drug discovery and development. To properly evaluate traditional Chinese medicines for their documented values that will lead to relevant and reproducible results, one must have some appreciation for how data on the properties and toxicities of Chinese traditional drugs have been generated and periodically revised over the past 3 millennia. This review attempts to provide some such information. Its focus will be on information directly related to CMM as a whole, and not on any isolated chemical components.
massage, and materia medica, the last is probably the most important. Chinese materia medica (CMM), often referred to simply as traditional Chinese herbs or traditional Chinese medicines, is important not just to TCM in modern practice, it is also important to modern American healthcare, as an increasing number of Americans is using it as an alternative therapeutic modality to conventional drugs as well as in dietary supplements. Although to date no systematic investigations have been conducted on the efficacy and safety of CMM that reflect its true toxicity and/or value as they relate to its long-documented literature, it is simply a matter of time before such studies need to be undertaken to protect the health of the American public and to provide them with truly alternative choices in their healthcare. For these reasons, it is imperative that the general facts regarding CMM documentation (hitherto still little known to the modern scientific and medical communities outside of China) be presented to interested researchers, providing them with some basic information regarding the traditional properties of these materials and their major differences from conventional drugs or botanicals intended for use solely as raw materials for modern drug discovery and development. To properly evaluate traditional Chinese medicines for their documented values that will lead to relevant and reproducible results, one must have some appreciation for how data on the properties and toxicities of Chinese traditional drugs have been generated and periodically revised over the past 3 millennia. This review attempts to provide some such information. Its focus will be on information directly related to CMM as a whole, and not on any isolated chemical components.
Interpretation and English Transliteration of TCM
One of the most difficult, but essential, tasks in studying the safety and efficacy of CMM is the interpretation of its writings. These are often esoteric and cannot be easily equated to modern pharmacologic and medical terms. Once a term or concept is misinterpreted using modern terminology, without simultaneously providing the original characters describing it, the original meaning will not be conveyed and even may be lost, which would lead to wrong conclusions and irreproducible results. A typical example is the Chinese term/concept of qu feng
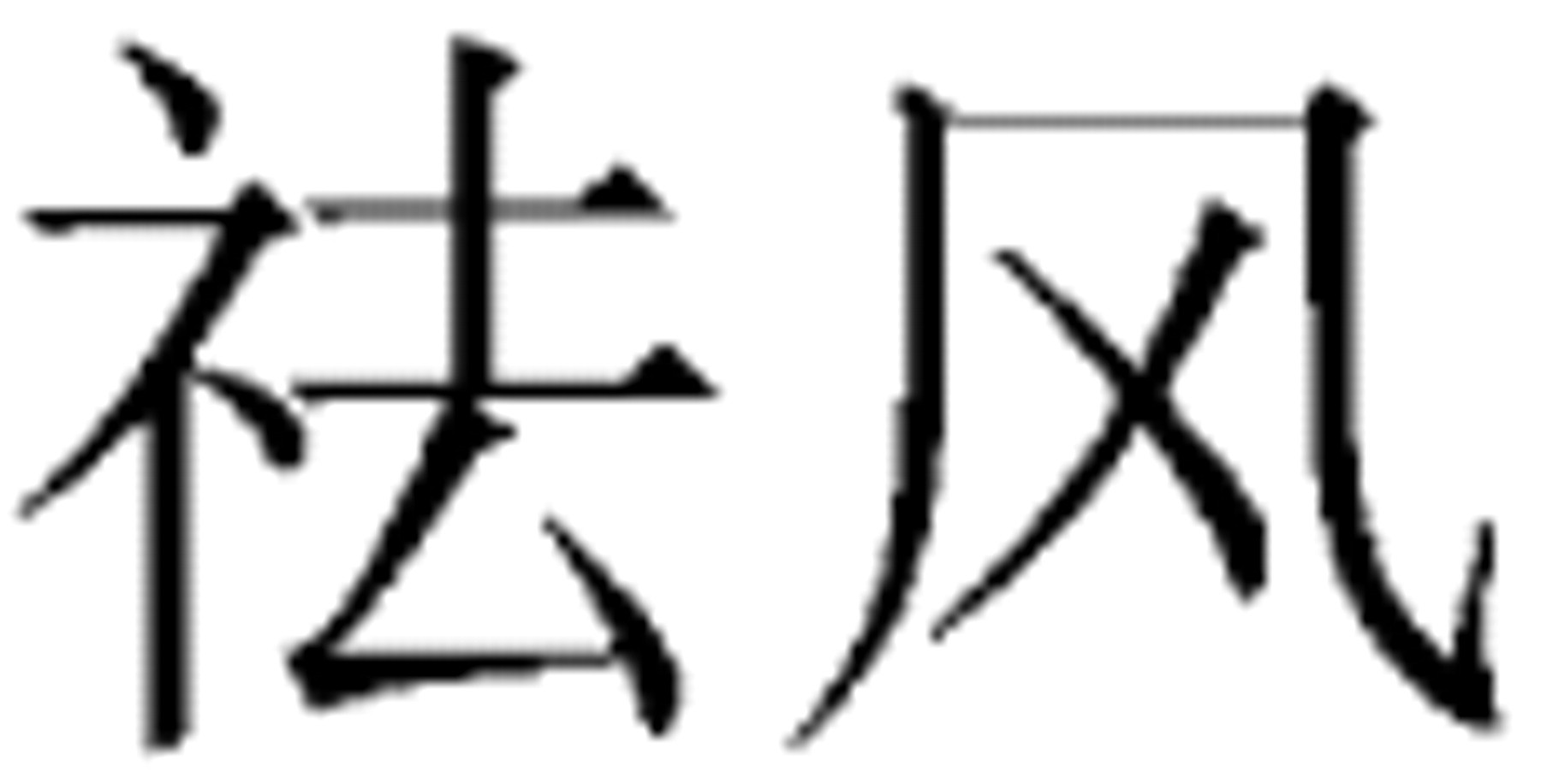 , literally meaning “removing wind or expelling wind,” but its true meaning has something to do with “relieving rheumatic pain and the common cold.” If a scientist who reads Chinese but does not understand TCM intended to study a CMM with qu feng properties misinterpreted it as related to “flatulence,” the expected “anti-flatulence” result of the subject herb would not be obtained. If this negative result were published without providing the original Chinese characters (or at least a proper transliteration) for the indication or the property studied, no other researchers could reproduce the results. Consequently, while modern science has just started to discover the potential of CMM and tries to validate its safety and efficacy, it is important always to accompany modern interpretations of TCM concepts with their original Chinese characters, or at least a standardized transliteration of them.
, literally meaning “removing wind or expelling wind,” but its true meaning has something to do with “relieving rheumatic pain and the common cold.” If a scientist who reads Chinese but does not understand TCM intended to study a CMM with qu feng properties misinterpreted it as related to “flatulence,” the expected “anti-flatulence” result of the subject herb would not be obtained. If this negative result were published without providing the original Chinese characters (or at least a proper transliteration) for the indication or the property studied, no other researchers could reproduce the results. Consequently, while modern science has just started to discover the potential of CMM and tries to validate its safety and efficacy, it is important always to accompany modern interpretations of TCM concepts with their original Chinese characters, or at least a standardized transliteration of them.
Over the past century, various methods of transliteration of Chinese characters have been used. The two best known are the Wade-Giles and Pinyin systems. The former dominated the TCM literature for many decades until the latter was widely adopted beginning in the 1980s. Currently, the Pinyin system is the predominant one used worldwide except for the literature emanating from Taiwan. The Library of Congress and the National Library of Medicine still hold a sizable amount of information in the Wade-Giles transliteration. Because of this lack of uniformity in one of the most important tools in the description and interpretation of TCM, much confusion exists in the identities of CMM. This consequently is a major source of irreproducible results in research and clinical applications involving TCM that requires the interpretation of traditional TCM concepts.
In addition, as with other traditional and botanical medicines, traditional Chinese drugs often have multiple names. Some of these are traditional ones recorded in well-known herbals over many centuries. However, the majority deals with vernacular names that vary from region to region. Sometimes numerous names apply to a single drug while other times a single name applies to multiple drugs. Thus, even without the transliteration (Wade-Giles or Pinyin) issues, correct identity of CMM is already a major problem. And this is another major source of irreproducibility in CMM research and modern clinical practice.
For lack of a universally standardized system for naming Chinese herbal drugs, the standardized names (standardized common names, Latin binomials, and Pinyin transliterations) in the Herbs of Commerce, 2nd Edition (McGuffin et al, 2000) for CMM are used in this review. Since it is the only authoritative work available with standardized names for botanicals in commerce, it was adopted by the United States Food and Drug Administration as an official standard for the labeling of botanical ingredients in dietary supplements effective January 1, 2006 [21 CFR 101.4(h)]. However, the FDA withdrew the Direct Final Rule on December 28,2005 after adverse comments by a Siberian ginseng company that objected to the new standard common name of “eleuthero” for “Siberian ginseng” used in this work, thus delaying its adoption as the labeling standard. Despite this status, the book contains only about 500 Chinese botanicals, which are a small fraction of the over 12,800 known CMM documented in the Chinese literature (Institute of Chinese Materia Medica, 1996). Hence, for CMM that are not found in the Herbs of Commerce but are quoted in this review, the common standardized Pinyin names and/or the corresponding Latin binomials recorded in credible publications, including recent Chinese Pharmacopoeias and major works compiled by joint efforts of major Chinese institutions are used (Pharmacopoeia, 1963–2005; Pharmacopoeia of the People’s Republic of China, 2000; Zhonghua Bencao, 1999; Luo, 2003).
Historical Background
Chinese materia medica consists of drugs derived from botanicals, minerals and animals, with the botanical ones making up the majority. It is a key and integral part of TCM, which also includes other therapeutic modalities, as mentioned earlier. The use of CMM dates back several thousand years. However, as evidence available to date shows, its written documentation did not start until only around 1,100 B.C. (Leung, 1990). Since then, information on Chinese herbs has evolved from sketchy mentions in ballads and geographic works, to works devoted to disease treatment, and eventually culminating in monumental works exclusively devoted to individual CMM and their prescriptions. All these publications contain information on safety, toxicity, incompatibility, precaution, and caution. Major works spanning a 3,000-year period are highlighted in Table 1 (Leung, 1990, 2001).
Due to the nature of CMM practice that relies totally on direct clinical experience with humans over time, there are no precise toxicity data like those of modern Pharmaceuticals generated from animal and in vitro experimentation. Nor are there any data on enzyme inhibitors, receptor agonists, receptor antagonists, or the like, which are often used to describe mechanisms of action of chemicals, including toxic ones. What is available in traditional toxicity data includes general information accumulated over time that ranges from decades to centuries, based on collective data from different accounts, not from a single episode based on any particular herbal sample from any particular locality. Such documentation can be viewed as word-of-mouth, but it has been generally based on multiple occurrences vetted over time before it entered the traditional literature. As a corollary, in the modern documentation of scientific research, any published report from a single source (laboratory, institution, or clinic) is only considered credible and the reported results valid when these results are reproduced by others.
There are no uniform methods of assessing the toxic effects of herbs in general and CMM in particular. Modern scientific techniques may involve acute and subacute testing that have been devised for modern chemical drugs, and are thus not readily applicable to multicomponent systems like CMM and foods. Hence, it is extremely difficult to predict the adverse effects of herbs based simply on such evaluation of a toxic chemical present in these herbs. Yet, such interpretative and extrapolative assessments are often used exclusively to define the toxic effects of CMM. Many of the recorded adverse reactions thus far attributed to CMM in the modern literature have been equivocal, due to problems inherent in current CMM research. These problems include dubious identity of subject herbs, misappropriation of a single chemical for the total activity of an herb, and the bias of conventional medical and pharmacological researchers, who undertake the studies or report on the findings, albeit often unintentional. The most notorious yet well-publicized and widely quoted “adverse effects” of “ginseng” is the classic report by Siegel (1979) which coined the term “ginseng abuse syndrome,” consisting of nervousness, insomnia, hypertension, skin rash, and diarrhea. It is a typical example of how the use of dubious test materials (in this case, commercial herb-powder capsules labeled as “ginseng”) combined with a lack of knowledge in CMM have produced and perpetuated a set of adverse reactions that may have nothing to do with any particular ginseng, Asian (Panax ginseng C.A. Mey.) or American (Panax quinquefolius L.), material(s) intended for that study.
Consequently, despite voluminous publications on ginseng (identity seldom well-defined) over the past few decades, the traditional properties (including toxicity) and efficacy of renshen (Chinese or Korean ginseng) as it has been known, documented and used over time, are still unclear, thus perpetuating the controversies on ginseng. Unfortunately, this problem extends beyond ginseng; yet, it is currently not widely recognized.
General Misconceptions
To most modern scientists who have only taken a cursory look without trying to understand it, CMM is commonly regarded as nonscientific and anecdotal. This is quite understandable because, over millennia, as part of TCM, CMM has developed into a complicated and, to many, esoteric system of theories and practice based primarily on empirical human experience.
Unlike modern allopathic drugs, the efficacy and safety of CMM cannot be readily verified in a short term with animal experimentation, because the two are inherently very different systems of therapeutics. While the development of allopathic drugs starts with toxicity testing in animals before moving on to human toxicity and efficacy studies, the traditional development of CMM skipped the animal step and started with human experimentation right at the onset. The former may take a few to several years to establish their pre-approval or premarket safety and efficacy before actual human experimentation begins. Of all the modern drugs in extensive use today, few, if any, have more than 75 years of human experience. In contrast, some of the Chinese medicines have been used in humans for several millennia, with documentation of their safety and toxicity for at least 3,000 years. In addition, as in the case with modern allopathic drugs, new uses and toxicities continue to be discovered and recorded, albeit in Chinese, which makes these data generally unavailable to the modern scientific and medical communities. Consequently, there is a general belief among scientists and healthcare professionals that there are no toxicity or efficacy data of any kind in CMM. But, that is not true. In fact, such data exist. It’s just that they are not universally available in any major non-Chinese literature or databases. In this review, attempts will be made to present major publications, both classical and recent, some of which specifically deal with traditional toxicity data of CMM. Although most are in Chinese, they can be readily accessed and its relevant portions be translated by any reasonably knowledgeable Chinese-reading scholars or scientists, who need not conduct extensive research to discover these sources, as the basic information and approach will be provided in this review.
Many toxicity studies using modern scientific methods have been conducted on CMM-related materials (isolated chemicals, chemical fractions, extracts, etc.) over the past 75 or so years. However, most toxicity data resulting from these studies cannot be correlated to the traditional properties and indications of the intact CMM and thus may not be relevant to the traditional materials concerned. Reliance on such data will only serve the interest of conventional drug development efforts, which are not relevant to efforts to evaluate and validate the safety and efficacy of traditional CMM as they have been known and documented over time.
Basic Differences between CMM and Modern Drugs
In order to conduct meaningful toxicity studies on traditional medicines that will generate reproducible results, it is important to consider the following facts:
The toxicity of traditional herbs or formulas is not the same as that of their isolated chemicals.
The toxicity resulting from a use out of context cannot always be attributed to the traditional medicine in question.
Most scientists conducting research in this field have been trained in modern medical, pharmacological and/or pharmaceutical sciences. They are accustomed to viewing test materials as pure chemicals with rather well-defined pharmacologic activities that are readily measurable using modern technologies. Their knowledge of these primarily single-entity chemicals is accumulated over a relatively short period of time (normally within a generation), where initial manageable animal data lead to much more complicated human use data. Consequently, it is difficult for most of them to accept human toxicity and efficacy data that seem to have come from nowhere, with no easy traceability, and certainly not along the same path with which they are familiar. Considering that few of these scientists have prior experience with CMM, it is understandable that there is much skepticism concerning its toxicity data. However, it must be borne in mind, while pre-clinical toxicity data of modern drugs are fairly precise and primarily experimentally derived (e.g., via animal and/or in vitro studies), their human toxicity data are mainly obtained through actual human usage, which are much less defined, and the accumulation of these human toxicity data only starts after the drugs have been approved for clinical trials and later use in humans. And as long as a drug is in human use, accumulation of its toxicity data is an ongoing process which is not unlike what has happened with CMM, except that, with CMM, the process, has started centuries to millennia earlier.
Furthermore, due to the inherent differences in the practice of CMM as opposed to that of modern allopathic drug therapy, there are few or no parallels in many aspects between the two systems, including the following:
CMM practice is primarily empirical and holistic, while modern drug therapy is experimental science-based and targets specific symptoms and obvious disease-causative agents.
CMM practice emphasizes prevention and restoring balance that often are intangible, while modern medicine attempts to treat what is scientifically obvious or tangible.
While modern drugs are mostly single chemicals that are readily identified and quantified, CMM are multicomponent therapeutics whose strength is due to their unique combinations around which this whole therapeutic system is based.
While combination modern drug therapy only started a few decades ago with cancer chemotherapy and later AIDS treatment (e.g., anti-HIV cocktails), the practice of combination therapy in CMM started at least 3,000 years ago, which has since evolved into a well-documented treatment system unique to TCM.
Because of the many differences between modern pharmaceuticals and CMM, one would expect certain modern methodologies (e.g., identifying and standardizing against specific chemical markers) used in the evaluation of modern drugs not appropriate for studying the toxicity, safety and/or efficacy of these traditional medicines. Innovative methodologies that identify, characterize, and standardize CMM holistically and with consistency, which correlate their physical and chemical profiles with biological ones (e.g., genomic) to represent their well-documented traditional properties (in total) and indications will be more appropriate and useful.
The Documentation Process
Despite its esoteric nature, the theory and practice of CMM is not illogical as most scientists believe. If one takes time to learn some of its basics, one will find it continues to undergo evolution as more and more human clinical experience is being accumulated. Not unlike modern single-component chemical drugs (though the longest of which being in use, such as aspirin and morphine, have only a history of less than 200 years), the safety and therapeutic properties of Chinese herbal medicines are continually being determined and updated as new human experiences are acquired. While the human clinical experience with most modern allopathic drugs is relatively short (e.g., over decades), that of most CMM generally extends over centuries to millennia. Thus, aspirin may have initially been used as an analgesic in rheumatism, lumbago, and neuralgia in the early 1900s, human experiences with it since then have expanded its properties and uses into other areas, including the most recent use in reducing the risk of recurrent heart attack and stroke. And, its toxicities over this period have been found to include gastrointestinal upset, ulcer, and bleeding, as well as Reye’s syndrome, among others. These toxic properties of aspirin have not been determined by typical modern toxicologic studies required for any drug before its approval, but rather by clinical observations and experience after it has been put in clinical use. This scenario of documenting new properties (including toxicities) and new indications is basically the same as with Chinese herbal medicines, except what has been observed and documented in CMM has lasted much longer, most for centuries or millennia as opposed to only decades with most allopathic drugs.
A typical example is the familiar herb, American ginseng (Panax quinquefolius), a native North American species. The Jesuits first introduced its root to China at the beginning of the 18th century, believing it was the same as Chinese ginseng. It was well received. However, within a period of only a few decades, the Chinese found that it was not the same as their own ginseng and documented this difference. They did this strictly based on organoleptic identification and human clinical experience over time, without the help of modern taxonomic botany or other modern scientific techniques used in identification and quality control (chemical, spectral, chromatographic, biochemical, etc.), nor the modern sciences of pharmacology. Thus, by the mid 1700s, American ginseng was first described in writings as a distinctly different ginseng than Chinese ginseng and later became a favorite of the Cantonese. American ginseng is known since then as a tonic with cooling (as opposed to Chinese ginseng’s warming) properties ; and this fact has not changed for over two centuries. Now, American ginseng is normally used in persons with excessive yang who need a tonic to strengthen their resistance but cannot tolerate yang medicines like Chinese ginseng, which may aggravate the problems they may already have, caused by this imbalance. American ginseng is thus a different herbal drug than Chinese or Asian ginseng. They are derived from distinctly different yet closely related plant species. American ginseng was later botanically identified as Panax quinquefolius and Chinese, or Asian ginseng, as Panax ginseng. Although both are tonics, they have some different indications. Due to its cooling nature, American ginseng is used to reduce fever while Chinese ginseng is seldom, if ever, traditionally used for this purpose. These traditional properties were first observed and differentiated through organoleptic examination followed by human usage over time (in this case, relatively short), before the 2 materials were finally subjected to the modern science of botany which made the
The documentation of CMM toxicity is a dynamic process that keeps updating and revising the record whenever new data arise. The highly toxic nature of certain traditional drugs was already known and started to be documented about 3,000 years ago. For example, among the drugs described in the Wu Shi Er Bing Fang (See Table 1), are the toxic wu tou (aconite), ban xia (Pinellia ternata root), and li lu (Veratrum nigrum root/rhizome), many of which are also provided with processing methods to reduce their toxicity.
In the Shennong Bencao Jing (Shennong Herbal), the drugs were classified into 3 therapeutic categories based on their properties and relative toxicities. Despite the toxic nature of some of the drugs being revised centuries later, most have remained unchanged.
In the Bencao Gangmu (Compendium ofMateria Medico), 381 among its 1892 drugs are clearly described as toxic. In one of the most extensive modern records, the Zhongyao Da Cidian (Encyclopdia of Materia Medico), published in 1977, 495 of its 5,767 drugs are reported as toxic. In the newly published Zhonghua Bencao (The Chinese Herbal), which contains monographs on 8,980 drugs, the number of toxic drugs is not immediately apparent. However, in the Duyao Bencao (Toxic Drugs Herbal), a book that specifically deals with toxic traditional Chinese drugs published in 1993, 903 toxic drugs are described in detail. And according to its editors, 55 of these toxic drugs were previously considered nontoxic in historical records.
There is much latitude in the determination of which Chinese medicine is toxic and how severely so. Traditionally, toxic drugs are classified into 3 categories: highly toxic, toxic, and slightly toxic. Depending on the authors and the periods in which the herbals were published, drugs considered “highly toxic” by one author in a particular period might be considered only “toxic” by another. And certain drugs considered “nontoxic” might later be described as “slightly toxic.” Nonetheless, when a drug exhibited toxic effects over time, these effects were recorded. For example, in the Pharmacopoeia of the People’s Republic of China (Pharmacopoeia, 1963–2000), mylabris (Chinese cantharides) was changed from “toxic” in the 1963 edition to “highly toxic” in later editions (1977–1995), ginkgo seed from “slightly toxic” in 1963 and 1977 editions to “toxic” in the 1985–1995 editions, herb-paris rhizome (chong lou) from “nontoxic” in the 1977 edition to “slightly toxic” in 1985–1995 editions, orostachys herb (wa song) from “highly toxic” in the 1963 edition to “nontoxic” in the 1977 edition, honeycomb (feng fang) from “toxic” in 1963 to “slightly toxic” in 1977 and finally to “nontoxic” in 1985–1995 editions, houttuynia herb (yu xing cao) from “slightly toxic” in 1963 to “nontoxic” in 1977–1995 editions, and omphalia (lei wan) from “slightly toxic” in 1963 to “nontoxic” in 1977–1995 editions.
So, the dynamic process of documentation continues since the Shennong Bencao Jing started the toxicity documentation and classification 2,000 years ago. What are recorded as toxic drugs only apply to those that have been found to be consistently toxic over time, when used in the traditional manner. There are countless others that ordinarily are not toxic but may be toxic if used outside of tradition or out of context. These latter are the ones that are being increasingly known to produce adverse effects in recent years.
While documentation of the safety and adverse effects of herbs used in the West and those used in various ethnobotanical systems may be scanty, that of traditional Chinese drugs has been extensive and continuous since around 1100 BC. In fact, except for some CMM commonly used as tonics, few traditional Chinese drugs (plant, animal, and mineral) lack toxicity or safety records, including cautions and contraindications for their use. Practitioners of TCM are taught to use toxic drugs that play an important role in TCM practice. Historically, TCM physicians who were skilled in the use of toxic medicines to treat illnesses were highly revered. And highly toxic traditional drugs such as Sichuan aconite (both the main root called chuan wu, and the less toxic cured lateral root called fu zi), chan su (toad venom), and minerals containing arsenic (e.g., realgar orxiong huang), mercury (e.g., mercuric sulfide or zhu sha), and lead (e.g., lead tetroxide or qian dan) are still routinely used by TCM physicians in China (Pharmacopoeia, 1963–2005). Poisoning or adverse effects due to Chinese herbs are often caused by ignorance and/or their inappropriate use. Because of the laxity in adhering to truly traditional Chinese medical principles in training modern-day TCM physicians (Fruehauf, 1999) and the misconception of modernized TCM, such adverse events are bound to increase rapidly in the near future, further spurred on by a misinformed public who frequently use these medicines as dietary supplements, and outside of traditional practice. Consequently, before one starts evaluating any CMM for its toxicity and efficacy, it is extremely important to consult the classical traditional literature to assure that one does not miss some important toxicity information. This will not only save time and money in one’s research efforts, but also avoid toxic effects unnecessarily inflicted on human subjects or patients if the recorded toxicities are known or the cautions and precautions heeded by the investigators conducting the research.
Access To CMM Toxicity Data
Major Classic Works with Toxicity Data
There are at least 300 major classic works on CMM, known commonly as ben cao (
 ), published over the past 3,000 years. All of the major ones reviewed contain toxicity information. The following 4 are selected as examples for this review because they are the most important ones that provide a historical perspective, as well as a sense of the scope of toxicity documentation of traditional Chinese drugs.
), published over the past 3,000 years. All of the major ones reviewed contain toxicity information. The following 4 are selected as examples for this review because they are the most important ones that provide a historical perspective, as well as a sense of the scope of toxicity documentation of traditional Chinese drugs.
The Shennong Bencao Jing (Shennong Herbal)
 (ca. 100 BC–200 AD)
(ca. 100 BC–200 AD)
This is the first work exclusively devoted to CMM, compiled during the period from 100 BC to 200 AD. It is also the first that attempted a systematic classification of CMM based primarily on their toxicities. Thus, its recorded 365 drugs of plant, animal, and mineral origins were divided into 3 categories:
Superior (120)-jun
 (monarch), nontoxic, suitable for long-term use
(monarch), nontoxic, suitable for long-term use
Medium (I20)-chen
 (minister), can be toxic, depending on usage
(minister), can be toxic, depending on usage
Inferior (l25)-shi
 (envoy), toxic, for treating diseases
(envoy), toxic, for treating diseases
Despite the fact that the information on the toxicities and indications of the drugs was recorded 2,000 years ago, much of it is still valid. When used as traditionally prescribed, the toxic properties of these drugs, as classified, seem to have withstood the test of time.
Examples of common drugs/herbs grouped under the 3 categories include:
Superior–Asian ginseng (renshen), licorice (gancao), reishi (lingzhi), chrysanthemum (juhua), astragalus (huangqi), Job’s tears (yimi), sesame seed, lycium, schisandra, Chinese wild ginger (xixin, Asarum spp.), Chinese salvia (danshen), etc. A review of the CMM in this category shows that most items are still appropriately classified here. For example, ginseng, astragalus, licorice, Job’s tears, schisandra, and lycium to this day continue to be regarded as nontoxic and suitable for long-term use after a period of more than 2,000 years
Medium–Chinese ephedra (mahuang), kudzu root (gegen), dried ginger, dong quai (danggui), zhuling, seaweed, zicao, Chinese skullcap (huangqin), epimedium, etc. As expected, many of the drugs in this category can be toxic, especially if not used properly, such as mahuang.
Inferior–Rhubarb, aconite (juzi and wutou), mang coo (Illicium lanceolatum) platycodon, bletilla (baiji), pinnellia (banxia), Japanese knotweed (huzhang), heal-all (xiakucao), almond (xingren), peach pit (taoren), forsythia (lianqiao capsule), earthworm, arsenic sulfide (xionghuang), mercuric sulfide (zhusha), etc. Drugs classified under this group are supposed to be toxic and used for treating diseased conditions as opposed to those in the Superior category where they are used for health promotion and maintenance. Many of the drugs here can still be considered as toxic as they had been 2,000 years ago (e.g., aconite, mang cao, xiong huang, etc.). However, over time, some have been found to be nontoxic and have since been upgraded in recent times (e.g., bletilla, forsythia capsule, platycodon).
Bencao Gangtnu (Compendium of Materia Medica)
 (ca. 1592–1596)
(ca. 1592–1596)
This work is probably the best known of all Chinese classic herbals. It was compiled by Li Shi-Zhen who is generally considered as China’s greatest herbalist. In this work, the number of CMM items described had increased from 365 recorded in the Shennong Bencao Jing to close to 1,800 plus 11,100 prescriptions (Leung, 1990). Over the past four centuries, parts of it have been translated into numerous languages. The newest English version has recently been published, which was translated and compiled by Xi-Wen Luo (2003). This English version is in 6 volumes, totaling over 4,400 pages. For the first time, a complete translation of the Bencao Gangmu is available. A brief, but detailed, comparison of the description of a few drugs in the English and Chinese versions by this reviewer has revealed a close-to-text and reasonably readable translation, with practically no obvious omissions or rationalization on the part of the translator and editors. Pending further use and review, it is safe to assume this English translation as equivalent to the original Chinese version in scope and content.
In this work, among other categories described (preparation, indications, prescriptions, etc.), the “Properties” (quality and taste) category provides a brief description of whether a drug is toxic, slightly toxic or nontoxic; it also provides cautions and precautions as well as contraindications for some CMM items. Some of the drugs originally described in the Shennong Bencao Jing have been reclassified in this compendium from toxic to nontoxic and vice versa. For example, baiji or bletilla rhizome (toxic, under the inferior category in the Shennong Bencao Jing) is described as nontoxic in this 16th-century work. And xixin (Chinese wild ginger), formerly considered nontoxic is described here with cautions and incompatibilities with certain vegetables, meats and other herbs.
The English version has 8 appendices, including those for English drug names, Latin binomials, drug names in Pinyin and Chinese, and list of classic works quoted. Although this work only covers about 1,900 Chinese drugs, out of a total of around 12,800 with well-documented records, it at least would allow Western researchers access, for the first time, to a small portion of CMM toxicity data that has been buried and thus mostly inaccessible for centuries.
Zhongyao Da Cidian (Encyclopedia of Chinese Materia Medica)
 (Jiangsu, 1977)
(Jiangsu, 1977)
This was the most comprehensive work in 1977 on CMM since the Bencao Gangmu. It consists of 3 volumes and 3,518 pages, describing 5,767 traditional Chinese drugs, many in great detail. The modern botanical, chemical, pharmacological and clinical data included in this book are from the world literature up to and including 1972. The traditional information includes processing and properties (quality, taste, toxicity, cautions, precautions, etc.), among other historical data. A review of historical and classic works on CMM, including this one has been published (Leung, 1990).
Zhonghua Bencao (The Chinese Herbal)
 (Zhonghua, 1999)
(Zhonghua, 1999)
This monumental work consists of 10 volumes, totaling 9,282 pages, with 8,534 illustrations and 8,980 CMM monographs. This herbal was compiled by the Chinese State Administration of TCM involving over 500 scholars/experts from at least 60 academic and research institutions, and took 10 years to complete. For those who want lesser details, there is also an abridged 2-volume version available. The modern literature covered is up to 1994. It is the most extensive documentation of CMM to date. An English review of this work has been published (Leung, 2001). More recently, as part of the same work, two separate volumes dealing with ethnic
Major Contemporary Works Focused on Toxicities of CMM
Modern works on CMM toxicity documentation are primarily based on compilations of data generated from sources unrelated to traditional CMM, especially those from isolated chemicals, unverified or improperly characterized materials, uses out of context, etc. These have been increasing exponentially during the past decade and are still continuing. Unfortunately, they are not useful for the evaluation or validation of the safety and efficacy of traditional CMM, but are only relevant for use in the development of new chemical drugs for specific indications unrelated to traditional practice. However, these data are so pervasive that it is sometimes difficult to tell the data that are the result of premature scientific interpretations or extrapolations by investigators (in an attempt to confirm traditional data) and those that are traditionally derived. The examples of contemporary works selected for this review are ones that are dedicated to toxic CMM based primarily on data from historical sources and those continually accumulated from ongoing TCM practice, along with a certain amount of modern animal data that may or may not have been extrapolated to apply to humans. As the main purpose of this review is to provide as true a traditional perspective as possible on CMM toxicity, only a handful of publications in scope have been found suitable for inclusion.
Duju Zhongyao Gujinyong (Toxic Chinese Materia Medica: Past and Present)
 (Yang, 1991)
(Yang, 1991)
This 404-page work describes the history of toxic Chinese drugs, the prevention and treatment of their toxic effects, etc., with detailed monographs on 435 CMM.
Youdu Zhongcaoyao Da Cidian (Encyclopedia of Toxic Chinese Materia Medica)
 (Guo, 1992)
(Guo, 1992)
This work (853 pages) provides monographs on 503 traditional drugs (420 botanical, 50 animal, 33 mineral) along with data on 1,300 formulas; rating of toxicity as “extremely toxic” with lethal dose under 1g (22 drugs), “highly toxic” with very close therapeutic to toxic doses (50 drugs), “toxic” with relatively close therapeutic to toxic doses (230 drugs), and “slightly toxic” with big differences in therapeutic and toxic doses (201 drugs); some modern scientific data are provided.
Duyao Bencao (Toxic Drugs Herbal)
 (Yang, 1993)
(Yang, 1993)
This work (1,119 pages) describes in detail 903 toxic traditional drugs (55 of which not previously considered toxic), including both traditional and modern data; a good source for a quick review of any CMM as to its recorded toxicity, contraindications, cautions, precautions, and/or incompatibilities.
Youdu Zhongyao Xiandai Yanjiu Yu Heliyingyong (Toxic Chinese Materia Medica: Modern Research and Appropriate Applications)
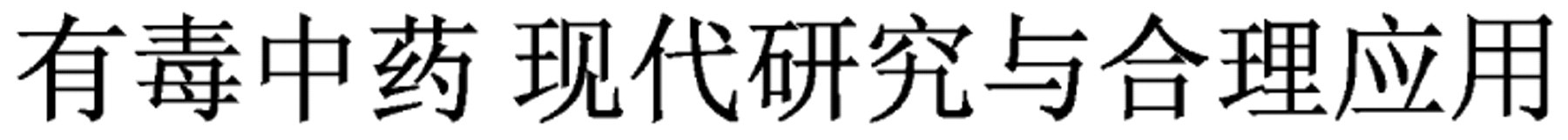 (Du and Fang, 2003)
(Du and Fang, 2003)
This work (1,029 pages) provides detailed monographs on 98 toxic traditional drugs, describing them from both historical/traditional and modern scientific perspectives; includes chemical (contamination by heavy metals, agricultural chemicals, pesticides, etc.), pharmacologic and clinical practice data (including use of zhu sha (HgS) for epilepsy in children), etc.
Xiandai Zhongyao Dulixue (Modern Toxicology of Chinese Materia Medica)
 (Xia, 2005)
(Xia, 2005)
This 934-page work describes 1,462 commonly used traditional drugs that have been recorded with toxic or potential toxic effects, cautions, and/or precautions, in any publication (ancient or recent) on CMM. In addition to the usual information on botanical sources, collection and processing, tastes and quality, indications, dosages, etc., most monographs also provide information on historical toxicity data, chemical composition, toxicology, and comments based on historical and modern data. Many also give methods and advice for preventing toxic effects, including heat processing, processing with other herbs, adherence to TCM tradition, and traditional recommended dosages, among others. Although there are some traditional toxicity data (obtained through TCM practice) in the “Toxicology” section, most of the toxicity data were experimentally derived, based on literature from the 1985–2002 period. Their relevance to the true nature of the respective CMM cannot be ascertained without careful review of each source cited, which would be a monumental task. Nevertheless, this work has both a Chinese drug index and an index for Latin binomials, which make it quite easy to access information. It is a good quick source of traditional CMM toxicity information.
National and Regional Journals of TCM and CMM
As a part of an earlier review of general TCM and CMM resources, examples of numerous of the over 100 better-known journals mentioned were given (Leung, 1990). Since then, over a 16-year period, many of the less popular and less extensively circulated ones have come to the fore and many of the more prominent ones have also greatly expanded in scope and content. The following are examples of some well-known ones:
Zhongcaoyao (Chinese Traditional and Herbal Drugs)

Zhongchengyao (Chinese Patent Medicines)
Zhongyaocai (Journal of Chinese Medicinal Materials)
Shizhen Guoyi Guoyao (Lishizhen Medicine and Materia Medica Research)

Shizhen Guoyao Yanjiu (Shizhen Journal of Traditional Chinese Medicine Research)

Zhongyiyao Xuebao (Acta Chinese Medicine and Pharmacology)
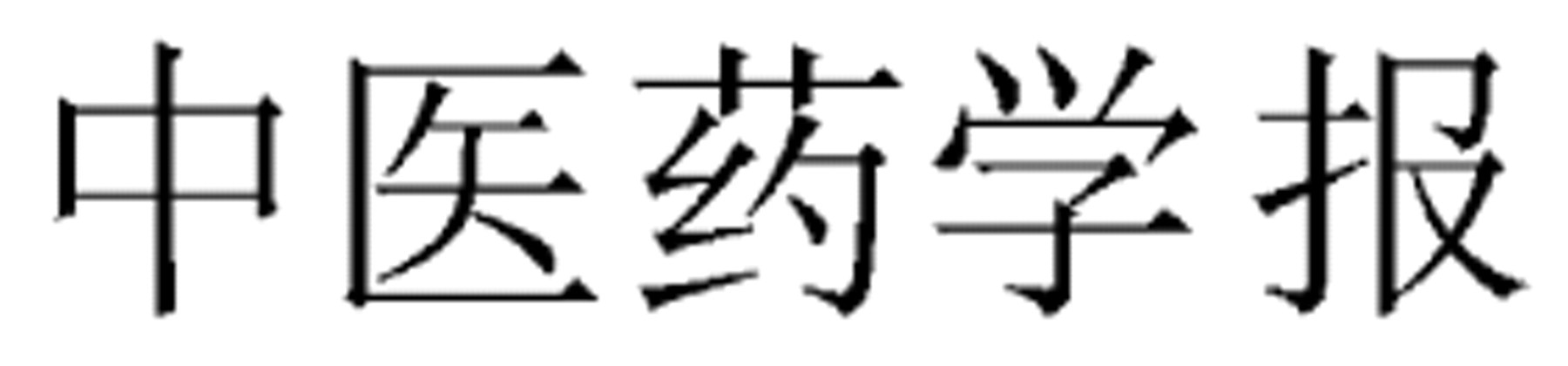
Zhongguo Zhongyao Zazhi (Chinese Journal of CMM)

Zhongguo Yiyao Xuebao (Chinese Journal of TCM and Pharmacy)
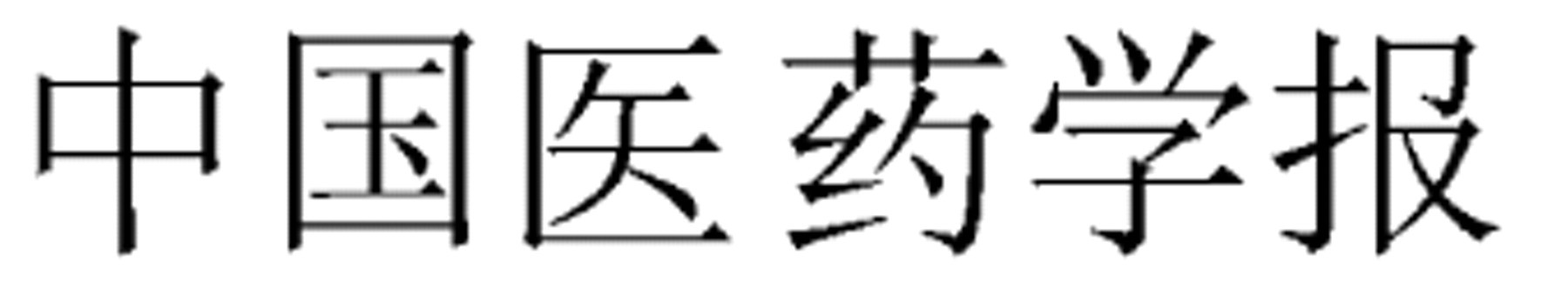
Beijing Zhongyiyao Daxue Xuebao (Journal of Beijing University of TCM)
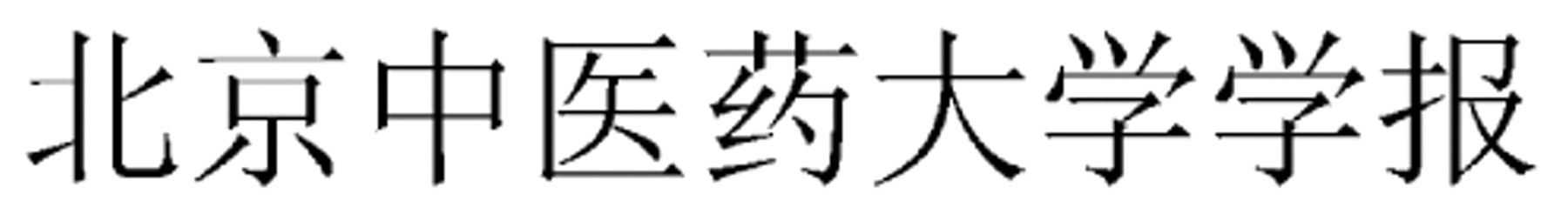
Chengdu Zhongyiyao Daxue Xuebao (Journal of Chengdu University of TCM)
Nanjing Zhongyiyao Daxue Xuebao (Journal of Nanjing University of TCM)
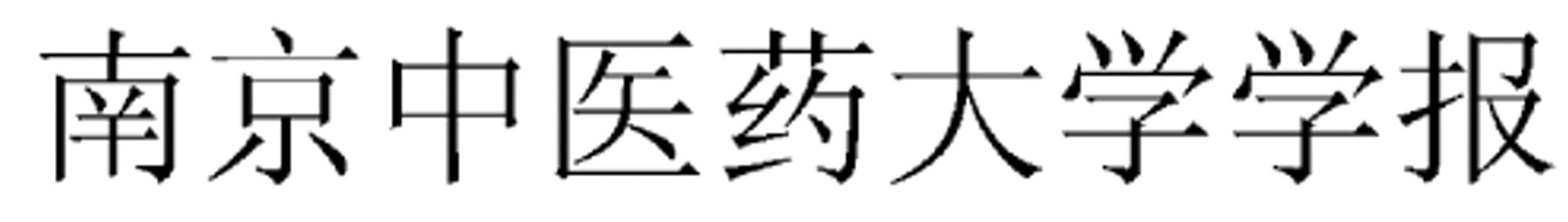
Shanghai Zhongyiyao Daxue Xuebao (Acta Universitatis Traditionis Medicalis Sinensis Pharmacologiaeque Shanghai)

Zhongyi Zazhi (Journal of TCM)
Yaoxue Xuebao (Acta Pharmaceutica Sinica)

Traditional toxicity information reported (based on traditional clinical practice with CMM) can be found especially in several dozen regional journals of TCM most of which have an annual index, with the best ones containing key words in Pinyin corresponding to their respective potential Chinese characters and the worst simply titles of the articles in that issue. In any case, it is not a major task to locate toxicity information if the journal resources are available in a single location. However, few of the major institutions in the United States, including the National Library of Medicine, have such resources, as they concentrate on acquisitions of journal titles geared at conventional medical and pharmaceutical interests that typically avoid TCM, as reported and practiced in the traditional manner in China for the past 3,000 years. Some toxicity information may occasionally appear in national journals like the Zhongyi Zazhi (Journal of TCM, which has an English edition), but the chances of such information also appearing in the English version are rather small, because typically not all information in Chinese publications enters the translated version, not only in this journal but also in other major works like the Chinese Pharmacopoeia (Pharmacopoeia, 2000), for whatever reason.
At present, it is unfortunate that in order to access ongoing traditionally acquired toxicity-data relevant to CMM, one cannot rely on most existing English sources (e.g., databases, institutions, books, and journals) due to the fact that most non-Chinese toxicity data are not due to the intact traditional CMM but to their specific, presumed active chemical constituents, which are generally irrelevant to the toxicity information one seeks for any particular traditional intact CMM. However, if one is at least aware of this problem, one will be able to ask the right questions when engaging a special consultant’s help in accessing such information. It is hoped this review can provide the reader with the basics for doing so.
Summary
Contrary to the common belief among the scientific and medical communities, traditional CMM toxicity data exists and is well-documented in the Chinese literature for over 3,000 years. However, only a minute fraction of this information has been translated into English, making it extremely difficult for non-Chinese-reading scientists and other interested professionals to access such information. This situation is not likely to change as experts in CMM or TCM who are proficient in English are dwindling instead of increasing, due to the continued misguided direction in efforts of modernizing TCM which equates it to evaluating CMM for their presumed active principles. This has greatly hindered true research efforts in the evaluation of the safety and efficacy of traditional CMM known for their traditional values which have been well-tested and well-documented over time. It is hoped that the basic information provided in this review will allow researchers, who are interested in CMM for their truly traditional values, to be aware of the deficiencies and pitfalls in relying on current English information on CMM toxicity and thus will start obtaining the desired and relevant results they have long been seeking.
