Abstract
The aim of the present study was to review the sexual side-effects of contemporary antidepressants in Australia, comparing the selective serotonin re-uptake inhibitors (SSRIs) with venlafaxine, reboxetine, mirtazepine, duloxetine, bupropion, desvenlafaxine and agomelatine. Double-blind, randomized comparative studies of these antidepressants that included assessment of sexual dysfunction with validated rating scales in patients with major depressive disorder were identified from the literature using MEDLINE, EMBASE and PsychINFO databases. Bupropion and duloxetine caused significantly less sexual dysfunction than the SSRIs in short-term studies and reboxetine significantly less in both short- and longer term studies. Bupropion and agomelatine caused significantly less sexual dysfunction than venlafaxine. The evidence for mirtazepine having an advantage over the SSRIs is lacking and there are currently insufficient data for desvenlafaxine. Well-designed comparative studies of contemporary antidepressants with direct assessment of sexual side-effects as the primary outcome measure are scarce. Future studies should be randomized, double-blind, active controlled trials in sexually active subjects with major depressive disorder. There should be direct assessment of sexual function and depression using reliable, validated rating scales before and during treatment. Studies should assess treatment-emergent effects in patients with normal function and resolution of baseline dysfunction over treatment, in both the short and long term. Further research should compare available instruments for measuring sexual function, and include separate analyses of both remitters/non-remitters and male/female subjects.
Sexual dysfunction is an exceedingly common but underrecognized side-effect of antidepressants. It is one of the most important reasons that patients stop taking these medications, with or without their treating physician's knowledge. Typically, sexual symptoms emerge once the patient has experienced a remission of depression. The depressed patient usually presents with diminished or absent libido and becomes concerned about their sexual difficulties only when their depression improves and their libido recovers. They may at first attribute their sexual dysfunction to persistence of their illness or old age or difficulties with their sexual partner. Despite increased sexual education and openness in many societies, surprisingly, long-standing sexual taboos persist and many patients continue to be embarrassed to raise the issue of sexual difficulty with their treating medical practitioner. This is particularly the case if the patient and doctor are of different gender or cultural backgrounds. The sexual impairment may be partial rather than complete and they may feel that the symptoms do not warrant the attention of a busy doctor. Nonetheless, these symptoms may still be subjectively problematic enough to result in the patient discontinuing their medication.
It is likely that sexual side-effects occur quite early, probably within a few days of treatment but only come to a patient's attention later on. Selective serotonin (5HT) re-uptake inhibitors (SSRIs) and other antidepressants are now being prescribed for an increasing number of psychiatric conditions, in particular, the anxiety disorders. Such patients may not have had pre-existing sexual dysfunction and may therefore become aware of newly emergent sexual difficulties sooner than the traditional SSRI patient who is diagnosed with major depressive disorder (MDD).
Sexual physiology
The parasympathetic (pelvic splanchnic nerves) and sympathetic nervous systems act synergistically to cause penile tumescence by relaxing the smooth muscles of the penile corpora cavernosa, allowing the penile arteries to dilate. Clitoral engorgement and vaginal lubrication also result from parasympathetic stimulation that increases blood flow to genital tissue. The sympathetic (adrenergic) system is responsible for ejaculation in men and contraction of the vagina, urethra, and uterus during orgasm in women.
Several neurotransmitters are known to be involved in the sexual response. Dopamine (DA) increases libido and promotes erection while serotonin inhibits ejaculation and orgasm. The post-synaptic 5HT2C receptors in the spinal cord, when activated, inhibit orgasm in the male and the female and may cause anorgasmia. Stimulation of β-adrenergic receptors enhances erections whereas α1 receptors when activated reduce tumescence. Nitric oxide (NO) causes relaxation of the smooth muscle and may be involved in arousal in both sexes [1].
Types of sexual problems
Assessment of sexual disorders can be complex because many contributing factors may be involved. Detailed diagnosis, classification and management of sexual disorders are beyond the scope of the article. Briefly, disorders of sexual functioning are usually classified into four categories: (i) sexual desire disorders, in which the individual has little or no interest in a sexual relationship and there is a partial or total lack of libido; (ii) sexual arousal disorders, in which the individual has the sexual interest or drive but cannot attain sexual arousal; (iii) orgasm disorders; and (iv) sexual pain disorders. The first two can be a primary condition in which the individual has never had a satisfactory sexual relationship. Alternatively and more commonly, sexual dysfunction can be a secondary disorder and follow a period of normal sexual functioning. Sexual arousal disorders include problems with erectile dysfunction in men or lack of vaginal lubrication in women and may be due to either antidepressants or other physical or psychological causes. Category 3, the orgasm disorders, involve a persistent delay or absence of orgasm following a normal sexual excitement phase affecting both women and men. The SSRIs are a frequent but not the only cause of such problems. They and other medications can delay the achievement of orgasm or eliminate it entirely. In men this results in either delayed or failure in ejaculation. The other common disorder of orgasm occurring in men is premature ejaculation where only a brief period of sexual excitement leads to premature ejaculation. The SSRIs are sometimes used to treat this condition. Category 4, the sexual pain disorders, affect women almost exclusively, and are known as dyspareunia (painful intercourse) and vaginismus (an involuntary spasm of the muscles of the vaginal wall, which interferes with intercourse). Dyspareunia may be caused by insufficient arousal or lubrication (vaginal dryness) in women. These are typically not directly caused by antidepressants, although insufficient lubrication (due to decreased libido or arousal) may lead to painful intercourse.
Sexual problems attributable to antidepressants
Depressive disorder itself is associated with disturbed sexual functioning. Estimates of prevalence rates of sexual dysfunction in depression vary due to different study designs, cultural mores and the complexity of studying sexual functioning. Recent reviews suggest that the prevalence of sexual dysfunction in major depression is between 35% and 50% [2] but may be as high as 70% [3]. Sexual desire tends to improve with alleviation of depression but may occur in tandem with the development of detrimental effects of the antidepressant treatment on sexual function.
Until the advent of the SSRIs, there was little research regarding sexual function and antidepressants. In fact, it has been only in the last two decades that this topic has been given much attention. The relative effects of antidepressants on sexual function are likely to be due to their action on specific neurotransmitter systems [3]. The older antidepressants such as the tricyclic antidepressants (TCAs) and monoamine oxidase inhibitors (MAOIs) have effects on several neurotransmitter systems, hence these agents were associated with many side-effects that possibly overshadowed sexual side-effects. Estimates of prevalence rates of sexual side-effects vary widely [3]. When the SSRIs were first introduced, their side-effects were underestimated [2]. Studies have reported the incidence of sexual side-effects associated with the use of the SSRIs varying from 24% to 73% of treated patients [3]. The wide range of findings can be attributed to differing methodologies, with those studies using more sensitive questionnaires and assessing more subtle symptom changes, reporting higher frequencies. In one study, the incidence of spontaneously reported SSRI sexual side-effects was only 14%, whereas 58% of patients acknowledged sexual dysfunction following direct questioning [4].
It is likely that the higher figures are more accurate and we need to assume that more than half of the patients who are taking an SSRI experience a sexual side-effect of one form or another. It is important, however, to determine which sexual symptoms have emerged during the course of treatment, while excluding pre-existing sexual problems due to depression or other comorbid conditions. Because sexual problems are such a common symptom of depression, as are other comorbidities that may cause sexual problems, determining the exact frequencies of sexual problems being caused by the antidepressants is fraught with methodological difficulties. It is important to determine which are treatment-emergent symptoms, excluding those that pre-date treatment. The ongoing persistence of sexual dysfunction such as a lack of libido may be due to partially treated depressive symptoms rather than a drug effect. Loss of libido is a common symptom of depression that usually improves with successful treatment. To accurately determine whether sexual side-effects are due to drug side-effects, patients must be randomly assigned in placebo-controlled trials, with concurrent measurement of depression and sexual function before and during treatment. A recent careful review of the extant literature concluded that all SSRIs were associated with sexual side-effects [2]. These include a loss of libido in approximately 20% of patients [5], and erectile dysfunction in approximately 10% of patients [6]; the most common side-effect was anorgasmia or delayed orgasm in women and ejaculatory failure or delayed ejaculation in men, which occurred in 30–40% of patients [2].
There are also some rare occurrences of other sexual problems such as priapism reported with paroxetine, sertraline, fluoxetine, citalopram, and trazodone [2]; isolated cases of increased sexual desire and spontaneous orgasm associated with clomipramine and SSRIs [7, 8], and persistent genital arousal disorder associated with the use of or withdrawal from SSRIs [9].
Comorbid sexual dysfunction not attributable to antidepressants
Comorbid sexual problems in patients who are treated with an antidepressant are common. The sexual problem can be a symptom of the illness itself, usually depression, or the patient may have a comorbid sexual disorder. A careful history dating the onset and nature of symptoms is essential. A patient with borderline personality disorder may have experienced childhood sexual abuse and have profound relationship problems, including long-standing sexual difficulties, such as vaginismus. The individual with a severe social phobia may never have had a sexual relationship and their sexual difficulty may be due to both ongoing anxiety and a lack of sexual experience. One of the most common causes of loss of libido and erectile dysfunction in psychiatric patients is comorbid alcohol abuse. Patients will typically not report this unless specifically questioned. Other forms of substance abuse, such as opiates, amphetamine, and marijuana may also lead to sexual dysfunction, usually a loss of libido.
Sexual desire disorders (decreased libido) may be caused by a decrease in the normal production of oestrogen (in women) or testosterone (in both men and women). In women, menopause and aging results in thinning of the mucous membrane of the vagina and loss of lubrication, resulting in dyspareunia. Comorbid physical illness, such as diabetes and degenerative neurological disorders are common causes because they affect the neurological pathways controlling the sexual response, resulting in anorgasmia, erectile dysfunction and/or ejaculatory failure. Any serious physical illness may impact on libido and sexual functioning. Disfigurements, such as burns or mastectomy, will also impact on an individual's perception of themselves as sexually desirable. Prostate problems in men, particularly total prostatectomy for prostate cancer, commonly result in erectile dysfunction (58%) [10] but are less common following surgery for benign prostatomegaly (13%) [11].
For optimal sexual function, in general, excellent physical and psychological health is required, whereas impairment in either is likely to result in sexual dysfunction. Ongoing relationship problems, difficulty maintaining relationships, or chronic disharmony with the current sexual partner will also interfere with sexual function. Antidepressants are not the only medications that affect sexual function. Most notably, the antipsychotics can affect sexual function probably by blocking dopamine, raising prolactin levels or possibly by their effects on other neurotransmitters or hormones. Other medications, such as benzodiazepines, antihypertensives (beta-blockers, angiotensin-converting enzyme (ACE) inhibitors, and calcium channel blockers), anti-epileptics (including those used as mood stabilizers), prostate medications and recreational drugs may all adversely affect sexual function [12].
Are all antidepressants the same?
There is some evidence that paroxetine may be more likely than other SSRIs to cause sexual dysfunction and that fluvoxamine may have some modest advantage [4, 13]. The mechanism by which the SSRIs induce sexual dysfunction is probably multifactorial but appears to be primarily related to their effect on serotonin through stimulation of 5HT2C receptors [1]. The older antidepressants, such as the TCAs and the MAOIs, have not been well studied but existing data suggest that they are not free of sexual side-effects [2, 3]. In fact, the most serotonergic TCA, clomipramine has been reported to cause anorgasmia or a marked delay in orgasm in up to 90% of subjects [14]. In contrast the reversible MAOI, moclobemide, has a reported very low incidence of sexual dysfunction (24%) compared to the SSRIs (62%) [15].
The prevalence of sexual dysfunction related to newer antidepressants was assessed in a cross-sectional, observational study of more than 6000 subjects from US primary care clinics [16]. In the overall population the prevalence was 37% and ranged from 22% to 43% across antidepressants. For those antidepressants taken by more than 500 patients, prevalence rates were significantly higher for paroxetine, venlafaxine XR, sertraline, fluoxetine and citalopram than for bupropion SR. Similar results were found in a subgroup without comorbid illness or concomitant medications that might be expected to affect sexual function. The study also showed a trend towards dose-related increases in sexual dysfunction for some drugs. A comparison of patient-rated and physician-perceived prevalence rates found that physicians underestimated prevalence rates. Rates were also higher than those reported in product labelling because the studies reported in the latter tend to rely on spontaneous reports of adverse effects.
Only a small number of comparative studies of the newer antidepressants approved after SSRIs have specifically investigated sexual dysfunction. A recent review by Haberfellner found 79 randomized double-blind clinical trials comparing antidepressants introduced in Europe after 1995 in MDD [17]. Of these 75% relied on spontaneous reports for assessment of sexual dysfunction, 18% on non-specific side-effects checklists and only 8% used specific sexual function assessment instruments. Moreover, many were short-term studies that would not pick up the emergence of side-effects that typically follow remission of depression. For such reasons, previous studies may not reflect the prevalence of sexual dysfunction in clinical reality.
Alternative antidepressants to the SSRIs have become available in Australia over the past decade and there are new antidepressants that may soon become available. There are now two serotonin and noradrenergic re-uptake inhibitors (SNRIs) available, venlafaxine and duloxetine. Also, desvenlafaxine, the major metabolite of venlafaxine, has recently been approved in the USA and Australia. Mirtazapine has a unique mode of action while reboxetine is a relatively selective noradrenergic re-uptake inhibitor (NRI). Buproprion has an indication in Australia for nicotine addiction but not depression, while agomelatine is a melatonin agonist that has received marketing authorization in Europe and is expected to become available soon. The pharmacological profiles of the newer antidepressants are varied, which may lead to differential effects on sexual function.
Comparative studies of contemporary antidepressants
Double-blind, randomized controlled trials of antidepressants with assessment of sexual side-effects

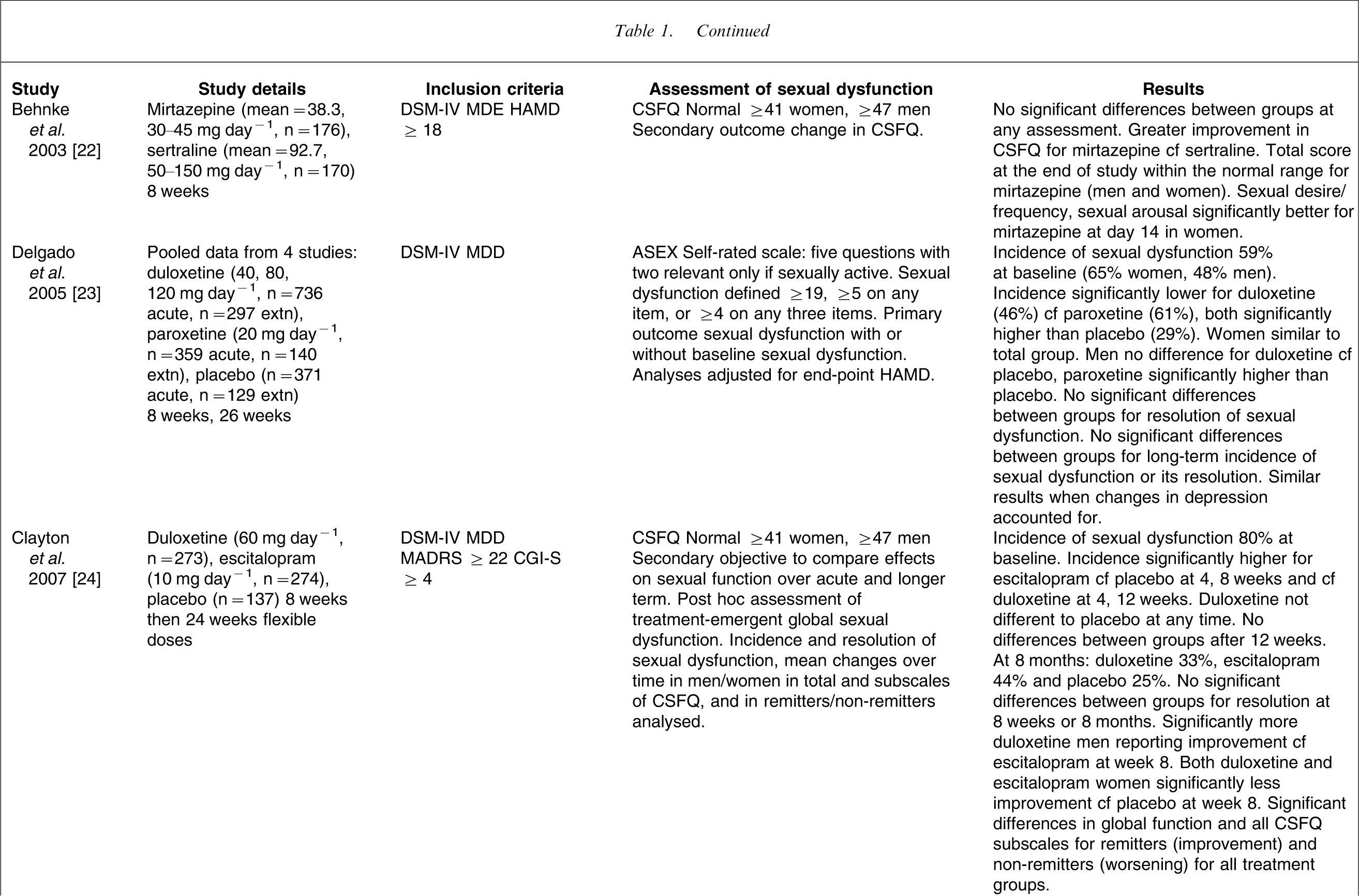
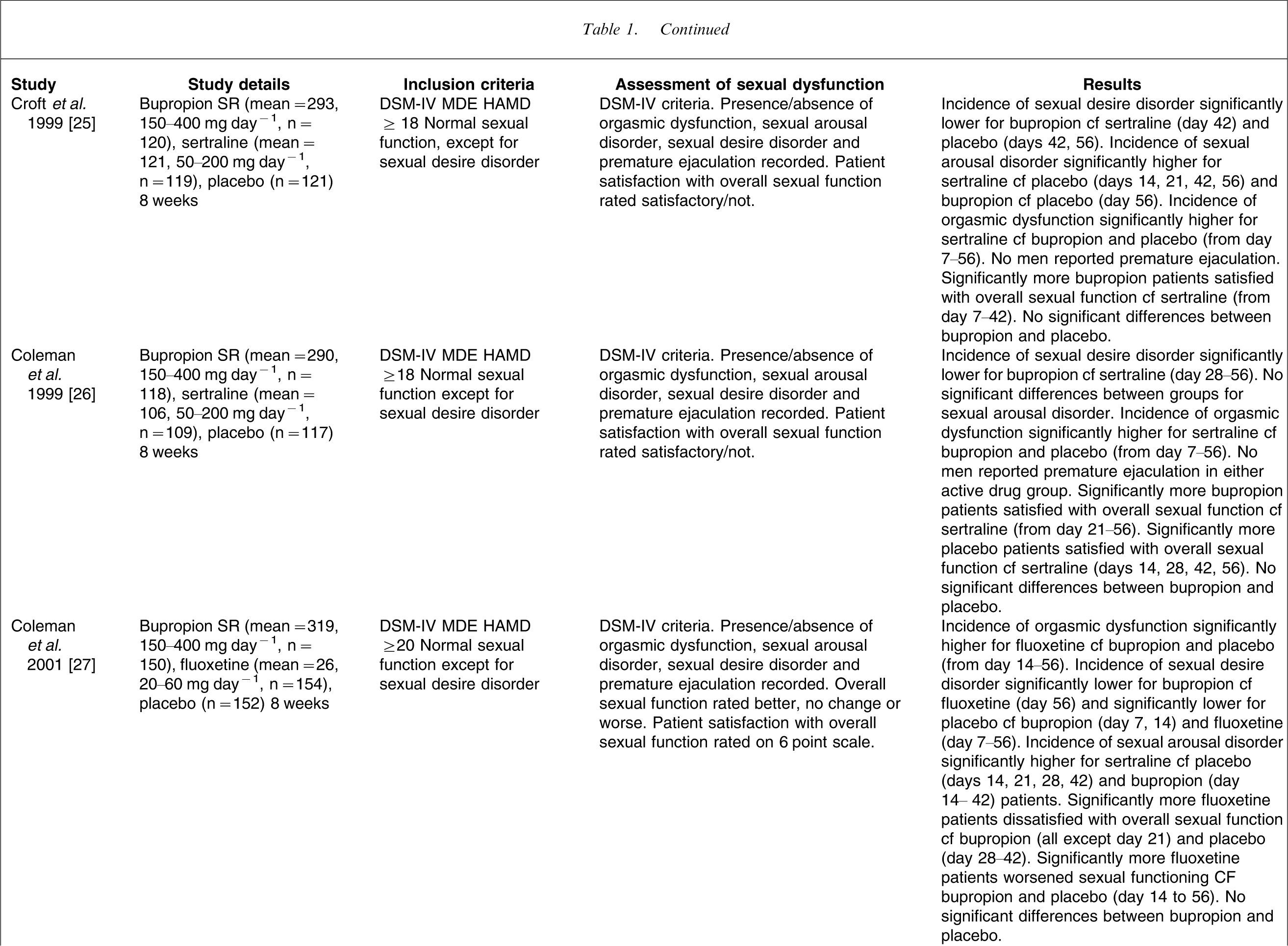

ASEX, Arizona Sexual Experience Scale; CGI-S, Clinical Global Impression of Severity; CSFQ, Changes in Sexual Functioning Questionnaire; extn, extension; HAMD, Hamilton Depression Rating Score; IRSD-F, Investigator-Related Sexual Desire and Functioning Scale; MADRS, Montgomery-Asberg Depression Rating Score; MDD, major depressive disorder; MDE, major depressive episode; pbo, placebo; RSI, Rush Sexual Inventory; Sex-FX, Sexual Function Scale, SF, Sexual Function Scale–Swedish version.
Venlafaxine
Venlafaxine was the first SNRI introduced in Australia. It is more potent in inhibiting 5HT than noradrenaline (NA) and is selective for 5HT at low doses. It is a weak inhibitor of DA re-uptake but has no significant affinity for muscarinic cholinergic, H1-histaminergic, benzodiazepine, opioid or α1-adrenergic receptors and does not inhibit MAO. There are no large studies comparing venlafaxine to any of the SSRIs with direct assessment of sexual dysfunction as a primary objective. One small comparative study of venlafaxine with paroxetine, sertraline and moclobemide found that rates of sexual dysfunction for venlafaxine were between those for moclobemide and the SSRIs [30].
Two studies comparing venlafaxine with newer antidepressants with assessment of sexual function as the primary outcome measure are included in Table 1. The first study compared the extended release formulations of venlafaxine and bupropion [18]. That study found that bupropion had a more favourable sexual side-effect profile than venlafaxine, spanning all subscales of the Changes in Sexual Functioning Questionnaire (CSFQ). Sexual function worsened with venlafaxine in both men and women and in subjects with normal sexual function at study entry but was improved or unchanged with bupropion. The differences were both statistically and clinically significant. The second study compared venlafaxine with agomelatine [19]. That study found that treatment-emergent sexual dysfunction was greater for venlafaxine than for agomelatine. There was significantly greater deterioration in sexual function for venlafaxine in both sexes and in both the total patient set and those subjects whose depression had remitted. Venlafaxine was associated with more sexual side-effects than the comparators in both these studies.
Reboxetine
Reboxetine is a selective NRI with little effect on 5HT or DA re-uptake, does not inhibit MAO and has low affinity for adrenergic, cholinergic, histaminergic, dopaminergic, serotonergic (including 5HT2) and muscarinic receptors. It is one of the few antidepressants with almost no effect on the serotonergic system. Three studies focusing on sexual dysfunction during treatment with reboxetine have been published. Reboxetine was similar to placebo and significantly superior to fluoxetine in overall sexual satisfaction in both male and female subjects (Table 1) [20]. In male patients, however, significant differences in favour of fluoxetine and placebo over reboxetine were found, which may reflect a noradrenergic effect on male arousal. Reboxetine was similar to placebo and fluoxetine worse than placebo in both clinical responders and non-responders, suggesting that differential effects of the two antidepressants were independent of improvement in depression.
A second study reported on the UK data from an international multicentre trial and examined the resolution of sexual dysfunction with reboxetine and paroxetine treatment over 8 weeks using the visual analogue items of the Rush Sexual Inventory [31]. Sexual function and satisfaction improved with both treatments over time with some significant differences in favour of reboxetine, but due to the small sample size (20–30) this needs confirmation in the total study population. Sexual function was investigated as part of an efficacy and tolerability comparison of reboxetine and citalopram (Table 1) [21]. Reboxetine was associated with significantly improved sexual function compared to citalopram, particularly in the later stages of this longer term study. In addition orgasmic dysfunction was the most common adverse event reported for citalopram. Overall, reboxetine appears to have less effect on sexual function than the SSRIs over the short and long term in the aforementioned studies as expected, with the exception of a possible noradrenergic effect on male arousal. There are no studies comparing reboxetine with the other newer antidepressants.
Mirtazepine
Mirtazepine is the first noradrenergic and specific serotonergic antidepressant (NaSSA) that enhances central noradrenergic and 5HT1 neurotransmission while directly blocking 5HT2 and 5HT3 receptors. There have been no double-blind comparative studies with the SSRIs that were specifically designed to primarily evaluate sexual side-effects. Three randomized, controlled efficacy studies with secondary assessment of sexual side-effects comparing mirtazepine with SSRIs [22, 32, 33] and one open naturalistic study have been published [34]. Behnke
An open 6 month naturalistic study evaluated the effects of mirtazepine on sexual function in depressed patients using the Psychotropic-Related Sexual Dysfunction Questionnaire (PRSexDQ) [34]. There was a significant reduction in mean total PRSexDQ scores, with 71% of patients returning to normal functioning by 6 months. The incidence of sexual dysfunction was lower in the remitter group at the end of the study compared to the overall group [34]. Mirtazepine, despite its 5HT2-blocking action, does not appear to have any major advantage over the SSRIs, but this requires confirmation in studies specifically designed to primarily evaluate sexual side-effects. There are no comparative studies with the newer antidepressants.
Duloxetine
Duloxetine is an SNRI with greater affinity for 5HT and NA re-uptake transporters than venlafaxine, with a more balanced NA/5HT ratio. It has low binding to the DA transporter, and little affinity for muscarinic, histamine H1, α1-and α2 adrenergic, muscarinic, serotonergic, dopamine D2 and opioid receptors and no MAOI activity. Several comparative studies with SSRIs that were primarily efficacy studies have measured changes in sexual function as part of the tolerability profile. Delgado
Clayton
Duloxetine was associated with a lesser effect on sexual function than the comparison SSRIs in the acute phases of both these studies but not in the longer term. This may be due to its more favourable NA/5HT re-uptake ratio. Longer term studies have methodological problems due to small numbers, which may be further compounded by the dropout of subjects experiencing significant sexual side-effects. Thus power to compare between active drugs and placebo may be compromised [24]. Again there are no comparative studies between duloxetine and the newer antidepressants.
Bupropion
Bupropion is an NA and DA uptake inhibitor (NDRI). It has no clinically significant effect on serotonergic neurotransmission or on muscarinic, histaminergic, dopaminergic, or adrenergic receptors. Three similar studies and a meta-analysis including these and other studies comparing sexual dysfunction after treatment with bupropion and the SSRIs, sertraline and fluoxetine, have been published [25–27, 35]. In the individual studies (Table 1) bupropion was found to have significantly less effect on sexual functioning than either sertraline or fluoxetine. The largest effect of the SSRIs was on orgasmic dysfunction, starting early in treatment (weeks 1 or 2) and occurring in responders and remitters [27], suggesting that it was not related to continuing depressive symptoms. The higher rate for the SSRIs did not appear to be a dosage effect because 20 mg day−1 fluoxetine produced greater dysfunction than 400 mg day−1 bupropion [27]. Overall, incidences of orgasmic dysfunction were three times higher for the SSRIs (37%) compared to both bupropion and placebo; incidences of sexual arousal disorder for the SSRIs twice (11%) that of either bupropion or placebo; and incidences of sexual desire disorder for the SSRIs 1.5 times higher than for bupropion or placebo [35].
Two other studies have compared bupropion with the SSRIs escitalopram [28] and paroxetine [29]. The first report included pooled data from two identical trials [28]. The incidence of orgasm dysfunction was again higher for escitalopram and of a similar rate (30%) to sertraline and fluoxetine, and was considered of clinical significance based on CSFQ changes. The relative risks for treatment-emergent orgasm dysfunction compared to placebo were 1.2 and 2.0 for bupropion and 2.9 and 3.6 for escitalopram, respectively, for the two individual studies. The second was a smaller study of bupropion and paroxetine that compared two different assessments of sexual function [29]. There was a gender difference both before and after treatment (Table 1). There was a significant correlation between the Sexual Function Scale (Sex-FX) and the Investigator-Related Sexual Desire and Functioning scale (IRSD-F) at all visits and there was a significant inverse relation between depression and sexual function in women but not in men.
There has been only one study comparing bupropion with a newer antidepressant, venlafaxine [18]. As detailed in Table 1 and, as already noted, bupropion had a significantly better sexual tolerability profile than venlafaxine in both men and women across all subscales of the CSFQ. These well-designed studies clearly show that bupropion is similar to placebo in its effect on sexual function and is superior to sertraline, fluoxetine, escitalopram, paroxetine and venlafaxine.
Desvenlafaxine
Desvenlafaxine is the major metabolite of venlafaxine and similarly is more potent in inhibiting 5HT than NA and DA and may also be serotonergic at low doses. It has little effect on other receptor systems (muscarinic cholinergic, H1 histaminic, α1 adrenergic) or MAO inhibition. No studies have yet evaluated sexual function using appropriate sexual function questionnaires. One report has pooled the data from two similar studies comparing desvenlafaxine (200–400 mg day−1) with venlafaxine and placebo [36]. Treatment-emergent adverse events incidences for impotence were desvenlafaxine 9%, placebo 1%, venlafaxine (75–150 mg day−1) 6%, and venlafaxine (150–225 mg day−1) 11%. Similar sexual side-effects might be expected for venlafaxine and desvenlafaxine given the similarity in pharmacological profile. Further studies are required to compare desvenlafaxine with other new antidepressants.
Agomelatine
Agomelatine is a melatonergic (MT1 and MT2 agonist) antidepressant with 5HT2C antagonist properties. There are no studies comparing agomelatine with any of the SSRIs in MDD with a primary focus on sexual side-effects, but one study in healthy volunteers has been reported [37]. Agomelatine 25 mg day−1, 50 mg day−1, paroxetine 20 mg day−1 and placebo were compared over 8 weeks in healthy men with no sexual impairment prior to the study. Sexual dysfunction was rated using the PRSexDQ and the International Index of Erectile Function. Both the agomelatine and placebo groups had a significantly lower incidence of treatment-emergent sexual dysfunction at week 8 than paroxetine for both measures. One study has compared agomelatine with venlafaxine [19] (Table 1) and, as already noted, agomelatine had a superior sexual side-effects profile compared with venlafaxine. The 5HT2-blocking action of agomelatine may explain the favourable sexual side-effect profile of this novel antidepressant. In addition, there is evidence that melatonin agonism may have a positive influence on sexual behaviour [19]. Further comparative studies with the other new antidepressants are required.
Management of sexual side-effects
Management of sexual side-effects during treatment with antidepressants is rarely simple and straight forward. In many cases, comprehensive patient education is vital to address the possibility of sexual side-effects to minimize non-adherence of treatment and subsequent relapse of depression. A thorough assessment of sexual symptoms is necessary to tease out if such symptoms pre-date the treatment or if they are secondary to other comorbid physical or psychosocial conditions. In particular, a sensitive approach to exploring the relationship issues is necessary to identify significant conflict and intimacy issues. Couple counselling may be an appropriate measure to address relationship problems that may contribute to sexual dysfunction. Primary sexual disorders may require referral for specific treatment by a sex therapist. In the presence of physical disorders, the treatment of the underlying condition can often alleviate or improve sexual symptoms. Rationalization of psychotropics and other medications especially in the case of polypharmacy may be an effective solution.
In terms of changing antidepressant treatment, reducing the dose of the antidepressant may be helpful because some studies have shown increases in sexual side-effects with higher doses [16], although this has not been consistently found [24]. Drug holidays, such as not taking the antidepressant on the morning of the day that sexual activity is planned, may help but probably not in the case of antidepressants with long half-lives. It may jeopardize compliance with the medication and does not allow for spontaneity in sexual activity. Switching to a different antidepressant, preferably one with less effect on sexual function, may be attempted, although this always has the attendant risk of relapse of depression because no two antidepressants have identical actions. Also sexual side-effects may always be replaced with different side-effects by the new antidepressant.
Addition of another agent that reverses the sexual side-effects has been attempted, also with varying levels of success. These approaches rely on the added agent reversing the pharmacological effect of the offending antidepressant that is causing the sexual dysfunction. There are anecdotal reports that reversal of negative sexual side-effects has been achieved with cyproheptadine, an antihistamine with anti-serotonergic effects; amantadine, a dopamine agonist; yohimbine, a central α2-adrenergic receptor antagonist; methylphenidate and dextroamphetamine, which are dopaminergic and have adrenergic effects; and buspirone, a 5-HTA agonist [2, 3]. The phophodiesterase inhibitors (PIs), however, such as sildenafil, vardenafil and tadalafil, have been shown by double-blind randomized placebo-controlled trials to be effective in overcoming erectile dysfunction and orgasmic problems induced by SSRIs in men [3, 38] and in reducing adverse sexual effects including reversal of anorgasmia in women [3, 39]. Adverse events were similar in these trials to those found for the general population, suggesting that combined use of PIs and SSRIs can be safely recommended. A review of strategies for managing antidepressant-induced sexual dysfunction found 15 randomized, controlled trials involving 16 different interventions of which the evidence base for the use of sildenafil for men with antidepressant-induced erectile dysfunction is the largest and most consistent [40].
Conclusion
Sexual side-effects from antidepressants remain a common problem and are a frequent cause of poor adherence. In individual patients it is important to tease out sexual dysfunction secondary to the depressive illness or that due to a comorbid condition from that being caused by the antidepressant. The studies listed in Table 1 indicate that the newer antidepressants have varying effects on sexual functioning, but there are few studies for each drug and there are still methodological deficiencies in some studies relating to placebo control, clinical significance of differences in sexual dysfunction, and a lack of longer term studies. From the data to date the propensity of each antidepressant to cause sexual side-effects is as follows: venlafaxine similar to the SSRIs and worse than either bupropion or agomelatine; reboxetine better than the SSRIs in both short- and longer term studies; mirtazepine similar to the SSRIs; duloxetine better than the SSRIs; bupropion better than the SSRIs and venlafaxine (and is the most investigated agent); agomelatine better than venlafaxine and thus expected to be better than SSRIs; and there are insufficient data for desvenlafaxine to comment. Thus, further comparative well-designed head-to-head trials of contemporary antidepressants are warranted. These should be randomized, double-blind, controlled trials in sexually active subjects with a DSM diagnosis of MDD. There should be direct assessment of sexual function and depression using reliable, validated rating scales before and after commencement of treatment. Further research comparing available instruments for measuring sexual function to determine the ideal assessment instrument is required because some discrepant results have been obtained [17]. More detail in reporting sexual dysfunction may be useful because summary scores may obscure differences between drugs that affect different aspects of sexual functioning. Studies should not only assess treatment-emergent effects in patients with normal function at baseline but also assess resolution of baseline dysfunction over treatment, in both the short and long term and after discontinuation of treatment. Separate analyses of both remitters and non-remitters and male and female genders appear important [24, 29]. The addition of medications, such as sildenafil, vardenafil and tadalafil, can counter some of the sexual side-effects of antidepressants and may be considered safe first-line agents. The authors recommend that further careful research is needed to investigate the nature of sexual dysfunction due to current antidepressant treatments.
Footnotes
Acknowledgements
Professor Schweitzer and Associate Professor Ng have served on pharmaceutical industry advisory boards over the past 3 years (Wyeth and Eli Lilly Advisory Boards). Associate Professor Chee has received research grant support from Wyeth and Lundbeck. Both have received honoraria for lectures and travel grants from pharmaceutical companies. This paper was supported by an unrestricted educational grant from Servier Laboratories.
