Abstract
Frontotemporal dementia (FTD) is the clinical term for patients who exhibit the behavioural variant of frontotemporal lobar degeneration (FTLD; for a review of current clinical and neuropathological classifications of FTLD see [1, 2]). The understanding of the causes of FTD advanced dramatically with the identification of tau mutations in families with FTD [3]. Subsequently, FTD patients who did not have genetic or neuropathological tau abnormalities were shown to exhibit ubiquitin-positive neuronal inclusions, so called FTD-u. TARDNA binding protein (TDP-43) is the principal component within ubiquitinated neuronal inclusions in FTD-u. This protein is normally present in neuronal nuclei and absent from the cytoplasm. In FTD-u, TDP-43 is no longer detectable in the nuclei but is seen in intracytoplasmic inclusions [4, 5]. Changes in TDP-43 expression have now been linked to sporadic and familial FTD and sporadic motor neurone disease [6] and have led to a new understanding of the clinical and neuropathological links between FTD and motor neurone disease.
In a recent study we described five subjects with young-onset FTD (mean age of onset = 35.6 years, range = 28–43 years), who presented with a schizophrenia-like psychosis [7]. The neuropathological diagnosis in these cases was that of FTD associated with motor neurone disease (i.e. TDP-43-positive FTD-u). In the second arm of that study we surveyed the presenting symptoms in 751 cases of FTD in the literature. We found that FTD one-third of patients who presented aged ≤30 and one-quarter who presented aged ≤40 had presented with a psychotic diagnosis.
These findings led us to undertake a pilot study to examine whether subjects with schizophrenia would exhibit abnormalities in hippocampal TDP-43 and ubiquitin. The current study focused on hippocampal dentate gyrus because it may be the earliest or only region to exhibit ubiquitin or TDP-43 abnormalities in FTD [8]. Few studies have investigated ubiquitin or TDP-43 in schizophrenia and none has reported on findings in the dentate gyrus. To date, studies have not found abnormalities of either TDP-43 [9] or ubiquitin [9, 10] in schizophrenia, although neither of those studies specifically examined hippocampal dentate gyrus. The current study examined post-mortem brains of patients with schizophrenia compared to control subjects and, in order to examine the specificity of any findings, patients with bipolar disorder.
Methods
Case selection
Subject characteristics
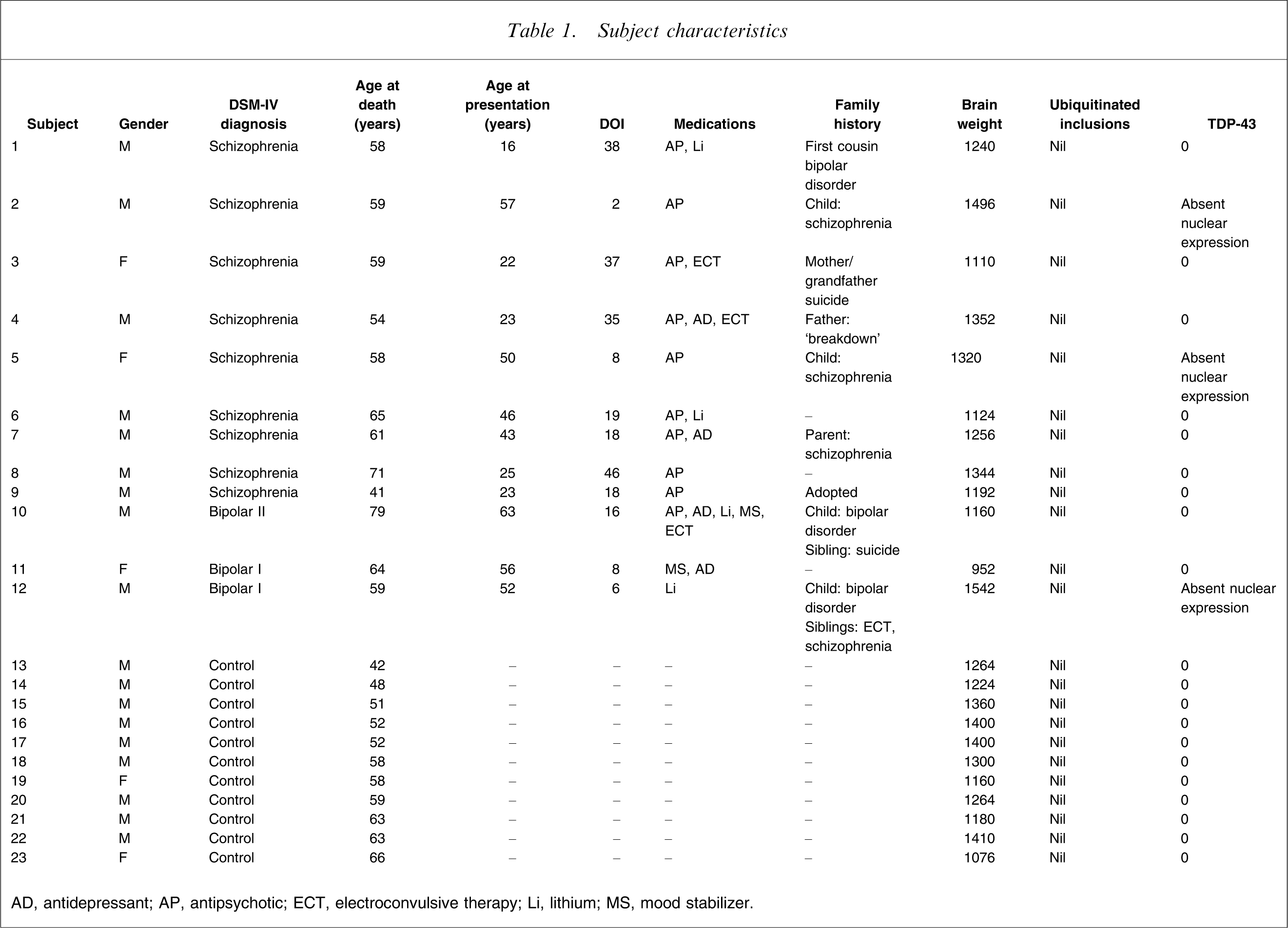
AD, antidepressant; AP, antipsychotic; ECT, electroconvulsive therapy; Li, lithium; MS, mood stabilizer.
Neuropathology and immunohistochemistry
All cases had been reported at autopsy and no subjects showed macroscopic or microscopic evidence of neurodegenerative, cerebrovascular or white matter disorder. Hippocampal sections (area B9/10) were obtained from the right hemisphere that had been fixed in formalin for 2 weeks. For immunohistochemistry, tissue sections were exposed to either a 1:200 dilution of a 1E8 (inhouse) antibody at 1:200, a 1:200 dilution of a Tau antibody (Dako, Carpinteria, CA, USA), a 1:100 dilution of a ubiquitin antibody (polyclonal rabbit, code no. Z0458; Dako) or a 1:8000 TARDBP antibody (clone 2E2-D3; catalog no. H00023435-M01; Abnova, Taipei, Taiwan).
A section of dentate gyrus hilus was photographed and rated blind to diagnosis for the presence or absence of a normal pattern of TDP-43 staining. In those cases of abnormal TDP-43 staining the proportion of neurones with abnormal staining was identified. The sections were examined for the presence or absence of ubiquitinated neuronal inclusions.
The study was approved by the Melbourne Health Mental Health Research and Ethics Committee. Following ethics approval, a tissue request application was approved by the Victorian Brain Bank Network.
Results
Demographic and clinical details
The study sample included 12 patients with psychosis (nine male, three female) and 11 control subjects (nine male, two female). The mean age of patients and controls at the time of death was 60.7±9.2 years and 55.6±7.3 years, respectively (Table 1). The age at first clinical presentation for the patients was 39.8±16.8 years and the duration of illness was 20.1±13.9 years. Ten subjects had been treated with antipsychotic medication during their lifetime and two had been treated with lithium or other mood stabilizers. Five patients had a first-degree relative with the same psychiatric diagnosis, while two subjects had a family history of suicide or unspecified mental illness.
TDP-43 and ubiquitin immunohistochemistry
Control subjects
All control subjects exhibited normal TDP-43 nuclear staining and no intraneuronal ubiquitin positive inclusions (Figure 1).
Patients
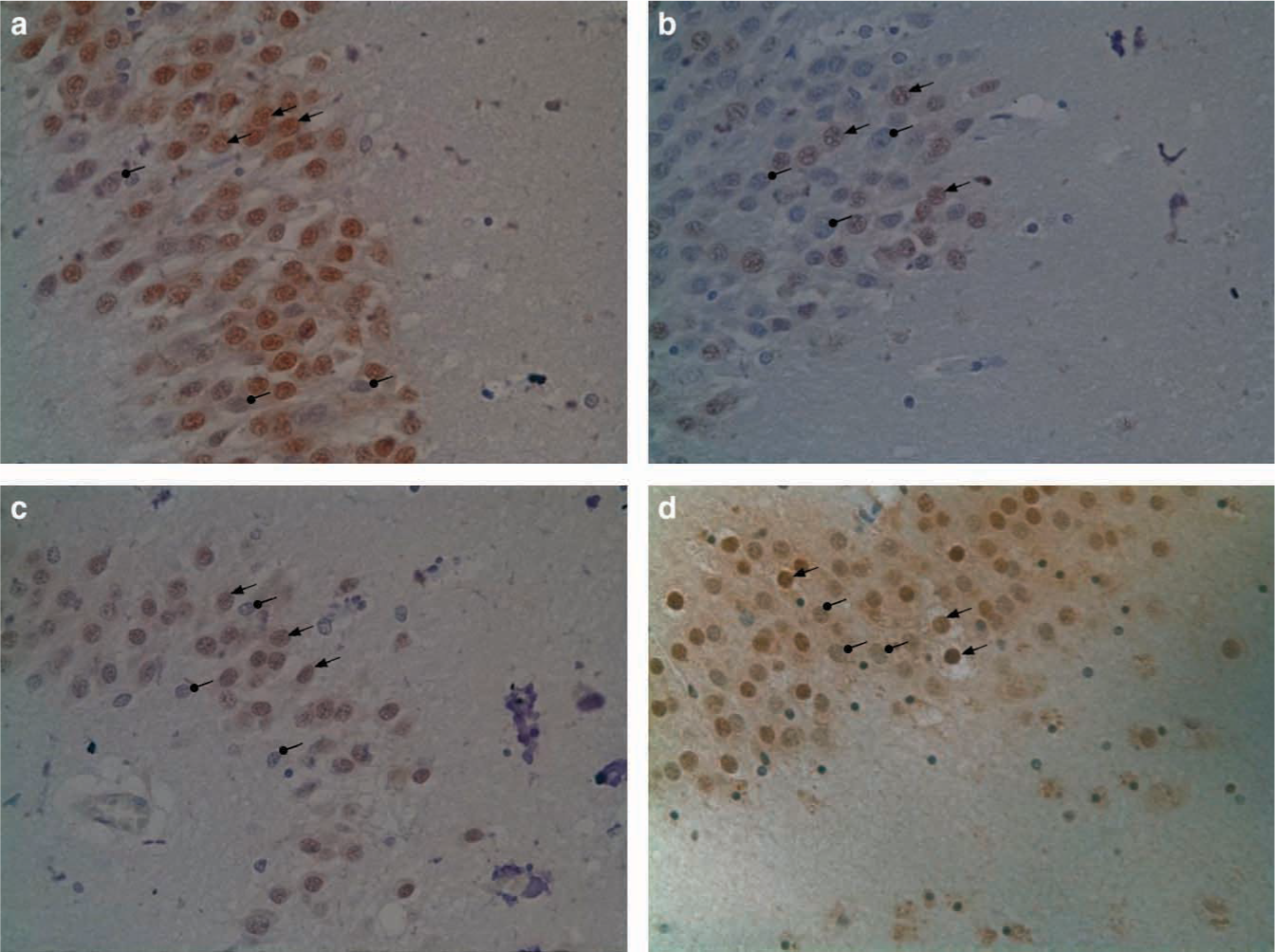
Three of the 12 patients (subjects 2,5,12) exhibited an absence of TDP-43 staining in a proportion of neuronal nuclei (16%, 26%, 70%; Table 1; Figure 2a–c). No cytoplasmic TDP-43-positive inclusions were identified in these three patients nor any other patients. No patients exhibited intraneuronal ubiquitin-positive inclusions. In order to exclude any effects of artefact, a second set of hippocampal dentate gyrus sections was stained for TDP-43 from each of subjects 2, 5 and 12. These were rated blindly and were found to be abnormal, showing the same patterns of TDP-43 distribution as the first set of sections.
Clinicipathological correlations
The histories of the three patients with abnormal TDP-43 staining are summarized in the following case reports. (Clinical information for the remaining nine patients is available from the author).
Subject 2
This subject first came to psychiatric attention aged 57 because of paranoid concerns regarding his neighbours. Friends described a severe deterioration in his level of functioning over several years including self-neglect, lowered mood, hoarding of junk and living with possums and rats in his house. The treating psychiatrist described the house as the worst he had seen during his work as a community psychiatrist. He came from a wealthy, upper class background in Europe and had migrated to Australia aged 55 for reasons that were unclear. His adult son was known to the same mental health service and was being treated for schizophrenia. On assessment the subject was a very articulate man who engaged well at interview. He had auditory hallucinations, paranoid delusions and exhibited formal thought disorder. Due to his late onset and presentation, magnetic resonance imaging and neuropsychological assessment were organized but he refused. He was transferred to a hostel but was non-compliant with psychiatric treatment. He died of myocardial infarct aged 59 years, 30 months after his original presentation. At autopsy the brain weight was 1496 g. Macroscopic examination was normal. Microscopic examination indicated lipohyalinosis of cerebral blood vessels. The post-mortem DIBS diagnosis was of schizophrenia.
Subject 5
This subject had her first psychiatric hospitalization at the age of 50. She believed she was being monitored by police and followed by terrorists. The subject's daughter described periods of depression, isolation and withdrawal when her mother was younger but no psychotic paranoid symptoms. She described auditory hallucinations, was suspicious and thought she was the cause of disasters in the world. A diagnosis of a paranoid delusional disorder was made and she was treated with antipsychotic medications. The diagnosis was subsequently changed to schizophrenia and she was followed by local mental health services. At the age of 54 she began to compulsively shop, buying thousands of items (5 m3) for which she had no use including baby clothes, toys and kitchen utensils. At one stage she told her family she was coming into money and went shopping for an expensive imported car. In the year she died her son, aged 26, was admitted to a psychiatric unit for the first time with a diagnosis of schizophrenia. She died of pneumonia aged 58. At autopsy she was found to have metastatic breast cancer with lung metastases. Brain weight was 1320 g and there were no significant findings on macroscopic or microscopic examination at autopsy. The post-mortem DIBS diagnosis was of schizophrenia.
Subject 12
This subject was first seen by a psychiatrist at age 52 with pressured speech, grandiosity, insomnia and anergia. A previous history of depression was noted at this time and a diagnosis of bipolar disorder was made. He was treated with lithium. At age 57 the subject had a manic psychotic episode during which he gambled heavily and lost his house. The subject was one of six children. One sibling had been treated with electroconvulsive therapy for depression and a second sibling had a diagnosis of schizophrenia. His eldest child had been diagnosed with bipolar disorder from age 17. For 3 months prior to his death he was treated for myasthenia gravis, although details of this diagnosis were not available. He collapsed and died of cardiovascular causes at age 59. At autopsy brain weight was 1542 g and there were no significant findings on macroscopic or microscopic examination. The post-mortem DIBS diagnosis was of bipolar I.
Discussion
The current study is the first to have examined TDP-43 and ubiquitin immunoreactivity in hippocampal dentate gyrus of patients with schizophrenia and bipolar disorder. The major finding of the study was the identification of three patients, diagnosed with a psychotic illness but not dementia during life, who exhibited a relative loss of neuronal nuclear TDP-43 staining. Clinicopathological correlation indicated that these patients had all presented after the age of 50 and all had an adult child with the same psychiatric diagnosis as themselves. The clinical histories of two of the patients (subjects 2 and 5) included behavioural and personality changes including compulsive behaviours, hoarding, poor personal care and disinhibition. Although such symptoms can be seen in FTD, no patient was suspected of having FTD in life.
The TDP-43 findings in the present three patients are not typical of a classic FTD-u pattern in which absent nuclear staining is associated with cytoplasmic inclusions, that is, the present patients had absent nuclear staining but no inclusions. This pattern of neuronal staining has not been previously described but we are confident that the observations are real, rather than artefactual, for several reasons. First, no control subject exhibited this distribution of TDP-43. Second, the findings were identical for the two sets of hippocampal TDP-43 staining for subjects 2, 5 and 12. Third, the TDP-43 staining of the abnormal neurones is very similar to that observed in an FTD-u subject with a known progranulin mutation (Figure 2d) [7]. Finally, a similar pattern of neuronal TDP-43 staining can be seen in the figures of other publications (e.g. Figure 2e [4]; Figure 1b [5]). These authors have not specifically commented on this pattern of staining neurones, perhaps because the focus has been on neurones with TDP-43 cytoplasmic inclusions.
Control subject showing normally staining neuronal nuclei (brown). The intensity of staining varies within normal nuclei (arrows 1–4 showing nuclei staining in descending order). Subjects (a) 2, (b) 5, (c) 12 and (d) patient with progranulin mutation showing neuronal nuclei with (arrows) normal and (•) absent neuronal nuclear staining.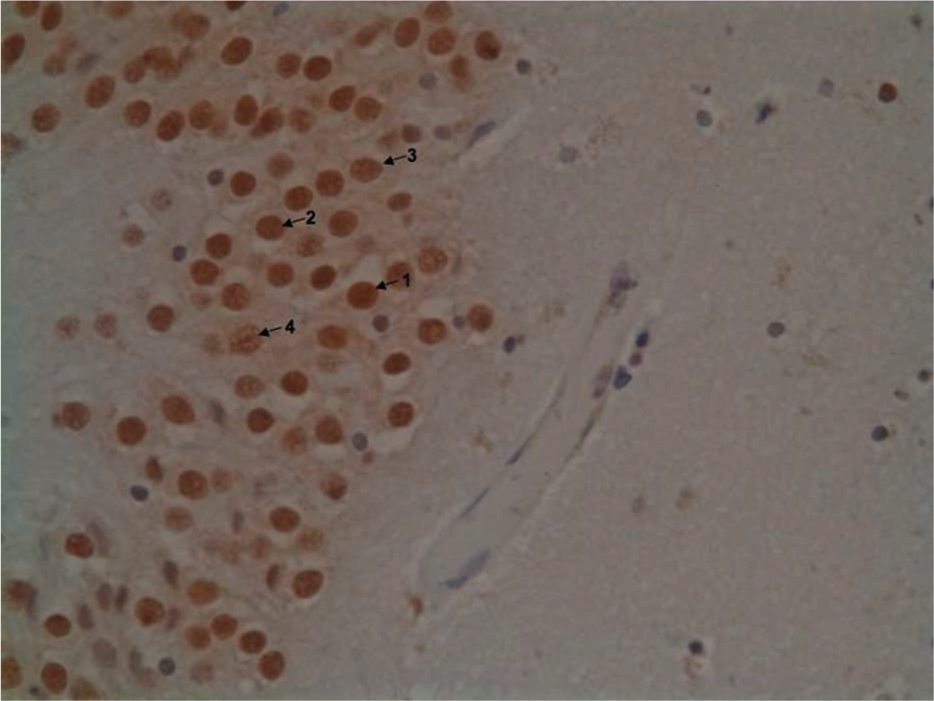
TDP-43 is an mRNA binding protein coded by the TDP-43 gene at 1p36.22 [15]. The known functions of TDP-43 include suppression of gene transcription [15], mRNA transport and translation [15], and the modulation of neuronal plasticity through dendritic spine growth in response to neuronal stimulation [16]. Reduced expression of nuclear TDP-43 may thus be associated with changes in gene expression or neuronal plasticity, but whether this is a normal physiological response to a pathological process or is in itself the pathological change remains unclear.
Unlike the large body of literature regarding the neuropathology of schizophrenia [17], the literature regarding the neuropathology of late-onset psychosis is scant. One relevant study by Casanova et al. identified hippocampal tau-positive glial fibrillary tangles in 20/34 late-onset (>40 years of age) schizophrenia patients and 11/30 early onset (<40 years of age) schizophrenia patients [18]. The authors termed their finding a ‘limbic tauopathy’, which was distinguishable from Alzheimer's disease. Similar glial tangles are seen in tauopathies such as FTD and progressive supranuclear palsy [19]. Taken together with the present study and our previous study [7], these findings suggest that pathology associated with tau or TDP-43 is associated with late-onset psychotic disorders.
The current study had several limitations. First, we examined a small number of patients and further studies are required to confirm our findings. Second, we cannot exclude the possibility that the observations are related to medication effects but if so, we might have expected to see similar changes in all patients who had been treated with psychotropic medication. Third, the finding that two patients with schizophrenia and one with bipolar disorder had abnormal TDP-43 raises the question of the specificity of the results. The subject with a diagnosis of bipolar disorder was reported to have a sibling with schizophrenia, suggesting diagnostic overlap within this pedigree. The existence of diagnostic heterogeneity within a pedigree is consistent with a recent Swedish population-based study of 9 million individuals that found that the schizophrenia and bipolar disorder ‘partly share a common genetic cause’ [20]. If confirmed, the present findings may reflect one such common genetic cause. Finally, it is possible that the three cases were clinically misdiagnosed FTD patients and that the present observations reflect a shared clinical phenotype between FTD and psychotic illnesses rather than shared neurobiology. In this context, follow up of the psychiatrically unwell children of these three patients would be potentially informative. The issue of misdiagnosis may be a particularly important clinical issue given that late-onset psychosis may constitute nearly 30% of all psychosis presentations [21, 22].
In conclusion, the current study has identified three patients who presented with schizophrenia or bipolar disorder after the age of 50, all of whom had adult children with the same psychiatric diagnosis as themselves. Both the clinical presentations and TDP-43 findings overlapped with, but were not entirely typical of those found in FTD. Further investigation of the present findings may potentially shed new light on our understanding of the pathogenetic mechanisms in psychotic illness, help to dissect out the heterogeneity of psychosis and potentially provide clinicians with the ability to identify patients who have FTD underlying a late-onset psychosis.
Footnotes
Acknowledgements
Tissues were received from the National Neural Tissue Facility, supported by the University of Melbourne, Mental Health Research Institute, Alfred Hospital, Victorian Forensic Institute of Medicine and funded by Neurosciences Australia and the National Health and Medical Research Council. The authors would like to acknowledge the input of Ms Fairlie Hinton, Ms Laura Leone, Mr Geoff Pavey, Ms Tiffany Cowie, Ms Qiao-Xin Li and Melbourne Health. We would like to thank those subjects and their families who donated brain tissue for research.
