Abstract
Impairments in both visuospatial working memory (VSWM) [1] and oculomotor pursuit of visual moving targets are considered trait markers of schizophrenia [2], but it is not clear whether a common neuropathological substrate underlies the two deficits.
The ability to coordinate the positioning and movement of the eyes to successfully maintain visual focus on a moving target requires both retinal and extraretinal signals [3]. Retinal signals are sensory signals produced by the motion of an attended target on the retina. During the maintenance of the pursuit, however, retinal information is limited or, in extreme conditions (i.e. the maintaining of pursuit during blinks or target occlusion), not available. Therefore, the system uses extraretinal signals processed in higher visual areas and motor cortex to extrapolate the predicted motion trajectory of the target in order to maintain pursuit.
It has been shown recently that an increased use of extraretinal signals during visual pursuit is associated with selective activation of a number of cortical areas. Three regions of interest here are the dorsolateral prefrontal cortex, parahippocampal gyrus and superior temporal gyrus [4]. These brain structures are known to be involved in memory, VSWM and attention and suggest a possible mechanism by which VSWM may help to optimize the reconstitution and the prediction of the target movements during the pursuit [5, 6]. This link between oculomotor mechanisms involved in pursuit processes and VSWM is important because it has been suggested that dysfunctions in the pursuit mechanisms in schizophrenia is related to VSWM impairment 7–10. A more recent study, however, by Hutton et al. showed that deficits in eye movements generated in response to smooth target displacements are not correlated to executive impairments, including working memory, involving visuospatial information in patients with schizophrenia [11].
In our view the contradictory nature of these previous results regarding the relationship between pursuit eye movement processes and VSWM are mainly due to the different tasks used to assess oculomotor function and VSWM. Furthermore, the relationship between oculomotor functions involved in visual pursuit and VSWM in patients with schizophrenia has been principally investigated using correlation analysis between independent assessments. This removes the possibility of directly assessing the association between oculomotor and VSWM processes, necessitating further investigations. Moreover, the impact of the general deficit in oculomotor pursuit mechanisms in patients with schizophrenia on true VSWM performance remains to be examined. Indeed, even if potentially independent to oculomotor pursuit mechanisms, VSWM abilities in schizophrenia might be additionally affected in dynamic stimuli condition by important inaccuracy in tracking dynamic targets.
To investigate whether a common neuropathological mechanisms underlies the two deficits, it is critical to first determine the nature of the relationship between VSWM and oculomotor pursuit abnormalities in schizophrenia. The present study of patients with schizophrenia was designed to investigate directly the interaction between oculomotor functions involved in dynamic target pursuit and VSWM, using a dynamic VSWM task [12]. Subjects were required to observe and remember the zigzagging trajectory of a disc moved across the screen (Figure 1a). In this task, dynamic stimulus encoding relies on two processes: (i) a sequential encoding of the complex visual target displacements in 2-D space; and (ii) a subsequent online integration of sequential visuospatial information to create a coherent internal representation of the whole stimulus. Unlike previous studies, this dynamic VSWM task used here, allows us to assess the general pursuit abilities in a realistic target tracking condition. Furthermore, the encoding of the disc trajectory is complex, and highly engages VSWM resources.
Ball flight test. (a) Illustration of the dynamic visuospatial working memory task design showing the time sequence for each trial. Note that the path taken by the stimulus is invisible during the encoding. (b) The three regions of interest (Stimulus_ROIs; limits not visible during the presentation) and the six Discrimination_ROIs (1–6 in the discrimination slide, each rectangle represents an ROI). Also included are the three specific ROIs defined to investigate the distribution of fixations on the correct path.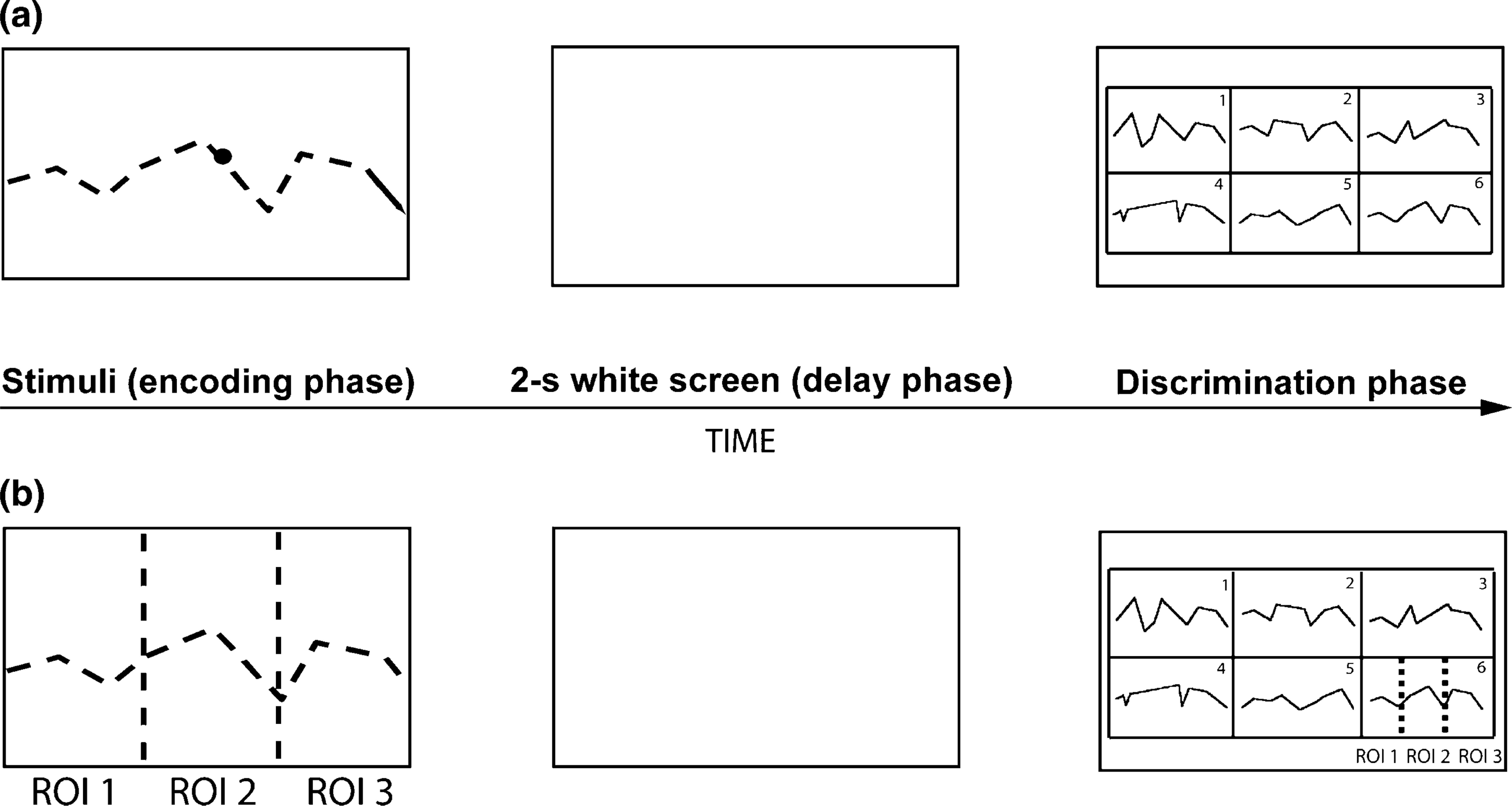
In a previous study with our dynamic VSWM task, we found that patients with schizophrenia showed control level recognition abilities, but significant qualitative differences in terms of encoding strategy [12]. Indeed, patients’ errors suggested that they relied on the last part of the trajectory, implying that they were unable to encode the whole sequence of information within VSWM. In that earlier study, however, it was not possible to determine whether the dysfunction in VSWM processes was related to selective encoding of the last part of the trajectory or to a general inaccuracy in tracking the dynamic target. Here, we extend our investigation using eye-tracking technology with an independent sample of participants with schizophrenia. The main objective was to examine how encoding and recognition strategies, as well as visual pursuit accuracy, related to dynamic VSWM performance.
Methods
Subjects
Subject details
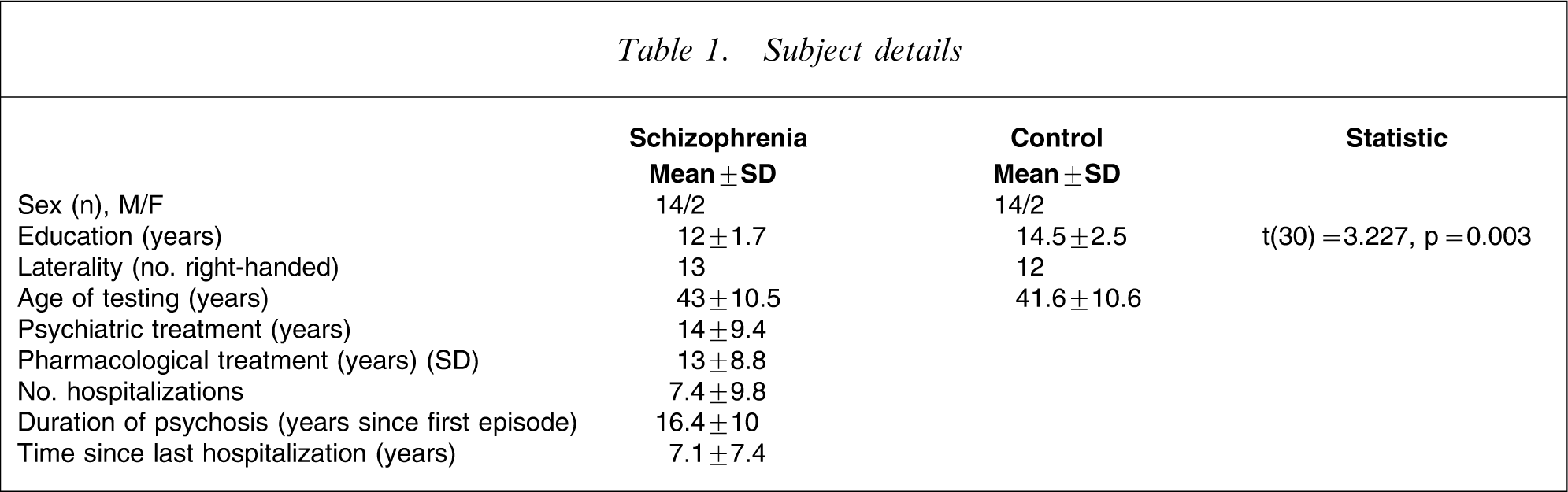
All subjects in the control group reported no history of substance or alcohol abuse and were free from psychiatric illness or symptoms, as investigated by a structured diagnostic psychiatric interview for DSM-IV and ICD-10 [13]. Control subjects and patients had normal or corrected to normal vision. Individuals with a history of neurological or medical illness likely to affect the nervous system, and traumatic brain injury were excluded.
Participants were provided with a complete description of the proposed study and given oral and written information. The University of Lausanne (School of Medicine) ethics commission approved the protocol and consent procedures. Subjects participating in this study were voluntary and were compensated with 25 Swiss francs after the test.
All the patients were treated with antipsychotic drugs at the time of testing. Fourteen patients received atypical antipsychotic drugs (chlorpromazine equivalents 325.71±204.50 mg day−1) [14, 15] and one received typical (chlorpromazine equivalent: 500 mg day−1) medication. Data were not available for one.
Procedure
Participants performed a task designed to investigate working memory processes involved in the encoding of dynamic visuospatial stimuli (Figure 1a; for a detailed description see [12]). Subjects sat on a chair 3 m from the screen and were instructed to keep their back on the chair and their hands on their legs throughout the task. They were instructed to observe and memorize the zigzagging trajectory (Figure 1a) of a black disc (diameter = 0.76) travelling from left to right across a screen (constant speed in the direction of the line segments, 6.35 cm s−1) along eight successive rectilinear segments of variable length. The start of each of the 15 successive trials was controlled by the subjects. A delay lasting 2 s (white screen) was imposed at the end of the stimulus presentation, followed by the presentation of the recognition slide displaying six static linear paths outlining the motion trajectory covered by the disc (the correct and five incorrect paths) in a 3 × 2 grid. Upon the presentation of the discrimination slide, participants were asked to verbalize the code number (from 1 to 6) corresponding to the target stimulus as they identified it. This discrimination slide was available as long as required.
The five incorrect paths were designed to partially match each target stimulus. The specific distortions leading to the five incorrect paths allowed these to be distributed in two different categories: (i) fragmentary, paths conserving two segments (first two: start paths; last two: end paths) or four segments (first and last two: start–end paths) identical to those of the target trajectory; and (ii) global, paths with either the same nine coordinates on the x axis (paths named rhythm) or on the y axis (paths named melody) as in the target stimulus. In this category, the incorrect paths (melody and rhythm) and the target trajectory had a common global structure but no identical segments.
A practice session was administered before the test. At the end of the test session, participants were asked to describe any strategy(ies) used to encode the stimuli.
Oculomotor data acquisition and analysis
Eye movement data were collected using an infrared technique (Applied Science Laboratory, Bedford, MA, USA; H6 model, pupil-corneal reflection tracking, sampling rate 120 Hz, accuracy 0.5° and resolution of 0.1°). Visual stimuli were presented with a projector (resolution of 1280 × 1024, with refresh rate of 70 Hz) on a white screen (horizontal dimension 147 cm (27.5° visual angle), vertical dimension 110 cm (20.8°)). A nine-point calibration procedure was performed after the practice session, and also at the end of the test as a control for measure stability. The head was not fixed during the tasks, but its position in space was recorded by a magnetic sensor integrated into the helmet (sampling rate of 103 Hz). Eye movement data were automatically corrected for head movements and vice versa. Post-experimental eye movement analyses were performed with ILAB [16] and they were executed on two specific tests phases (Figure 1b): (i) encoding (during the presentation of the trajectory); and (ii) discrimination phase (during the discrimination of the correct path between the six possibilities). Qualitative online video analysis of the target pursuit and the stimulus discrimination phase were used to ensure that all the subjects were able to perform, actively, the task.
Eye movement analysis during the encoding phase
We first assessed tracking accuracy during the dynamic presentation of the moving target disc by calculating an error vector (relative distance between disc segment and fovea) every 8.33 ms (120 Hz). Median of error vector magnitude (scalar value, in cm) was calculated for each stimulus and subjects, and an error vector magnitude median for each subject was then calculated and used for comparisons. Thus, median error vector analysis can be interpreted as a global measure of the distance to the precise target pursuit. In addition, we assessed whether there were regional biases in eye position during the encoding phase. As shown in Figure 1(b), three regions of interest (ROIs), that is, Stimulus_ROIs, were defined to investigate if subjects completely follow the disc displacements. Visual pursuit time was assessed in the three specific ROIs. The three first trajectory segments comprised the Stimulus_ROI 1, the two central segments comprised the Stimulus_ROI 2 and the three last segments defined the Stimulus_ROI 3.
Eye movement analysis during the discrimination phase
Visual scanning variables included the number of fixations (FXn) and the scanning time (ST) in specific ROI (dimension of each cell horizontally 9.2°; vertically 5.60°). Fixations were defined as a set of consecutive gaze coordinates confined within a circular area of 1–2° diameter for a period of at least 150 ms [17, 18]. In the present study a fixation was defined as a sequence of consecutive eye-positions with x and y coordinates falling within a diameter of 1.5° of visual angle for at least 150 ms. Blinks were identified from a loss of corneal reflection and were excluded from subsequent data analysis, as were gazes outside the screen. We defined six ROIs (Discrimination_ROI: 1–6; Figure 1b) corresponding to the six grid cells containing the correct and the five incorrect paths. An analysis of ST and FXn distribution was performed on the six specific ROIs. The distribution of individual fixations on the correct paths was also recorded. The distribution of FXn on the correct paths (Figure 1b) was estimated by the same criteria used in the encoding task phase (i.e. first three segments for the ROI 1; two central segments for the ROI 2 and the three last segments for the ROI 3).
Statistical analyses
Working memory performance
t-Test analyses were used to compare groups’ task performance. A mixed two-way ANOVA was used to analyse between-group differences (patients vs controls) on incorrect paths choices (repeated factor). Data sphericity was tested using Mauchly's approach. When sphericity was not respected, we used a Huynh-Feldt correction to rectify ANOVAs results. Cohen's d and partial η2 were used to estimate effect size.
Eye and head movements
Encoding phase
t-Tests were used to assess the median of error vector magnitude differences within and between groups. This parametric approach was used because data normality was confirmed. Cohen's d was used to estimate effect size and Pearson's correlations were used to evaluate the relationship between the median error vector magnitude and the number of correct trials.
Mixed between–within-subjects three-way ANOVAs on visual ST during stimuli encoding were used to investigate a potential interaction between group, trials and ST distribution in the three Stimulus_ROIs (repeated factor). Partial η2 was used to estimate effect size.
Horizontal head movements during the presentation phase were evaluated as follows: horizontal head movements and time of occurrence were recorded and then plotted on a 2-D scatter plot (head position on the y axis, time on the x axis; the axis intersection indicates the centre of the screen at time zero). The regression linear slope was then estimated, positive values indicating that the subject's head movements were coherent with the left to right movement of the target, negative values indicating non-coherent movements. For each subject, the average of the 15-stimuli slope values was calculated and used for group comparisons using the Mann–Whitney U-test. Additionally, Spearman's correlation was used to study the relation between the global horizontal head movements’ coherence (average of the 15 regression slope values for each subject) and the total number of correct choices. Spearman's correlation was also computed for each individual trial to evaluate the correlation between vertical head movements and target displacements. A Mann–Whitney U-test on Spearman's correlation averages was finally performed to evaluate a possible difference in vertical head–target-related movements between the two groups.
Discrimination phase
Mixed between–within-subjects two-way ANOVAs on FXn and ST were used (i) to assess the between-group visual exploratory pattern in the Discrimination_ROIs and (ii) to contrast the exploratory pattern (FXn) in specific ROIs of the correct paths in patients and controls. Post-hoc t-tests were performed to investigate global differences evidenced by ANOVAs.
Results
Working memory
Between-groups comparison of task performances yielded significant differences between patients with schizophrenia and healthy control participants. Subjects with schizophrenia had significantly fewer correct responses than controls (t(30) = − 3.435, d = 1.21, p = 0.002; Figure 2).
Variability (confidence intervals, 95%) and the mean of correct paths choices obtained by () controls and (○) patients with schizophrenia. Patients obtained a lower score than controls (t-test: ∗∗∗p = 0.002; Cohen's d = 1.21).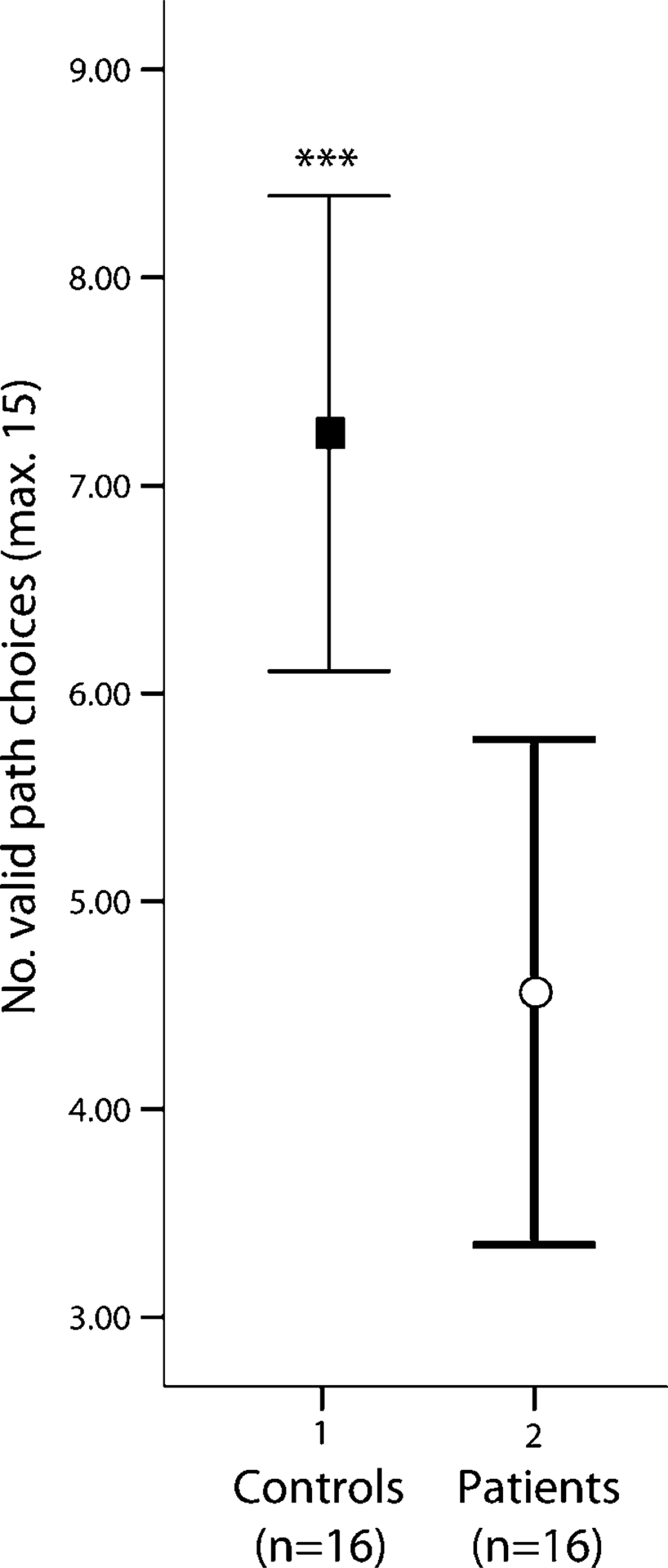
A mixed two-way ANOVA of error categories (2 groups × 5 categories as repeated factor) showed a significant group effect (i.e. more wrong choices overall in the patients F(1,30) = 11.797, partial η2=0.282, p = 0.002) but no significant error category effect (F(4,120) = 0.852, partial η2=0.028, p = 0.495) or group × category interaction (F(4,120) = 0.555, partial η2=0.018, p = 0.696).
Eye movements
Encoding phase
The comparison between the median error vector magnitude showed that controls and patients had similar disc pursuit in both correct and incorrect recognition trials (correct: t(29) = 0.107, d = 0.038, p = 0.915, incorrect: t(30) = − 1.219, d = 0.43, p = 0.232). In addition, pursuit precision was not significantly related to choice accuracy (dependent t-tests: controls t(15) = 0.785, d = 0.09, p = 0.445; patients t(15) = − 1.168, d = 0.42, p = 0.262). Effect size analyses, however, suggested globally diminished pursuit accuracy in incorrect trials versus correct trials in patients (correct 1.7±0.5° and incorrect 2.5±2.6°; controls: correct 1.8±0.8°, incorrect 1.7±0.6°). In spite of this, there were no significant correlations between the number of correct choices and the median of the 15 error vector magnitudes (patients: r = 0.151, p = 0.590; controls: r = 0.074, p = 0.785). Using the ROI analysis, no significant interaction was found between group and choice (correct vs incorrect) for ST in the three stimulus_ROIs in a mixed three-way ANOVA (F(1.619,774.012) = 0.267, partial η2=0.003, p = 0.654). This means that patients’ distribution of ST on the screen was not different from control level during the observation of disc trajectory.
Because of a technical problem in the generation of the head movement output file, head movements were analysed only for 11 patients and 10 controls. A significant difference between groups in horizontal head movements (i.e. values of the regression linear slope) was shown in 14 of the 15 stimuli (Mann–Whitney U-test; p < 0.05 for each). Specifically, control subjects performed horizontal head movements that were globally related to the target direction (left to right, i.e. positive values) during stimulus presentation. In contrast, patients with schizophrenia had globally negative values (in seven patients) or near zero values (in four patients). This means that patients’ horizontal head movements during target pursuit were frequently oriented in the reverse target direction, but there were no significant correlations between the amplitude of horizontal head movements and the number of correct choices (patients: ρ = 0.142, p = 0.696; controls: ρ = 0.060, p = 0.861). Finally, a Mann–Whitney U-test showed that global (average of 15 stimuli correlations) head–target correlations on the vertical axis were not different in the two groups (Z= − 0.775, p = 0.438).
Discrimination phase
Because the duration of grid presentation was not equivalent for all participants, the analyses were performed on data relative to individual total fixations and ST (%) in the six Discrimination_ROIs. Scanning of the six paths was analysed separately for the trials with a correct or an incorrect path choice. We also investigated if the distribution of FXn on the correct paths during correct or incorrect trials was different in the two groups.
Correct trials
A mixed two-way ANOVA (group×six discrimination ROIs) showed a significant main effects for ROIs (FXn: F(4.082,757.629) = 123.346, partial η2=0.011, p < 0.001; ST: F(4.403,840.937) = 186.171, partial η2=0.494, p < 0.001), suggesting a selective difference in the path inspection patterns. Post-hoc paired t-test analyses showed that subjects (controls and patients) spent more time viewing the correct path than the five incorrect paths (paired t-tests on FXn and ST: p < 0.001 for all the five correct vs incorrect Discrimination_ROIs comparisons in both groups; Figure 3a). No significant group main effects and interactions were found for FXn and ST (p > 0.05).
Mean and confidence intervals (95%) of the relative number of fixations performed by () controls and (○) patients throughout the discrimination phase. (a) Correct trials; (b) same representation for incorrect trials. n = total of correct/incorrect recognitions in each group. ∗∗p = 0.13, d = 0.38.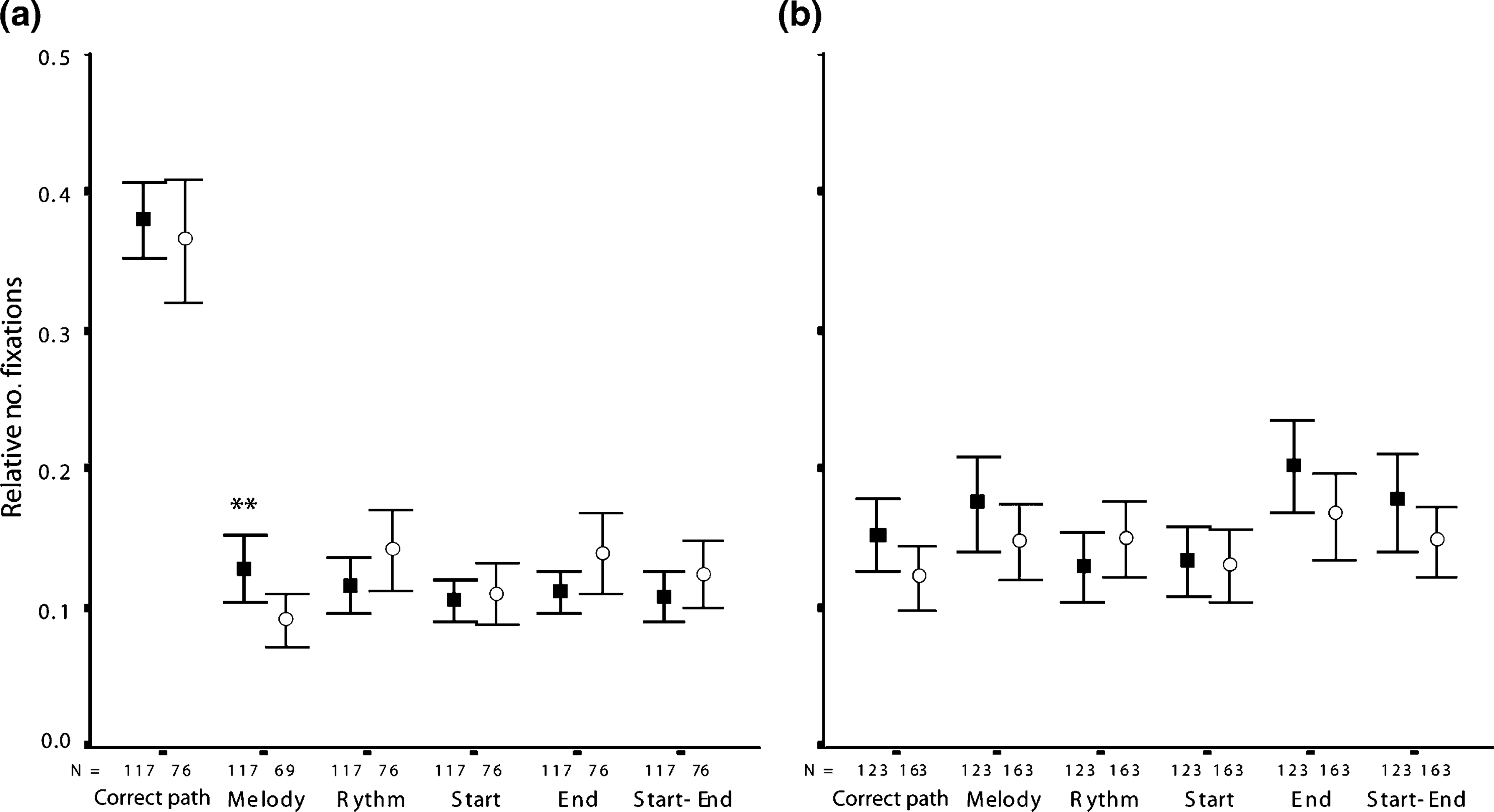
In spite of a similar exploration of the correct paths, the two groups differed in the scanning of the five incorrect paths (group×ROI interaction for FXn F(3.833,732.104) = 3.028, partial η2=0.016, p = 0.019). Specific comparisons illustrated that controls scan (FXn) more frequently the melody incorrect paths than did the patients (t(191) = 2.501, d = 0.38, p = 0.013).
Incorrect trials
A group×ROI (six discrimination ROIs) mixed two-way ANOVA showed significant ROI effects (FXn: F(4.889,1388.575) = 3.328, partial η2=0.012, p = 0.006; ST: F(4.918,1401.700) = 2.301, partial η2=0.008, p = 0.044), suggesting a difference in the pattern of paths inspection (Figure 3b). There were inconsistent group effects for each of the different visual scanning variables (FXn: F(1,284) = 10.234, partial η2=0.035, p = 0.002; ST: (F(1,285) = 0.760, partial η2=0.003, p = 0.384) and no significant interactions (FXn: F(4.889,1388.575) = 0.994, partial η2=0.003, p = 0.419; ST: (F(4.918,1401.70) = 0.473, partial η2=0.002, p = 0.794). Post-hoc paired t-test analyses suggested that subjects tended to spend more time viewing incorrect paths that preserved the last part of the trajectories (end paths). End paths were scanned significantly more often than correct (FXn: t(285) = 2.919, d = 0.26, p = 0.004; ST: t(286) = 2.975, d = 0.26, p = 0.003) and start (FXn: t(285) = 3.075, d = 0.27, p = 0.002; ST: t(286) = 2.493, d = 0.23, p = 0.013) paths.
Specific visual scan patterns (FXn) of the valid paths in correct and incorrect trials
A group × ROI (three specific correct path ROIs) two-way mixed ANOVA showed a significant interaction only when patients and controls selected the valid paths (F(2,374) = 3.264, partial η2=0.017, p = 0.039). As shown in Figure 4, patients addressed a lower number of fixations in the first three path segments than did controls in trials in which they recalled the path correctly (t(187) = 2.781, d = 0.526, p = 0.006).
Mean and confidence intervals (95%) of the distribution of fixations on the correctly chosen paths during the discrimination phase. (○) Patients and () controls had different exploratory patterns. In correct trials, controls scanned the first region of interest (ROI 1; Figure 1b) of the correct paths more, whereas patients concentrated on the second half of the stimulus (ROI 2 and 3). ∗∗p = 0.006, d = 0.52.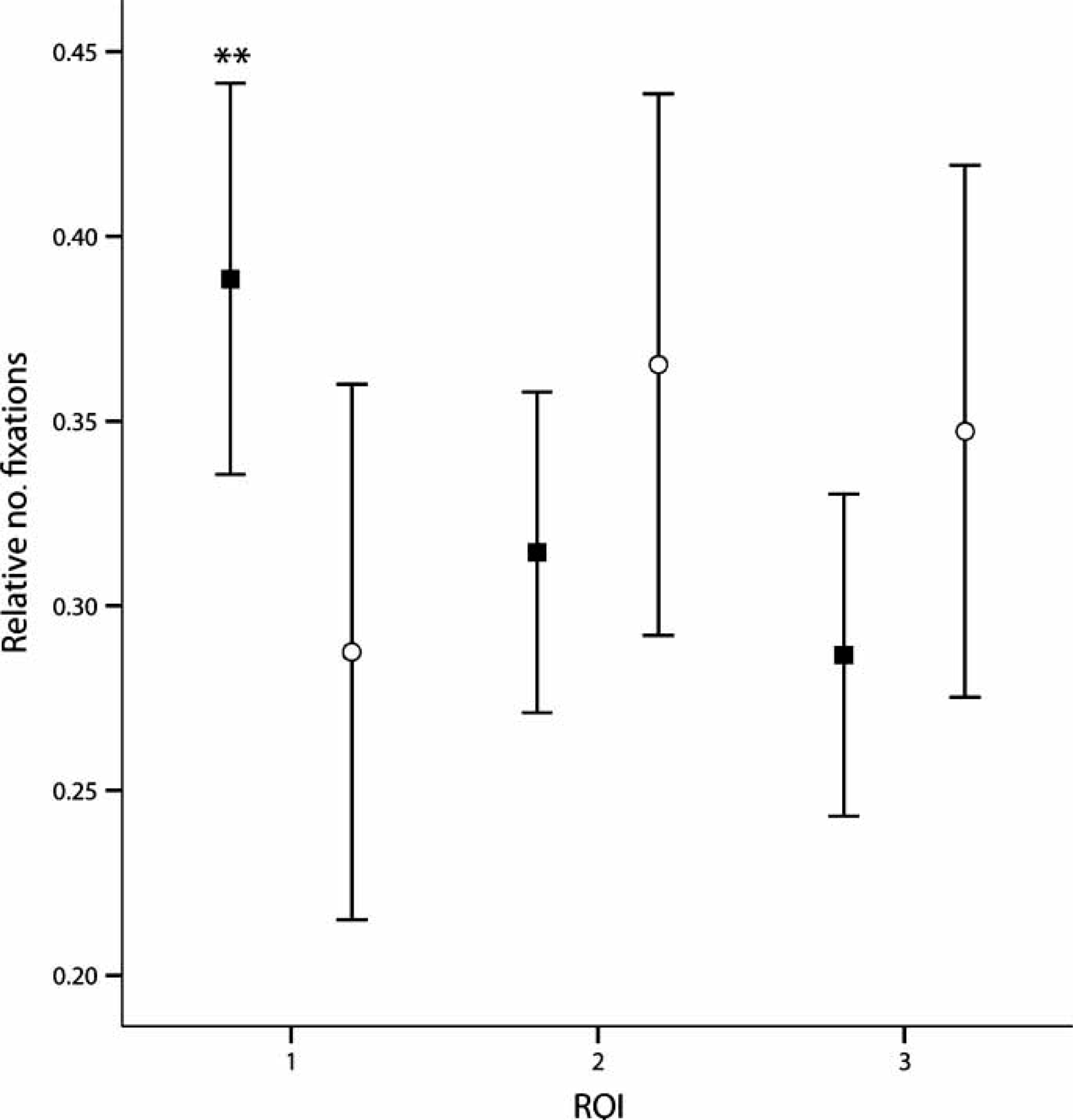
Discussion
The present study addressed the direct relationship between encoding and recognition strategies during a dynamic VSWM task. Specifically, we investigated whether differences in the accuracy of visual pursuit processes could account for any deficits in VSWM performance in patients with schizophrenia compared to control subjects. We used a working memory task presenting complex dynamic visuospatial stimuli [12] and found that patients chose the correct path less frequently than did controls. Eye tracking measures were analysed during the encoding of the disc trajectory and also during the subsequent path discrimination phase in order to provide deeper insights into the nature of the observed deficits.
Together with the increased frequency of wrong choices, the spatial biases in scanning expressed by patients during the path discrimination phase suggest that they have impaired VSWM and are unable to recall the accurate global representation of the disc trajectories. Two main findings provide evidence supporting this hypothesis. First, in correct trials, analysis of eye movement during the presentation of the discrimination grid showed that success was accompanied by increased scanning of the correct path relative to the five incorrect ones in both patient and control groups. In contrast, a homogeneous exploration of the six paths preceded incorrect path selection. In addition, even in trials when patients were able to discriminate the correct path, patients’ gaze behaviour seemed to be related to a less accurate and less global internal representation of the target displacements. Indeed, controls displayed an increased number of fixations on the melody incorrect paths during correct trials. Because of their specific distortion, melody paths are the most globally similar to the correct paths [12]. It could be argued that controls displayed greater fixations on the melody paths because they were more likely to use a verbal strategy to memorize the target trajectory (i.e. up, down, up, up, down, up, down, down; Figure 1). This appears unlikely, however, because both patients and control subjects rarely mentioned the use of verbalization when they were asked to describe any strategies used. Second, the analysis of fixations distribution performed on correct paths in correct trials showed that patients made relatively less fixations on the first three segments of the path than controls. Thus, patient inability to recall the first third of the target movement may have caused them to rely more heavily on their memory of later portions of the disc trajectory in trials when they chose the correct path. In our view, this loss of information from the earliest encoded segments is due to dysfunctions in sequential information organization and may be largely responsible for the observed deficit in VSWM capacity. This assumption is consistent with the general preference for the incorrect paths conserving the last part of the trajectories during incorrect trials and with previous findings obtained using the same task in an independent group of patients with schizophrenia [12].
Specific eye movement measures were conducted to control whether group differences in scanning performance during the discrimination phase were due to differences in encoding strategy and/or to a global deficit in pursuit accuracy during the encoding of the disc displacements. The present finding of similar pursuit time in the three stimulus zones by the two groups illustrates that patient discrimination inefficacy cannot be explained by differences in the encoding strategies (i.e. due to focussing on the end region of the trajectory). Overall error vector magnitude analyses did not indicate a significant global inaccuracy in following the disc displacements in patients. Effect size analysis, however, suggests that patients were less accurate than controls in tracking the disc in the incorrect trials. Due to the high frequency of incorrect path recognition it can be argued that patients were often unable to perform a sufficiently precise pursuit. This assumption is supported by results suggesting that a slightly higher tracking accuracy led to correct path recognition. The global precision of eye movement pursuit, however, was clearly not correlated with the number of correct choices. Generally, these findings are consistent with the established idea that patients have abnormalities in pursuit processes [19], but suggest that the important stimuli recognition impairment seen in patients is unlikely to be due to a general deficit in pursuit accuracy.
We also included additional analyses of head movement during the encoding of the dynamic stimuli because head movements play an important role in pursuit processes and share a similar brain network to smooth pursuit eye movements [20]. Head movement analysis showed that patients did not execute control-like horizontal disc-related movements during the disc trajectory presentation. But patients showed inconsistent horizontal head movements even during trials followed by a correct choice, and these movements were not correlated with the number of correct choices in both groups. Hence, the impaired patient performance in the dynamic VSWM task was not simply related to abnormal horizontal head movements during encoding.
The results of impaired path discrimination demonstrated by the schizophrenia group contrast partially with those of our previous study. No evidence of impaired performance in the patient group was found in the dynamic condition, but a merely different selection strategy (i.e. accurate retention of the last three segments [12]). A possible explanation for these results could be the difference in illness duration between the two samples: 5 years in the first study compared to 14 years in the present study. Illness progression could affect the early sensory processes and especially the prefrontal VSWM processes. This assumption is supported by studies reporting progressive grey matter loss over the course of a psychotic illness, especially in prefrontal cortex [21, 22].
Finally, it is necessary to note certain methodological limitations of the present study such as the small number of patients and their diverse medication. Medications were not considered in the statistical analyses as covariates because of the large heterogeneity in substances and doses used. We were also not able to match patients and controls in years of completed education. The performance of controls, however, was not correlated with years of education. The finding that poor pursuit performance and impaired recognition are unrelated may be due to insufficient power, and we cannot discount that there is some relation between them in schizophrenia. Further studies are needed to explore the magnitude of this hypothetical influence.
In summary, the present study provides direct support for the hypothesis that active integration of visuospatial information within VSWM is deficient in established schizophrenia [23, 24]. In contrast, a general impairment of oculomotor mechanisms involved in pursuit process did not appear to be a critical factor for the lower VSWM performance in the disorder.
Footnotes
Acknowledgements
The authors especially thank Professor Pierre Bovet and Dr François Grasset for allowing us to test patients under their care and Dr Pascal Vianin and Jerôme Favrod for his efficient collaboration. The Swiss National Foundation for Scientific Research supported this study (Professor F. Schenk) 3100A0–105765 and (Dr L. Cocchi) PBLAB–119622 and PBLA. Dr Wood is supported by a Clinical Career Development Award from the NHMRC (Australia). Dr P. Connus is supported by the Leenaards Foundation, Switzerland. The Robert Bosch research institute of Lonay (Switzerland) supported this study in providing technological facilities for eye movement recording and analysis.
