Abstract
Clozapine has been shown to be an effective antipsychotic treatment in a large percentage of patients with schizophrenia who are resistant to conventional and other atypical antipsychotic medications [1], [2]. Furthermore, it has been shown to have a beneficial effect in reducing rates of violence [3–5] and rates of suicide [6]. In a forensic psychiatric population, where the majority of offences have been violent in nature with a large proportion being committed in the context of psychosis, clozapine clearly has a part to play in the treatment and rehabilitation of patients [7], [8]. The adverse effects associated with clozapine, some of which are life-threatening, have been well documented [9–21]. It is not readily available in parenteral form, which raises concern regarding ensuring future treatment adherence in the community in patients perceived to be at high risk of relapse and potential violence. Mandatory blood monitoring can also cause annoyance, leading to patient-initiated cessation of clozapine. Patients prescribed clozapine have high rates of discontinuation due to a number of these factors [22].
This study was conceived as a survey to explore the recording in clinical notes of various parameters for patients prescribed clozapine. The purpose of the study was to review the use of clozapine in a forensic psychiatric population. A retrospective file audit was conducted. A group of patients prescribed clozapine were compared to a group of patients prescribed other antipsychotics, matched for sex (male) and diagnosis: a range of variables were recorded, such as demographic details, comorbidity, physical observations, rates of adverse effects, investigations and effect of treatment. In the clozapine sample, reasons for discontinuation and rates of relapse of psychosis were noted. Highest dosages, highest serum levels and rates of other medications being prescribed were recorded. This study gives an overview of use of clozapine in a forensic population, records many parameters relevant to its use and compares data with patients prescribed other antipsychotic medications. It represents an opportunity to study the use of clozapine in a patient group treated in a heavily restricted environment, with relatively long lengths of admission, low levels of illicit drug use and where medication adherence can be ensured.
Methods
The clinical file of patients who had been admitted to the forensic acute admission ward at Long Bay Hospital between 1999 and 2004 were reviewed and those prescribed clozapine were identified. In this period, 608 patients were admitted. The majority of these patients were diagnosed with psychotic illnesses (mainly schizophrenia, schizoaffective disorder, bipolar disorder and drug-induced psychosis). Of these 608 patients, 330 patient clinical files were reviewed. The remaining 278 patient files were not reviewed. This was due to many of the patients having been discharged from the forensic psychiatric hospital to outlying prisons, with their clinical files being transferred with them to rural and remote centres. While these inmates were incarcerated and receiving care clinicians in outlying centres required access to these files. It was therefore not feasible to have these files returned to a central location for review. The pharmaceutical companies who manufacture clozapine maintain monitoring systems for clozapine patients. These systems were not used to identify patients in order to avoid potential interference or bias in this study.
Once identified, the subjects’ clinical files were reviewed and data entered onto a data-recording sheet. Data recorded were as follows: age at first diagnosis of psychosis, age first prescribed clozapine, time between diagnosis and prescription of clozapine, index offence, psychiatric, drug and alcohol, and medical diagnoses, prevalence of hepatitis C, history of i.v. drug use, treatment with methadone, data before and after clozapine initiation for weight, pulse, blood pressure; blood glucose, triglycerides and cholesterol, changes in liver function tests, incidence of neutropenia and seizures, highest dose of clozapine prescribed, length of time prescribed clozapine, concurrent medications prescribed, recording of clozapine plasma levels, number of antipsychotics prescribed prior to clozapine in the forensic setting, clinical effect of clozapine using the Global Improvement item of the Clinical Global Impression scale [23], whether clozapine prescription was continuing or ceased, change of mental state following clozapine cessation and reasons for cessation of clozapine. Similar data were recorded for the non-clozapine group. Once collected, the results were collated onto a spreadsheet and analysed.
The retrospective nature of the study, non-synchronous collection of data and inaccessibility of a number of clinical files were a weakness in the study and may result in bias.
Data analysis
Results are presented as proportions with 95% confidence intervals (95%CI) for each of the main findings. The data relating to medical parameters are presented as means for each parameter.
Results
Results are presented here for both the clozapine and non-clozapine groups. Table 1 provides a summary of the main findings.
Main findings
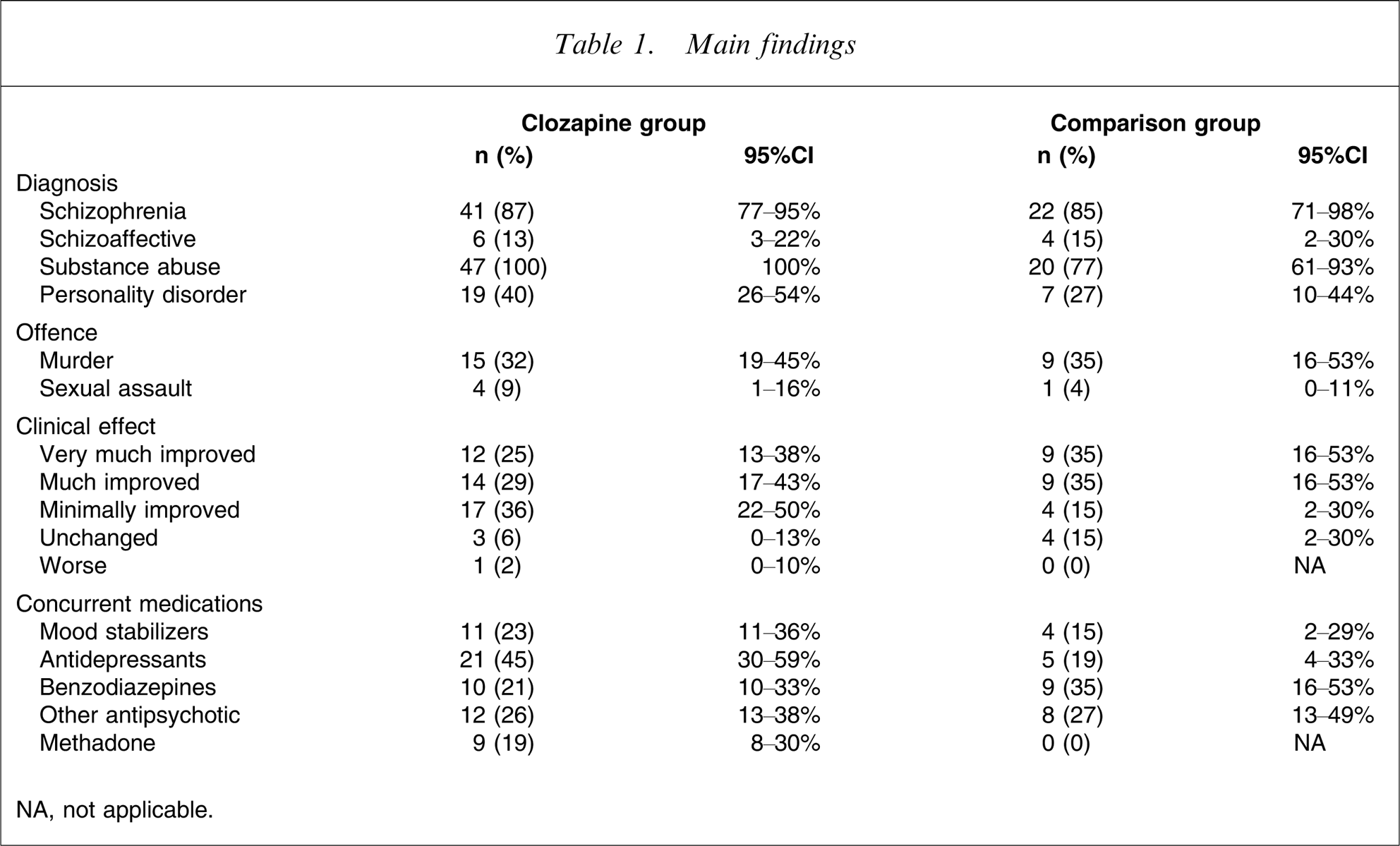
NA, not applicable.
Demographics
Age
Of those prescribed clozapine the mean age at first diagnosis of psychosis was 22.31 years (range 14–37 years); mean age first prescribed clozapine was 30.74 years (range 20–46 years); and mean time between diagnosis of psychosis and prescription of clozapine was 8.23 years (range 1–23 years). In the comparison group the mean age at diagnosis was 32 years (n = 18, range 20–49 years).
Diagnosis
In the clozapine group 41 subjects (87%; 95%CI = 77–95%) had a diagnosis of schizophrenia; and six subjects (13%; 95%CI = 3–22%) were diagnosed with schizoaffective disorder. All of the subjects fulfilled criteria for substance abuse or dependence (47, 100%; 95%CI = 100%); 26 subjects (55%; 95%CI = 41–70%) had a history of i.v. drug use; 19 subjects (40%, 95%CI = 26–54%) were diagnosed with personality disorder and 16 subjects (34%; 95%CI = 20–48%) had comorbid pre-existing major medical problems.
In the comparison group 22 subjects (85%; 95%CI = 71–98%) had a diagnosis of schizophrenia; four subjects (15%; 95%CI = 2–30%) had a diagnosis of schizoaffective disorder; 20 subjects (77%; 95%CI = 61–93%) met the criteria for substance abuse or dependence and seven subjects (27%; 95%CI = 10–45%) had a diagnosed personality disorder. Five subjects (19%; 95%CI = 4–34%) had comorbid major medical problems.
Index offence
The index offence was recorded as the most serious offence leading to incarceration at the time of data collection. In the clozapine group murder was the most common index offence (15 subjects, 32%; 95%CI = 19–45%). Sexual assault was recorded in four cases (9%; 95%CI = 1–16%); break and enter in three cases (6%; 95%CI = 0–13%); abduction of child in one case (2%; 95%CI = 0–6%); assault in seven cases (15%; 95%CI = 39–77%); malicious wounding in eight cases (17%); armed robbery in four cases (9%; 95%CI = 1–16%); breach of Apprehended Violence Order in one case (2%; 95%CI = 0–6%); malicious damage in one case (2%; 95%CI = 0–6%); firearms offences in one case (2%; 95%CI = 0–6%) and grievous bodily harm in one case (2%; 95%CI = 0–6%).
The most common index offence in the comparison group was also murder (n = 9, 35%; 95%CI = 16–35%); sexual assault was recorded in one case (4%; 95%CI = 0–11%); assault, n = 2 (8%; 95%CI = 3–18%); malicious wounding, n = 3 (12%; 95%CI = 0–24%); armed robbery, n = 3 (12%; 95%CI = 0–24%); malicious damage, n = 1 (4%; 95%CI = 0–11%).
Physical parameters
Physical health data were easily accessible for the clozapine group but there were limited data available in relation to physical parameters for the comparison group. Therefore the results reported here are for the clozapine group only.
Weight
The mean weight before clozapine was 86.76 kg (n = 34); 6 months after clozapine, 90.44 kg (n = 27); 12 months after clozapine initiation, 93.7 kg (n = 20); 24 months after clozapine, 100.55 kg (n = 11) and 5 years after clozapine, 119.67 kg (n = 3). Average weight gain in patients prescribed clozapine after 12 months was 6 kg; after 24 months it was 15 kg. Over 5 years the individual weight changes ranged from a gain of 55 kg to a loss of 13 kg.
Blood pressure
The mean arterial blood pressure before clozapine was 94.07 mmHg (n = 41); 1 month after clozapine initiation it was 103.38 mmHg (n = 40); 6 months after clozapine it was 102.58 mmHg (n = 31); 12 months after clozapine, 101.4 mmHg (n = 25); 24 months after clozapine, 91.54 mmHg (n = 13); and 60 months after clozapine, 101.33 mmHg (n = 3).
The mean changes in blood pressure 1 month after clozapine was an increase of 9.31 mmHg; at 6 months, an increase of 8.31 mmHg; at 12 months, an increase of 7.33 mmHg; at 24 months, a decrease of 2.53 mmHg; and at 60 months an increase of 7.26 mmHg.
Pulse
The mean pulse before clozapine was 87.51 b.p.m. (n = 41); 1 month after clozapine, 99.53 b.p.m. (n = 40); 6 months after clozapine, 97.42 b.p.m. (n = 31); 12 months after clozapine initiation, 96.28 b.p.m. (n = 25); 24 months after clozapine, 95.15 b.p.m. (n = 13); and 60 months after clozapine, 70.67 b.p.m. (n = 3).
The average changes in pulse 1 month after clozapine was an increase of 12.02 b.p.m.; at 6 months, an increase of 9.91 b.p.m.; at 12 months, an increase of 8.77 b.p.m.; at 24 months, an increase of 7.64 b.p.m. and at 60 months, a decrease of16.84 b.p.m.
Blood glucose
Before clozapine the mean blood glucose was 4.44 mmol L−1 (n = 31); at 6 months after clozapine initiation the blood glucose was 4.9 mmol L−1 (n = 18); at 12 months after clozapine the blood glucose was 4.89 mmol L−1 (n = 15); at 24 months, 5.62 mmol L−1 (n = 5) and at 60 months, 2.5 mmol L−1 (n = 1).
Triglycerides
Before clozapine the mean triglyceride level was 2.38 mmol L−1 (n = 19); at 6 months after clozapine it was 3.1 mmol L–1 (n = 13); at 12 months, 4.33 mmol L−1 (n = 16); at 24 months, 3.75 mmol L−1 (n = 8) and at 60 months, 2.1 mmol L−1 (n = 1).
Cholesterol
The mean total cholesterol level before clozapine was 5.54 mmol L−1 (n = 19); at 6 months after clozapine it was 5.35 mmol L−1 (n = 13); at 12 months, 5.18 mmol L−1 (n = 16); at 24 months, 4.25 mmol L−1 (n = 8) and at 60 months it was 4.5 mmol L−1 (n = 1). Ten subjects (21%) were treated with lipid-lowering drugs.
Liver function
Seven subjects in the clozapine group (15%) had abnormal liver function tests at some point during admission in the forensic setting. Twenty-one subjects (47%) in the clozapine group were hepatitis C positive.
In the comparison group four subjects (15%) were hepatitis C positive and two subjects (8%) had increased liver function tests at some point during admission in the forensic setting.
Pattern of treatment
The mean highest dose of clozapine was 514 mg daily (range 200–900 mg), with a mean highest clozapine plasma level of 1.57 mmol L−1 (recorded in 38 subjects, 59.9%). The highest clozapine plasma level recorded was 5.2 mmol L−1.
Of the clozapine subjects, 37 were continuing with clozapine and 10 had ceased, the mean length of time prescribed clozapine was 18 months. The mean number of antipsychotics prescribed in forensic setting prior to clozapine was 3.95 (range 2–9). In the comparison group the average number of antipsychotics prescribed in the forensic setting was 4 (range 1–8).
Clinical effect
As noted here the clinical effectiveness of treatment was determined using clinical global impression scale. In the clozapine group 12 subjects (25%; 95%CI = 13–38%) were very much improved; 14 subjects (29%; 95%CI = 17–43%) were much improved; 17 subjects (36%; 95%CI = 22–50%) were minimally improved; three subjects (6%; 95%CI = 0–13%) were unchanged and one subject (2%; 95%CI = 0–10%) was much worse.
Nine subjects (35%; 95%CI = 16–53%) of the comparison group were very much improved on treatment; nine subjects (35%; 95%CI = 16–53%) were much improved; four (15%; 95%CI = 2–30%) were minimally improved and four subjects (15%; 95%CI = 2–30%) were unchanged. None of the comparison group had deteriorated on treatment.
Serious adverse effects
Information in relation to serious adverse effects was collected for the clozapine group only. Two subjects (4%; 95%CI = 0–10%) developed neutropenia while on clozapine while three subjects (6%; 95%CI = 0–13%) had seizures.
Reasons for cessation of clozapine
Fifteen subjects discontinued clozapine at some point during their admission in the forensic setting. Reasons were not documented clearly in two cases (13%; 95%CI = 0–30%); sedation was recorded as the reason for cessation for two subjects (13%; 95%CI = 0–30%); weight gain, one subject (7%; 95%CI = 0–19%); patient refusal (poor insight), n = 2 (13%; 95%CI = 0–30%); seizures, n = 1 (7%; 95%CI = 0–19%); hypersalivation, n = 1 (7%; 95%CI = 0–19%); hyperglycaemia, n = 1 (7%; 95%CI = 0–19%); neutropenia, n = 2 (13%; 95%CI = 0–30%); and ineffective (ceased by treating team), n = 1 (7%; 95%CI = 0–19%).
Deterioration following discontinuation
Nine subjects clinically deteriorated after ceasing clozapine (25%; 95%CI = 35–85%). Five subjects who ceased clozapine were later re-prescribed clozapine (33%; 95%CI = 9–57%). Six subjects did not clinically deteriorate after ceasing clozapine (25%; 95%CI = 15–65%).
Concurrent medications
In the clozapine group 11 subjects (23%; 95%CI = 11–36%) were prescribed mood stabilizers; antidepressants, n = 21 subjects (45%; 95%CI = 30–59%); and benzodiazepines, n = 10 (21%; 95%CI = 10–33%). Nine subjects (19%; 95%CI = 8–30%) were treated with methadone while on clozapine and other antipsychotics were prescribed for 12 subjects (26%; 95%CI = 13–38%). General medications prescribed were cardiac medications, n = 3 (6%; 95%CI = 0–13%); hypoglycaemic medication, n = 6 (13%; 95%CI = 3–22%); lipid-lowering medication, n = 10 (21%, 10–33%) and other medications, n = 29 (62%; 95%CI = 48–76%).
In the comparison group four subjects (15%; 95%CI = 2–29%) were prescribed mood stabilizers; five subjects (19%; 4–33%) were prescribed antidepressants; nine subjects (35%; 95%CI = 16–53%) were prescribed benzodiazepines and eight subjects (27%; 95%CI = 13–49%) were prescribed other antipsychotics. Two subjects (8%; 95%CI = 0–18%) were prescribed lipid-lowering medications and none of the comparison group were prescribed hypoglycaemics.
Discussion
This study highlights many of the potential benefits and risks involved in the use of clozapine in the treatment of forensic patients suffering psychotic illness. Managing patients within a well-structured and secure environment with high levels of direct observation and reduced access to illicit drugs may be seen as beneficial. A study comparing clozapine patients in a forensic setting with patients in a general community setting may be useful in examining this issue further.
The age at first diagnosis of psychosis was significantly lower in the clozapine group than the comparison group. This might be because earlier age of onset is associated with a poorer prognosis, differing course and features of illness [24]; clozapine is associated with treatment-resistant patients. The mean time between first diagnosis and first prescription of clozapine was 8 years. This is much longer than has been recommended because evidence suggests that earlier treatment with clozapine is more likely to improve prognosis of patients identified as treatment resistant [25–27]. But other factors may have been involved in decisions not to use clozapine, such as perceived likelihood of non-adherence in the community because of the extremely high rates of substance misuse. There is a striking difference in the incidence of substance misuse in the clozapine group and the comparison group. This difference may suggest that substance misuse is closely associated with treatment resistance or a more severe form of illness. The clozapine group had higher rates of diagnosis of personality disorder but caution should be exercised because it is problematic to make a diagnosis of personality disorder in the presence of a psychotic disorder (and substance misuse disorder). It is possible that a person with a more severe psychotic illness might attract a label of personality disorder due to more severe behavioural disturbance. It could be said that the clozapine group showed evidence of more severe behavioural disturbance. The clozapine group had a higher rate of comorbid medical problems prior to being prescribed clozapine.
Both groups had high rates of violent index offences, with murder being the most common index offence in both groups. Given the evidence of clozapine being effective in reducing rates of violence in patients suffering psychosis, clozapine should be given consideration early in the course of illness, particularly when risk factors associated with future violence are present, for example substance abuse, personality disorder and previous violence.
The clozapine group had higher rates of i.v. drug use than the comparison group with correspondingly higher rates of positive antibodies for hepatitis C. This is important because it suggests that patients considered to be treatment resistant for psychosis have a different, and perhaps more severe pattern of drug use. The higher rates of hepatitis C in the clozapine group might have ramifications for use of clozapine considering the possibility of liver impairment. It also suggests that being hepatitis C positive is a marker for i.v. drug use.
It could be postulated that forensic patients might have more severe forms of psychotic illness and greater disability, with high rates of comorbidity (personality disorder, drug and alcohol problems and medical illnesses) and high rates of violence and suicide. If this is so, then clozapine clearly has an important part to play in their management and rehabilitation. In the present sample we found clozapine to be effective in treatment-resistant patients. Against its beneficial effect in the treatment of psychosis needs to be balanced the potential for clozapine to cause adverse effects and the issue of ensuring medication adherence in the absence of a depot formulation. In the present sample we found high rates of adverse effects, some of which led to being prescribed other medications and to discontinuation of clozapine.
Anecdotally in the NSW Forensic Mental Health Service, there is reluctance among many patients and some staff to prescribe clozapine because of the perceived dangers and adverse effects (such as weight gain, sedation and sialorrhea). Even the fact that many patients who might benefit from clozapine had a background of i.v. drug use meant that the poor quality of venous access became another potential barrier to being prescribed clozapine because of the associated difficulty in obtaining regular blood samples. Other patients who decided to refuse to continue to take clozapine ensured that clozapine was not continued by refusing to have blood taken.
For the future, in order to reduce morbidity associated with clozapine and to reduce perceived barriers to prescribing in cases where clozapine is indicated, we recommend education for patients and staff of the risks and benefits of clozapine. We found that it was difficult to obtain some data from file review that would have been helpful in making more direct comparison between patients prescribed clozapine and other antipsychotics (e.g. changes in weight in the comparison group). In patients being treated with antipsychotics it is essential that there is comprehensive, objective recording of mental state changes (e.g. using validated scales such as the Brief Psychiatric Rating System), and well-documented monitoring of physical and metabolic parameters, with rapid interventions when adverse effects are identified, and rigorous risk/benefit analysis conducted as part of treatment review. This would improve safety for patients, clinical decision-making for patients and their treatment teams and aid research for the future.
