Abstract
Story of Jenny
Jenny was a 28-year-old woman with an 8 year history of schizophrenia. Following treatment with a series of typical antipsychotics that had brought with them multiple side-effects, Jenny was prescribed olanzapine at a dose of 20 mg oral daily; her symptoms were contained and her cognition improved.
Jenny became pregnant at the age of 28. At 6 weeks gestation her general practitioner stopped prescribing all medications for fear of teratogenic complications, and in view of Jenny's lack of psychotic symptoms. Two weeks later, however, she began experiencing hallucinations and paranoid delusions. Jenny became very suspicious of work colleagues and at 20 weeks gestation she physically assaulted a customer, following which her employment was terminated. At 28 weeks gestation Jenny was certified to hospital after attempting to slash her abdomen in response to hearing voices. As a result of this, Jenny's boyfriend told her she could not return to their apartment, rendering Jenny homeless.
Jenny was given oral chlorpromazine, an antipsychotic medication deemed to be safe for the baby, at a dose titrated to 750 mg oral daily. She had postural hypotension, experiencing fainting and possible seizure in hospital, and was subsequently given oral dilantin for 1 week. An ultrasound was performed, and results indicated the baby was small for dates. At 32 weeks gestation Jenny experienced side-effects, complaining of feeling very drowsy, with excessive salivation and upper limb cogwheel rigidity. She continued to hear voices, which instructed her to kill herself and ‘the alien within’, by stabbing her abdomen.
A Caesarean section was planned and performed at 36 weeks gestation, at which the baby girl delivered was flat (unresponsive) and small. The infant was managed in the neonatal intensive care unit for 4 weeks, where she was treated for pneumonia. Her father applied for sole custody, and as Jenny's mental state report detailed a potential risk for infanticide, the application was granted. Jenny was transferred to the psychiatry ward, and was recommenced on a second-generation antipsychotic, aripiprazole 15 mg oral daily. Seven days later her mental state greatly improved with respect to psychotic symptoms, and 3 weeks later she was discharged from hospital, soon after which she developed postnatal depression. Jenny was treated with a selective serotonin re-uptake inhibitor; there was some improvement in her mood but she continued to experience prominent and overwhelming grief over the separation from her baby, with whom she had experienced no contact.
While social workers, legal aid lawyers and psychiatrists attempted to obtain access for Jenny to see her child, the legal processes were slow and Jenny became impatient. She angrily confronted the infant's father, and attempted to take the baby. He called the police, and Jenny was forcibly escorted to hospital. An emergency department doctor made an appointment for Jenny with the psychiatry team for 2 weeks later, and discharged her to their care. Jenny failed to arrive for her appointment; a search revealed that she had committed suicide 2 days earlier. She was found lying next to a tiny pink dress, with a note attached for her baby.
The desire to reproduce is both a powerful urge and a basic human right for women regardless of mental health status. Deinstitutionalized treatment for schizophrenia, better pharmacotherapies, and generally higher expectations for a normal quality of life have the potential to raise the incidence of pregnancy in women with psychosis [1]. A recent epidemiological study found that in a sample of 110 women with schizophrenia, there were a total of 257 pregnancies, resulting in 198 live births [2]. Furthermore, researchers such as Gregoire and Pearson have suggested that changing antipsychotic medication from types such as risperidone, that elevate prolactin levels, to newer brands that do not elicit this effect, may further increase the likelihood of pregnancy [3]. Indeed, birth rates among a small cohort of patients treated with clozapine, which does not cause sustained increase in serum prolactin levels, was higher over a brief period than rates among those who were not receiving the same or similar medication [4]. The right of women with schizophrenia to become parents, in addition to the potentially increasing birth rate, places responsibility upon health-care professionals to ensure that sound antenatal and ongoing care is available.
Ethical duty of care aside, good antenatal treatment appears prudent given the greater risk of obstetric complications in women with psychosis [5]. A recent review of the obstetric records of 618 births to women with schizophrenia found that this cohort was more likely to experience placental abnormality and to need the administration of a narcotic antagonist (naloxone) than women in the general community [6]. This remained the case after adjustments were made for maternal age, parity, plurality, marital and indigenous status [6]. Moreover, an odds ratio of 1:3.8 was detected for these babies to be within the lowest 10th percentile of intrauterine growth, again following statistical control for the confounding variables [6]. Interestingly, maternal onset of mental health problems might also bear a relationship to outcomes. Jablensky et al. discovered that women whose psychiatric illness commenced before the birth of their child were significantly more likely to experience obstetric complications than women whose first admission for mental illness was after the delivery [6]. Taken together, this collective body of knowledge seems to suggest that maternal psychosis contributes a potential risk to obstetric outcomes, which indicates an area ripe for investigation.
Mortality rates and other post-partum measures for infants additionally provide considerable impetus to facilitate exemplary obstetric intervention. Evidence from a meta-analysis points to a small but statistically significant increase in the risk of low birthweight and inferior neonatal condition in infants born to women within the schizophrenia cohort [5]. Results from another meta-analysis by Webb et al. were less conservative; the authors concluded that exposure to maternal psychotic illness is associated with a ‘higher than expected’ risk of perinatal and infant mortality, including an almost twofold chance of fetal death or stillbirth [7]. Similarly, the likelihood of stillbirth, infant death, preterm delivery, low birthweight and small size for gestational age among 2096 infants born to mothers with schizophrenia were generally double that of 1 555 975 babies of women from the general population, even after smoking and other maternal factors were controlled for [8]. Looking further into the future, the offspring of women with schizophrenia also carry with them an increased risk for developing psychosis [9]. This observation has driven the neurodevelopmental hypothesis about the aetiology of schizophrenia, which posits that alterations to early fetal brain development in utero might lead to a predisposition to the formation of schizophrenia later in life [10].
Antipsychotics in pregnancy
Despite these clear arguments for a thorough understanding of the needs of pregnant women with schizophrenia, questions remain about appropriate care and treatment. This is especially the case with regard to the safety of administering antipsychotic medication upon fetal development and maternal health. Psychotropic medication passes through the placenta [11], and furthermore, lower levels of protein in fetal plasma render a greater portion of the drug unbound, facilitating entry into the fetal brain [12]. Metabolism of medication is less efficient in the fetus due to slower rates of excretion, which, coupled with their less active liver enzymes, the immaturity of the nervous system and the blood–brain barrier, causes unborn babies to be at a potentially greater risk of toxicity and more susceptible to drug effects [12].
Typical antipsychotics
Over the last four decades intermittent studies have sought to investigate the risk of teratogenicity and other problems of fetal development associated with typical antipsychotic use. Earlier reports were generated from pregnancy outcomes of women treated with these medications for hyperemesis gravidarum, not from women with schizophrenia [13]; as such, it is difficult to extrapolate initial findings across these two cohorts with confidence. Sobel that found women given chlorpromazine during pregnancy had infants with a higher rate of fetal damage or death than did women from the general population, although differences did not reach statistical significance [14]. In their review, Patton et al. concluded that a significant increased chance exists for babies exposed to phenothiazines of developing congenital malformations compared with the incidence within the general population [15]. Results from a meta-analysis of 74 337 births were similar, indicating that the administration of low-potency phenothiazine during the first trimester of pregnancy increased the risk of congenital abnormalities from 2% to 2.4% [16].
Impact upon postnatal behavioural sequelae for infants exposed to typical antipsychotics tells a similar story of increased risk, albeit with less deleterious consequences. The reported incidence of neonatal toxicity comes largely from single case studies reviewed by Altshuler et al. [16]. Findings indicated that infants exposed to such medication may experience motor restlessness, tremor, hypertonicity and transient heart block. While these perinatal syndromes may persist for up to 10 months, most are ephemeral. The authors also concluded that outcome measures of behaviour functioning and IQ did not differ between children with and without a history of neuroleptic exposure [16].
Atypical antipsychotics
Prescribing guidelines laid out in MIMS (2003) for all current second-generation antipsychotics include pregnancy as a ‘special precaution’. As with the older class of medications, there are a modest number of publications examining atypical antipsychotic use in pregnant women. In their review, Patton et al. noted that for a total five cases cited, no congenital abnormalities were reported following clozapine use, although one infant experienced low-grade fever and seizure, with gastro-oesophageal reflux [15]. Grigoriadis and Seeman suggest that the increased risk of agranulocytosis and seizures in babies, coupled with the identification of both pregnancy and clozapine as potentially increasing the likelihood of thromboembolisms, render this a medication to be avoided during pregnancy [17]. Reported findings of quetiapine use in expectant women are scarce, which is to be expected because quetiapine is a relatively new antipsychotic in the Australian pharmacopoeia. One case study exists describing a patient treated with quetiapine throughout her pregnancy and post-partum period, without maternal or infant complications [18]. Twenty-three olanzapine-exposed pregnancies were observed by Goldstein et al., who found that there was no increased risk of spontaneous abortion, stillbirth, prematurity or major malformation [19]. The authors were swift to assert that studies of greater sample sizes, over a protracted time period, are necessary before olanzapine can be declared safe with confidence.
Indirect risk factors may also call into question the suitability of antipsychotic prescription in pregnancy. A review by Patton et al. found that either olanzapine or clozapine were associated with a danger of weight gain and diabetes mellitus, both of which can adversely influence obstetric outcomes [15]. In another investigation a random sample of 70 inpatients treated with atypical antipsychotics were examined with regard to factors associated with neural tube abnormalities; results indicated that not only were the majority of patients overweight or obese, but only eight had folate intake at or above the levels recommended for protection against neural tube defects [20]. Serum folate levels were also significantly lower than those attained from a non psychiatric patient sample [20]. The authors suggest that many women being treated with atypical antipsychotics are at increased risk of having babies with neural tube defects because of both their low intake of folate and maternal adiposity [20]. But the sample was not pregnant, meaning that this projected risk was based upon behaviour and management in a sample for whom folate levels may not be a current significant concern.
To further complicate the issue of medication for psychosis during pregnancy, the mental health of a woman may alter during her expectant state. Seeman outlines physiological changes, including increased blood plasma volume, body fat and renal elimination, plus faster drug metabolism, all of which lower plasma drug concentrations during pregnancy [12]. Investigations have indicated that symptoms of psychosis ameliorate in pregnancy [21] and the need for medication is reduced [22]; conversely, symptoms of psychosis may prevent women from seeking appropriate health care [23]. Deterioration in mental state is not only highly distressing for the woman, it can affect her ability to bond with, and parent, her child. To this end, more data that plot the course of symptoms for women, both antenatally and in the post-partum period, in combination with antipsychotic medication information, are required to enable judicious treatment advice.
Guidelines published by the American Academy of Pediatrics Committee on Drugs acknowledge that information to date regarding the risks of drugs administered during pregnancy is derived largely from either case studies or from retrospective epidemiological observations [24]. Biases, therefore, are potentially inherent due to confounding factors such as maternal age, health, use of other licit and/or illicit substances, presence of more than one type of medication, compliance, time of fetal exposure, maternal genetic and obstetric history [24]. A proven, evidence-based practice for achieving a balance between maternal mental health, with minimal risk to the fetus, must be researched and substantiated. Ethical considerations restrict the use of randomization in this patient population [25], therefore we have established a National Register of Antipsychotic Medication in Pregnancy (NRAMP), which was loosely based on the Australian Register for Women on Antiepileptic Medication [26]. NRAMP is a non-interventionist means of gathering information about pharmacotherapy during pregnancy, and outcomes for women with psychosis, and their babies. A secondary aim is to capitalize upon the opportunity to track alterations of mental health symptoms for women during pregnancy and in the post-partum period, and to garner valuable insights into the attitudes of this cohort during their transition into parenthood.
The objective of this paper was to present details regarding the methodology and preliminary demographic information about this exciting and highly needed register.
Methods
Participants
Locality and sample size
Women are recruited across Australia, from sites including hospitals, mental health services and universities. Using national projections of pregnancy rates and an estimation of the number of women of child-bearing age with psychosis, it is anticipated that 100 women could be enrolled over a 2 year period.
Inclusion criteria
Enrolment into NRAMP is open to pregnant women treated with an antipsychotic medication, with a history of psychosis such as schizophrenia, schizoaffective disorder, first-episode psychosis and bipolar affective disorder with psychosis. The inclusion criteria are necessarily broad because the basic question pertains to the relationship of the antipsychotic medication to maternal and baby health. There are no exclusion criteria, although other substance use details, including medications for concomitant conditions, alcohol, nicotine and illicit substances are carefully recorded.
Recruitment
Recruitment has occurred indirectly by targeting clinicians, health services and other professional bodies, and has incorporated the use of extensive mail outs, national and state advertising campaigns, establishment of an 1800 telephone number, development of a website and direct contact with community mental health services and clinics. Participants have not been directly approached by researchers, but this may be a future step.
Materials
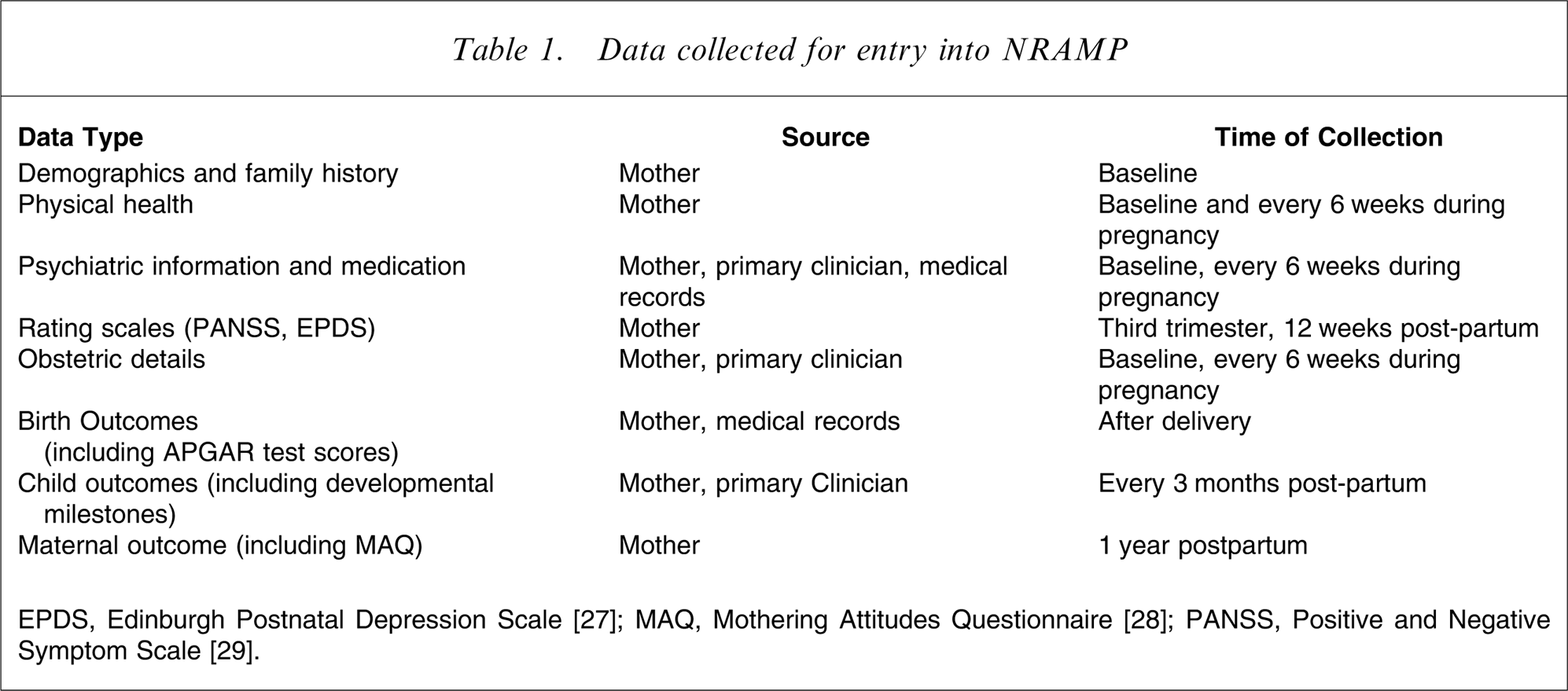
Information regarding each woman and her infant is gathered every 6 weeks throughout her pregnancy, at the time of delivery, and at 6 weeks, 12 weeks, 6 months and 12 months after delivery. It is supplied by both direct interview and medical records, and encompasses personal history, physical and mental health of the mother, psychiatric management and outcomes, and pregnancy and delivery details, as well as health and developmental outcomes for the infant. A more detailed inventory of each assessment is presented in Table 1.
Data collected for entry into NRAMP
Data analysis
Preliminary analyses are based on demographic data. Future analyses will explore relationships between psychotic symptoms, medication (dose and type), and pregnancy stage and infant outcome measures, such as APGAR test scores and developmental tasks.
Results
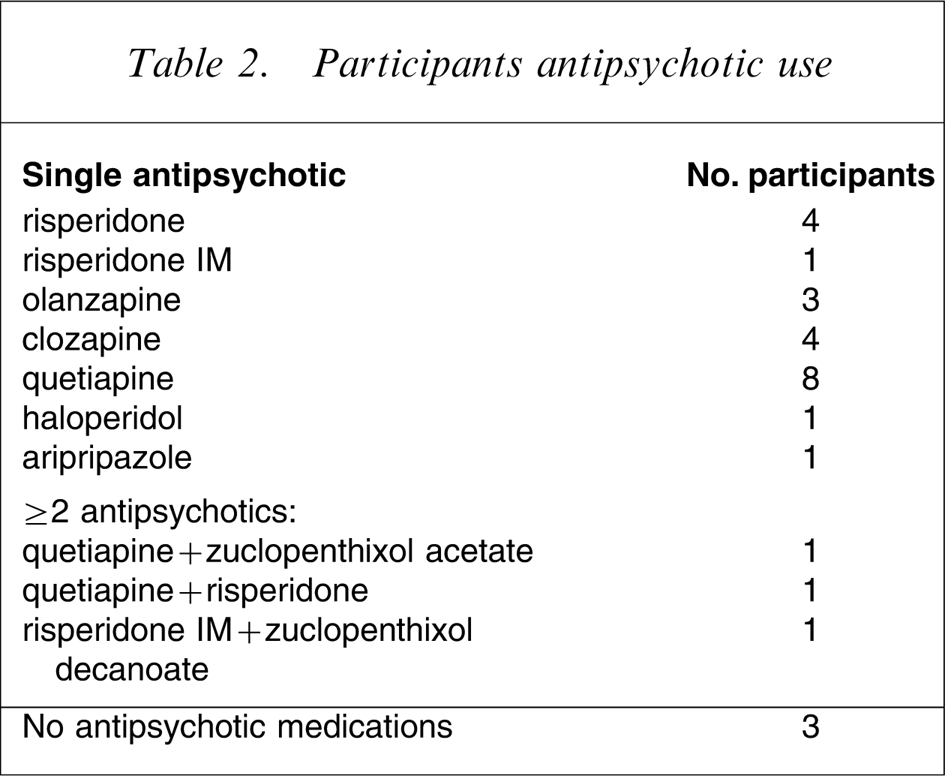
Approval for the study has been granted by 14 ethics boards overseeing 21 sites across Australia to date, with approval from additional sites pending. Demographic information is available for the first 30 participants, who were aged 29–40 years (Figures 1–3). Antipsychotic medications received antenatally are presented in Table 2. Many of the participants were also receiving concomitant medications, including mood stabilizers such as lithium carbonate and Sodium valproate; selective serotonin re-uptake inhibitors such as citalopram, sertraline, Venlafaxine and fluoxetine; anticonvulsant medications such as lamotrigene; anxiolytics such as diazepam, temazepam and clonazepam; and other medications such as methadone and salbutamo.
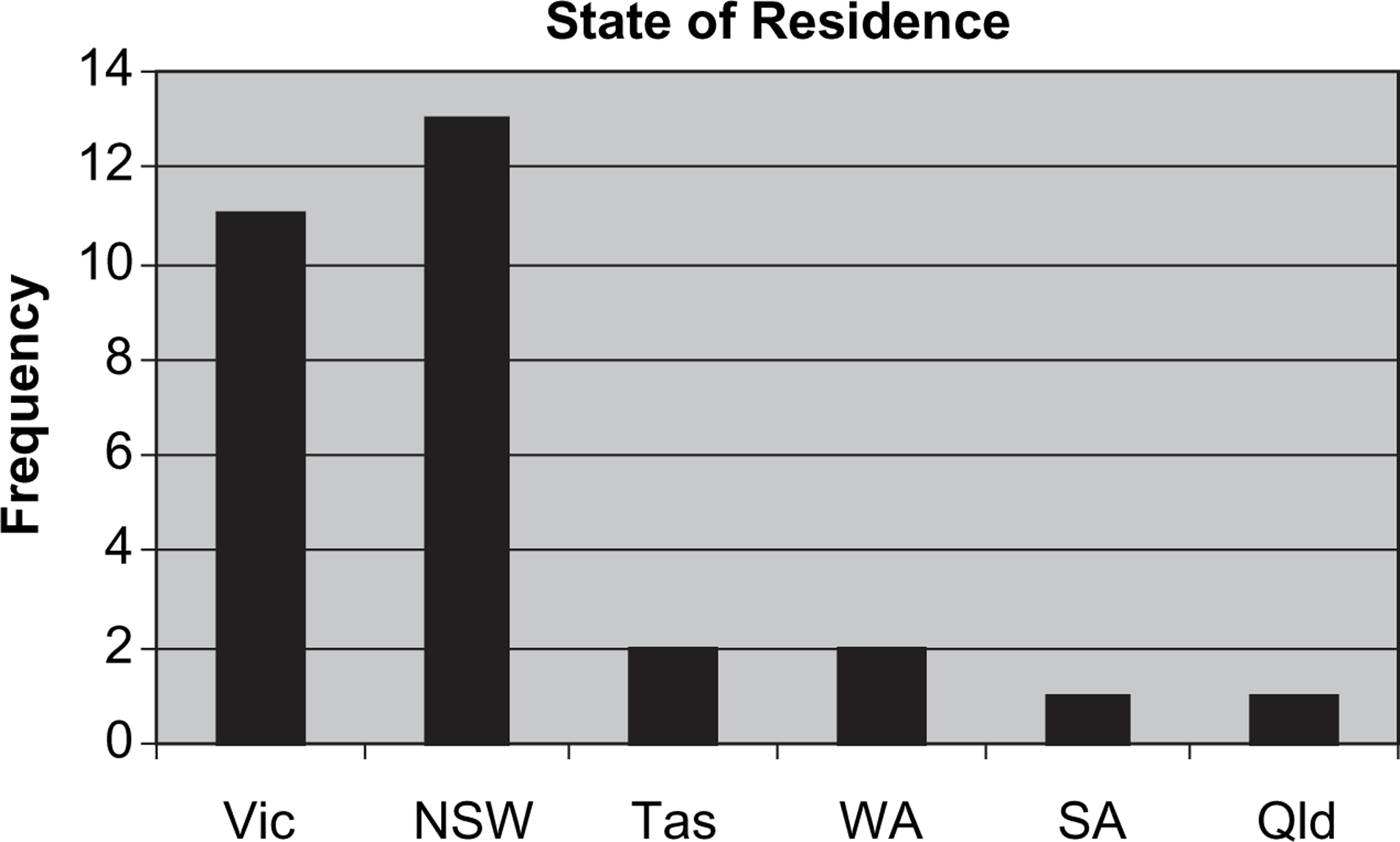
State of residence for the first 30 participants in the National Register of Antipsychotic Medication in Pregnancy.
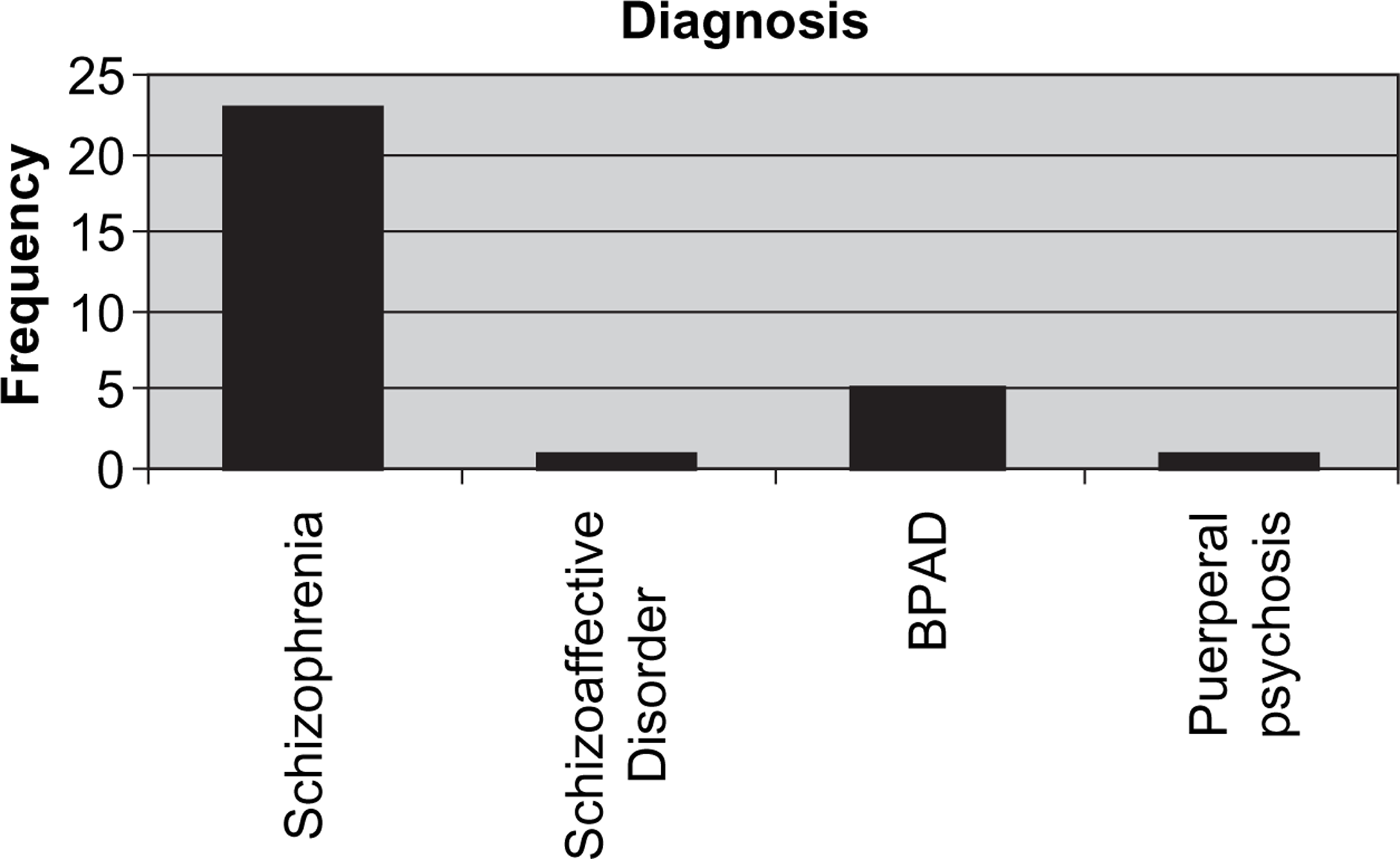
Diagnoses of the first 30 participants in the National Register of Antipsychotic Medication in Pregnancy. BPAD, bipolar affective disorder.
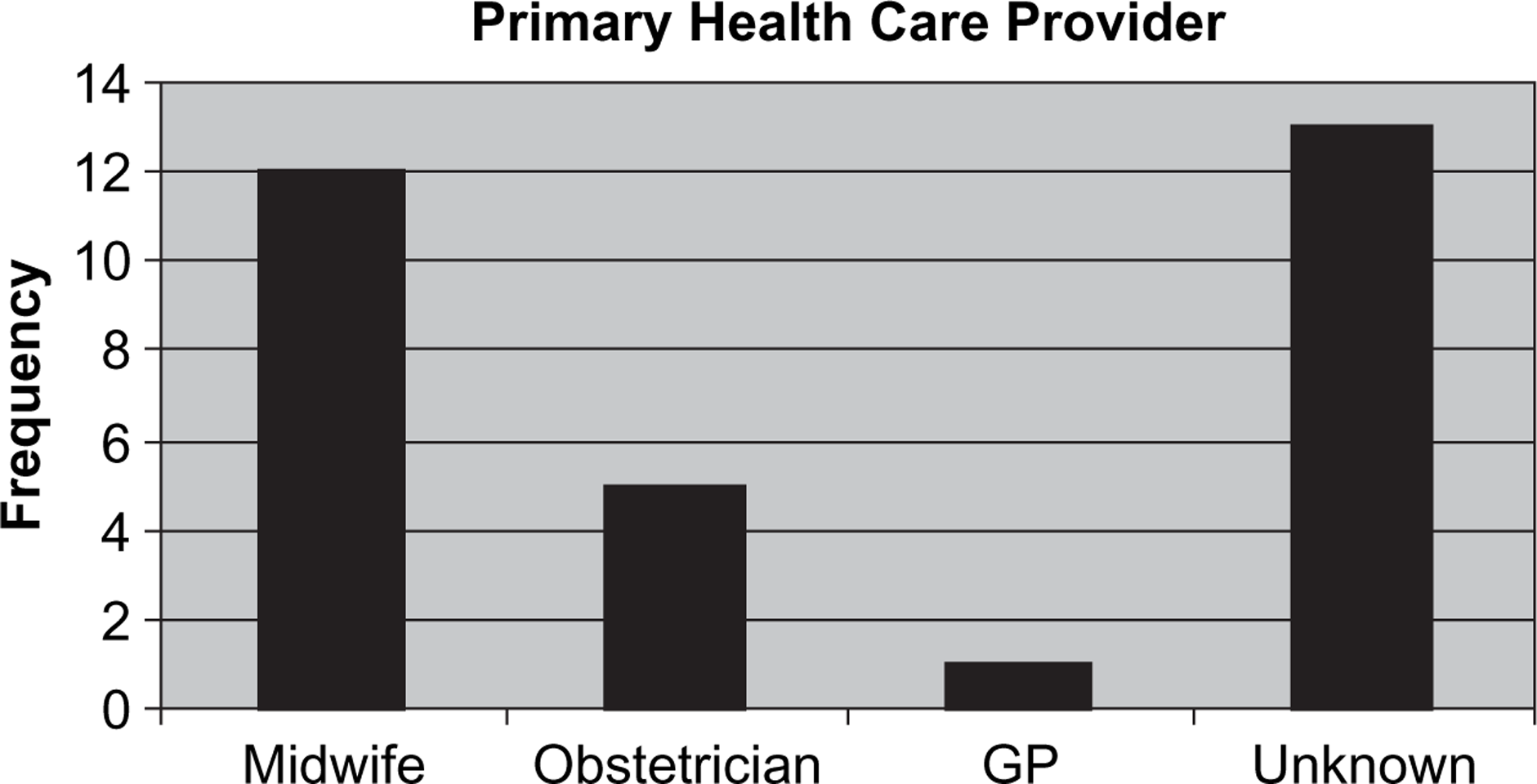
Primary health-care provider for the first 30 participants in the National Register of Antipsychotic Medication in Pregnancy. GP, general practitioner.
Participants antipsychotic use
Conclusion
The story of Jenny not only highlights the vulnerable and desperate situation in which women in this population group can find themselves, but illustrates the very clear need for information surrounding the pregnancy and outcomes for women taking antipsychotic medication. The establishment of NRAMP, focusing as it does on the broader needs and vulnerabilities of pregnant women requiring antipsychotic treatment, will enable us to develop guidelines for their optimal health-care interventions. It is hoped that NRAMP will become a national resource, created with the collaboration of patients, carers, clinicians and researchers, which will improve confidence in management strategies, and enhance the quality of life and the experience of parenting for participants and their families. Through the establishment of the register we also have an opportunity to potentially improve the mental health of future generations of Australians, thereby providing a truly protective approach to mental illness.
Footnotes
Acknowledgements
This study has been approved by 14 sites nationally, with further approvals pending. We gratefully acknowledge the support of Astra Zeneca Pharmaceuticals, Janssen-Cilag Pharmaceuticals and Mayne Pharmaceuticals in providing grants to assist the funding of this project.
