Abstract
Forty-five years ago the surprising discovery was made, in a Melbourne University laboratory, that peripheral synapses exist that release neither noradrenaline nor acetylcholine. The same laboratory went on to show that one of these then novel transmitters is adenosine 5′-triphosphate (ATP), for which a class of receptors has been dubbed P2X7. Recent linkage studies have shown that the P2X7 gene is associated with major depression and bipolar disorder. This speculative paper considers possible mechanisms that could link polymorphisms in the P2X7 gene with the functioning of neural networks, especially in the hippocampus. A selective review of the neurobiological literature on the location and function of the P2X7 receptor at synapses and on astrocytes as well as microglial cells was performed in the context of determining viable hypotheses as to the function of these receptors during synaptic transmission in the neural networks of the hippocampus. It is suggested that P2X7 receptors participate in a regenerative loop at central glutamatergic synapses. In this loop glutamate-evoked release of ATP from both astrocytes and microglia cells, as well as ATP derived from an autocatalytic release from astrocytes, provides purines that can act on presynaptic P2X7 purinergic receptors. This increases glutamate release to further the amount of ATP at the synapse, leading to a new functional state of the neural network in which the synapse participates. This synaptic ATP can also act on microglia P2X7 receptors to release the cytokine tumour necrosis factor-α (TNF-α), as can glutamate, with this TNF-α acting on the post-synaptic neuronal membrane to increase glutamate alpha-amino-3-hydroxy-5-methyl-4-isoxazolepropionate (AMPA) receptors there. As synaptic ATP and glutamate are maintained by the regenerative loop they provide a sustained release of TNF-α, and therefore of AMPA receptor enhancement, increasing synaptic efficacy, and so contributing to the new functional state of the neural network. Infections can change this state by activating toll-like (TOL) receptors on the microglia concomitantly with their P2X7 receptor activation by the regenerative loop, thereby releasing the cytokine interleukin-1β, which decreases the AMPA receptors in the neural membrane, so decreasing synaptic efficacy and changing the functional state of the neural network in which the synapse resides. Polymorphisms in the P2X7 gene that modify operation of the regenerative loop or the release of cytokines, as can infections, change the functional state of neural networks, which may then lead to vulnerability to mood disorders.
At Christmas 1962 synaptic potentials were discovered in the peripheral nervous system with very different characteristics to those expected of nerve terminals that release either noradrenaline or acetylcholine, the only transmitters thought at that time to exist, because the synaptic potentials could not be blocked by appropriate receptor antagonists and so were called non-adrenergic non-cholinergic (NANC; [1, 2]; for a history see Ch. 7 in [3]). Identification of the first NANC transmitter did not take long, when the same superfusion technique introduced by Loewi some 40 years earlier to identify acetylcholine as the vagal transmitter to the heart [4] was used to suggest the purine adenosine 5′-triphosphate (ATP) as a likely NANC transmitter [5, 6]. Subsequently a distinction was made between ionotropic receptors for ATP (P2X), through which ions flow on agonist activation, and metabotropic receptors for ATP (P2Y), which activate intracellular biochemical pathways ([7]; for a history see [8]) of which one is the P2X7 receptor, formerly called the P2Z receptor [9]. In the central nervous system P2X7 receptors are principally found on microglial cells, to some extent on astrocytes and on nerve terminals [10–13]. Very surprisingly, polymorphisms in this receptor have recently been implicated in both bipolar disorder and major depression [14, 15]. This speculative essay presents a hypothesis linking these conditions to the function of P2X7 receptors found at synapses.
Astrocyte processes at the neuronal synapse
Each astrocyte occupies a discrete volume of the brain such that the processes of one astrocyte do not overlap those of another but come into direct apposition with those of adjacent astrocytes, with each astrocyte encompassing on average approximately 10 000 synapses in the neuropil [16]. Calcium waves, originating in the triggered release of calcium from the endoplasmic reticulum, propagate through networks of astrocytes [17]. The propagation of the calcium wave occurs as a consequence of purinergic transmission between individual astrocytes, involving the release of ATP from astrocytes in particular parts of the brain such as the hippocampus as well as the spinal cord (Figure 1; [18]). There calcium waves can propagate for long distances through astrocyte networks (>600 µm in vitro; [19]; for a review see [20]), as a consequence of the autocrine release of ATP, in which ATP acting on P2Y receptors promotes its own release (Figure 1; [21]). Glutamate released from nerve terminals has the potential to evoke ATP release from astrocyte processes at the synapse [22]. Such extracellular synaptic ATP can establish an autocrine release of further ATP from the astrocyte independent of further glutamate release (Figure 1; [21, 23]). This synaptic ATP can act on a variety of presynaptic P2 receptors, including P2X7 receptors, if ATP reaches sufficient concentrations, to increase transmitter release ([24]; for a review see [25]). A feedback loop is thereby established in which ATP increases glutamate release from the nerve terminal, which in turn can enhance ATP release from astrocytes, enhancing the autocrine ATP release mechanism further (Figure 1). In addition presynaptic P1 receptors that decrease transmitter release may be activated if suitable ectoenzymes are present to metabolize synaptic ATP to adenosine [26].
The tripartite synapse. This consists of pre-synaptic and post-synaptic processes of the neuronal synapse together with those of an astrocyte. In the hippocampus astrocytes conduct intracellular calcium waves that are transmitted between astrocytes using the purine adenosine triphosphate (ATP). In addition, ATP acting on astrocytes releases glutamate from them. Thus ATP and glutamate released from an astrocyte process in the tripartite synapse can act on the neuronal pre-synaptic purinergic receptors for nucleotides (P1 receptors for adenosine that inhibit the release of glutamate; P2, including P2X7, receptors for ATP that enhance the release of glutamate) as well as on post-synaptic glutamate alpha-amino-3-hydroxy-5-methyl-4-isoxazolepropionate (AMPA) receptors. In addition, glutamate released from the pre-synaptic process can release ATP from the astrocyte process. It is important to note that the release of ATP from astrocytes is regenerative: that is, it recruits an autocatalytic mechanism by which ATP released from the astrocyte acts back on the astrocyte to promote its own release, as indicated by the large black arrow.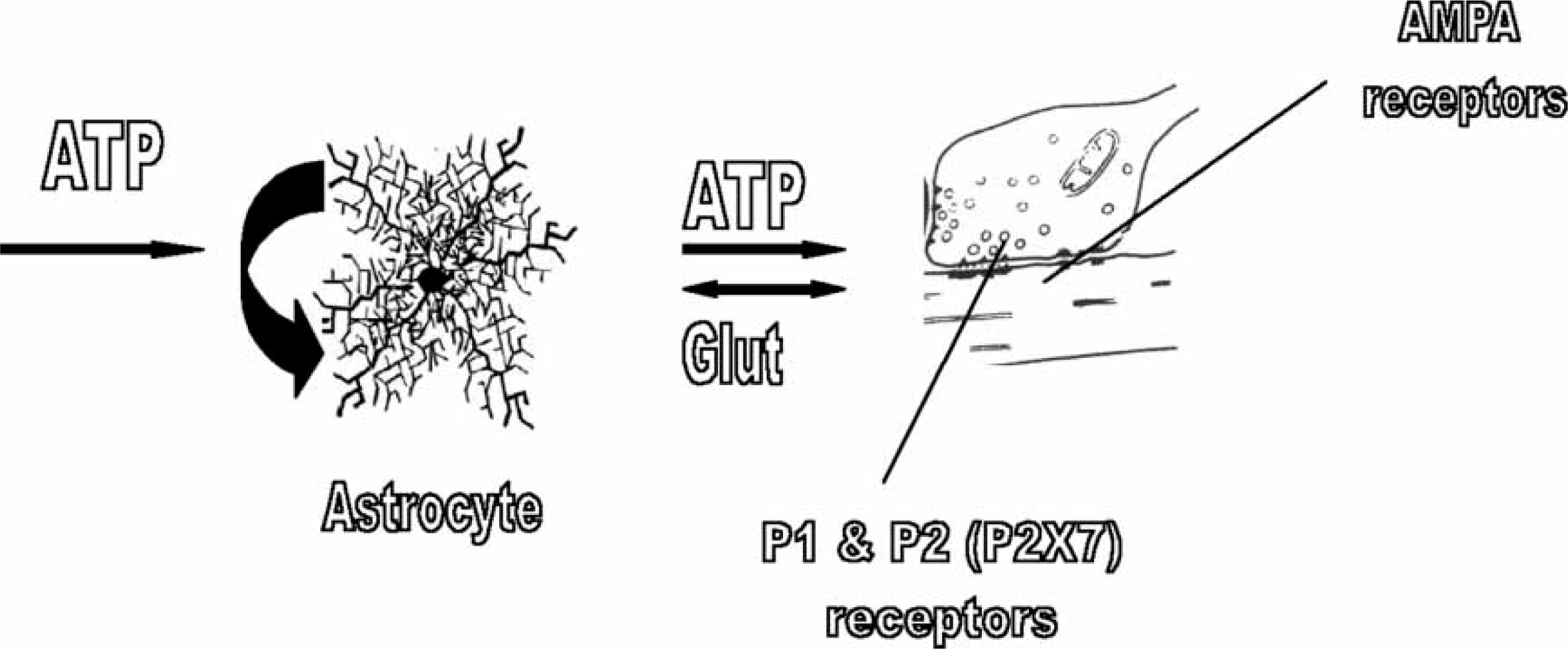
The discovery that the calcium wave leads to the secretion of glutamate from astrocytes and that this acts on neuronal synapses within the neuropil radically changed our conception of the function of astrocytes [27]. This, together with the fact that ATP released at junctions between astrocytes could also act on presynaptic P2X receptors at neuronal synapses gave rise to the idea of the tripartite synapse consisting of pre- and post-synaptic neuronal processes as well as an astrocytic one (Figure 1; [28, 29]). The concept is now well established that all parts of the brain must be considered as consisting of neuron–astrocyte networks the function of which is dependent on the integrity of the tripartite synapse [30, 31].
Microglia and astrocytes processes at the neuronal synapse
Microglia cells were thought to occur in low density throughout the brain but recent studies show that they too contribute a dense coverage of the neuropil [32]. Microglia possess a higher density of P2X7 receptors than any other cell type in the brain, with these receptors requiring a larger concentration of ATP to be activated [10, 13]. Importantly these cells respond to ATP with a calcium transient, which modulates the release of cytokines, nitric oxide and superoxides [33].
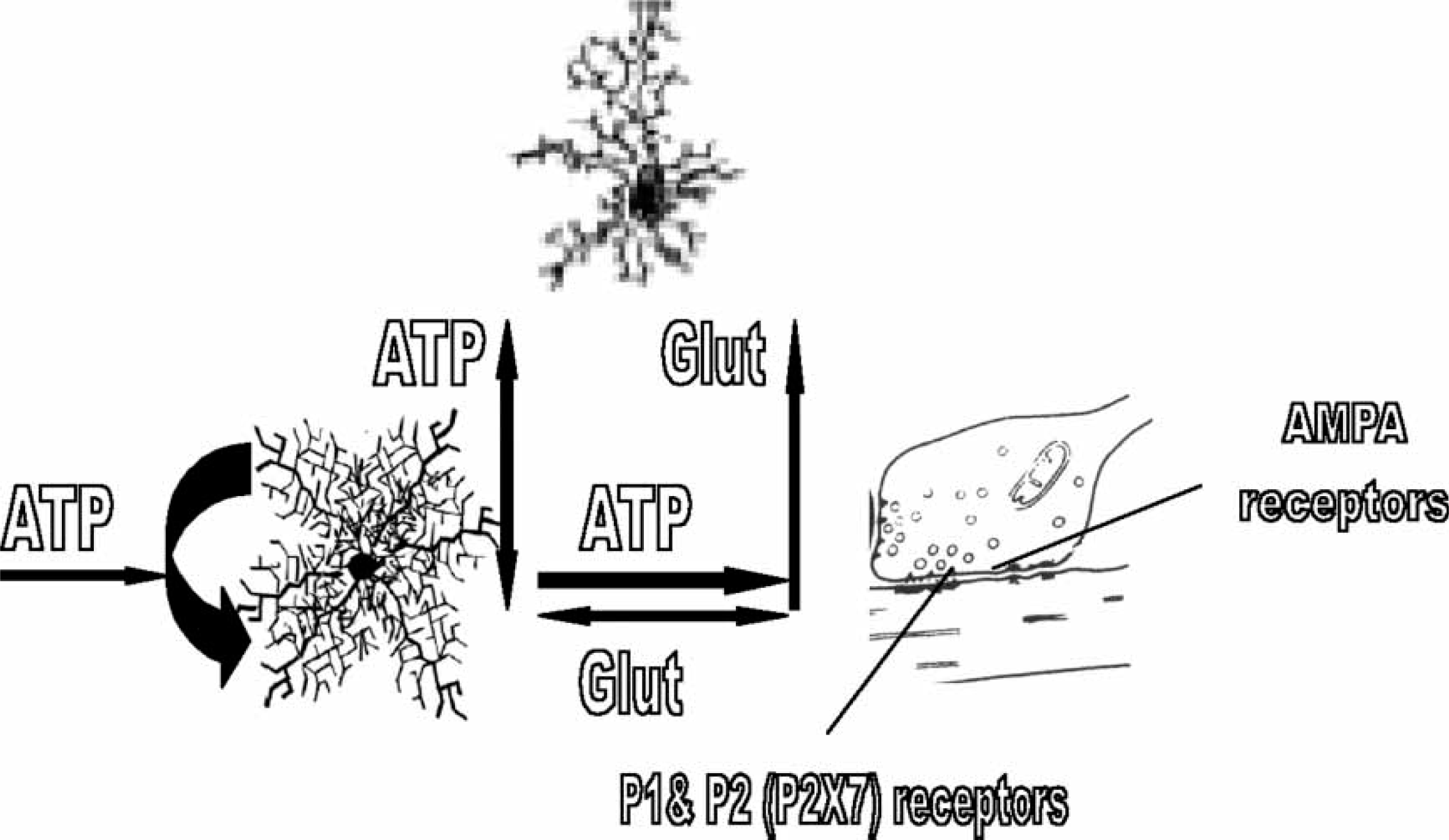
Because microglia have a high level of motility they are readily attracted to sites that release suitable chemoattractants. Microglia processes can be attracted into intimate contact with synapses by the release from nerve terminals of chemokines such as fractalkine, which is specific for microglia (Figure 2; [34]). A chemokine which is a major chemoattractant is monocyte chemoattractant, protein-1 (MCP-1), released from astrocytes as a consequence of the action of ATP on their P2X7 receptors (Figure 2; [35, 36]). Furthermore ATP itself acts as a chemoattractant to recruit microglia to synapses by acting on P2Y receptors (Figure 2; [37, 38]). The process of chemoattraction involves first, the directional movement of microglia processes towards the source of the chemoattractant, and later, migration of the whole cell body can occur at velocities between 20 and 35 µm h–1[38, 39]. Once microglia processes are intercalated at the synapse they can themselves release ATP due to nerve-terminal-derived glutamate promoting this secretion (Figure 3; [40]). Microglia processes may then be stabilized at the synapse by glutamate-evoked ATP release from both microglia and astrocytes, with the latter contributing further ATP through an autocrine release that, once triggered, may sustain synaptic ATP even in the absence of glutamatergic transmission (Figure 3). The tripartite synapse then evolves into the quadpartite synapse consisting now of pre- and post-synaptic neuronal processes as well as astrocytic and microglial processes (Figure 3).
The quadpartile synapse. The release of purine adenosine 5′-triphosphate (ATP) from astrocyte processes at the tripartite synapse, through the action of glutamate and ATP on these processes (Figure 1), promotes the chemoattraction of nearby microglia as does the release of fractalkine from pre-synaptic processes. This leads to the intercalation of microglia processes into the tripartite synapse, so forming a quadpartite synapse composed of pre- and post-synaptic processes together with those of astrocytes and microglia. Pre-synaptic P2X7 receptor function to enhance glutamate release and so synaptic efficacy. The pre-synaptic process at the quadpartite synapse releases glutamate onto both astrocyte and microglia processes, which triggers their release of purine adenosine 5′-triphosphate (ATP) that adds to that provided by the autocatalytic ATP release from the astrocytes. The resulting high concentration of ATP can then act on pre-synaptic P2X7 receptors to promote the release of glutamate. There is then a positive feedback pathway in which the release of glutamate maintains its own high level of release through glial-derived ATP and the pre-synaptic P2X7 receptor. This maintains the synapse in a high state of efficacy. AMPA, alpha-amino-3-hydroxy-5-methyl-4-isoxazolepropionate.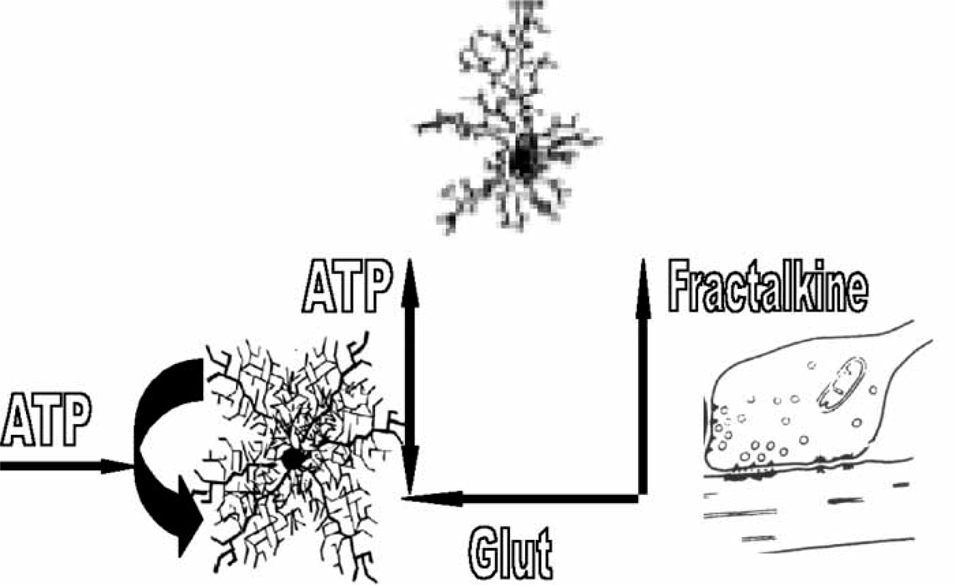
ATP may also act on synaptic microglia P2X7 receptors to evoke the release of cytokines such as tumour necrosis factor-α (TNF-α), with this cytokine also released by glutamate itself (Figure 4; [41–44]). TNF-α then increases AMPA receptors for glutamate in the post-synaptic membrane processes [45]. Thus the P2X7 receptor mediates mechanisms that give rise to significant changes in the efficacy of synapses both through changes in transmitter release and the expression of receptors for the transmitter. Once begun these mechanisms may be sustained by the autocrine release of ATP by astrocytes. The efficacy of synaptic transmission can then be set by mechanisms involving the P2X7 receptor, so that the function of the neural network in which these synapses operate is set by mechanisms mediated by P2X7 receptors.
Microglia P2X7 receptors function to release the cytokine tumour necrosis factor-α (TNF-α), increase glutamatergic receptors and so synaptic efficacy. If adenosine 5′-triphosphate (ATP) reaches sufficient concentrations at the quadpartite synapse to activate P2X7 receptors on microglia it can trigger the release of the pro-inflammatory cytokine TNF-α. Glutamate can also release TNF-α from the microglia processes. This TNF-α then acts of the post-synaptic processes to enhance the expression of glutamatergic AMPA receptors in their membranes. This then maintains the synapse in a high state of efficacy.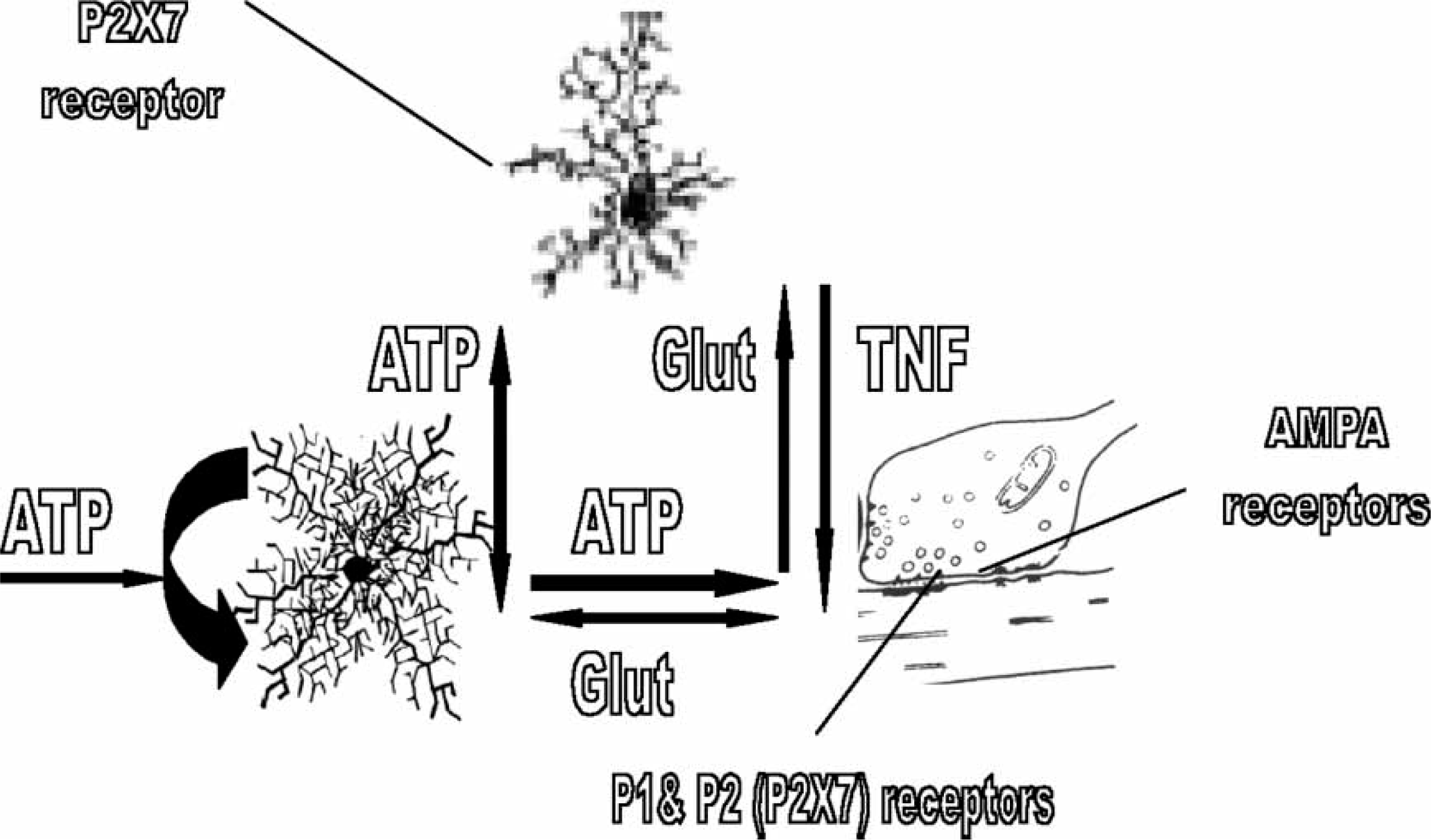
Infection and sustained activation of microglia processes at the neuronal synapse
The potential for forming quadpartite synapses varies throughout the brain because the distribution of microglial cells in the brain is not uniform. For example, the rat substantia nigra has a much higher resident population of microglia than does the hippocampus, so that direct injection of endotoxins (lipopolysaccharides, LPS) in the former produces much greater neurodegeneration than in the latter [46]. However, there is probably variation in the density of microglia within a single structure because injections of LPS into the hippocampus give localized microglia activation in the granule cell area of the dentate gyrus, spreading somewhat later to the hilus, but leaving the rest of the hippocampus unaffected [47]. Such heterogeneity in microglia density within a single structure has been observed directly in the mouse cerebellum, varying in distinctive ways between different histological layers [48].
There is also heterogeneity in the extent of expression on microglia of different Toll-like receptors (TLRs), the cell surface receptors that are involved in recognition of microbial structures and in the release of cytokines. Direct injection of infective agents into the brain shows that among the Toll-like receptors it is TLR2 whose expression is most enhanced. Injection of Gram-negative bacteria (Escherichia coli, meningitis) initiates an 80-fold increase in TLR2 and a fourfold increase in TLR4, whereas injection of Gram-positive bacteria (Streptococcus pneumoniae) gives a 10-fold increase in TLR2 and a fourfold increase in TLR9, and injection of the Herpes simplex virus a fourfold increase in TLR2 only [49].
The circumventricular organs, namely hippocampus, caudate and hypothalamus, show the greatest potential for activated microglia to participate in quadpartite synapse formation following a bacterial or viral infection [50, 51]. The systemic blood to lateral ventricle cerebrospinal fluid pathway provided by the choroid plexus is clearly the main route for such infections as experimental autoimmune encephalomyelitis to reach the brain [52], although other pathways also exist [53–55]. This pathway regularly supplies a range of growth factors and hormones to the circumventricular organs [56]. Microglia in these organs show a constitutive and transcriptional expression of the LPS receptor TLR4 minutes after LPS appears in the systemic circulation, appearing some hours later in the rest of the cortex, with high levels of expression in the circumventricular organs maintained long after the systemic LPS has been cleared [50, 57].
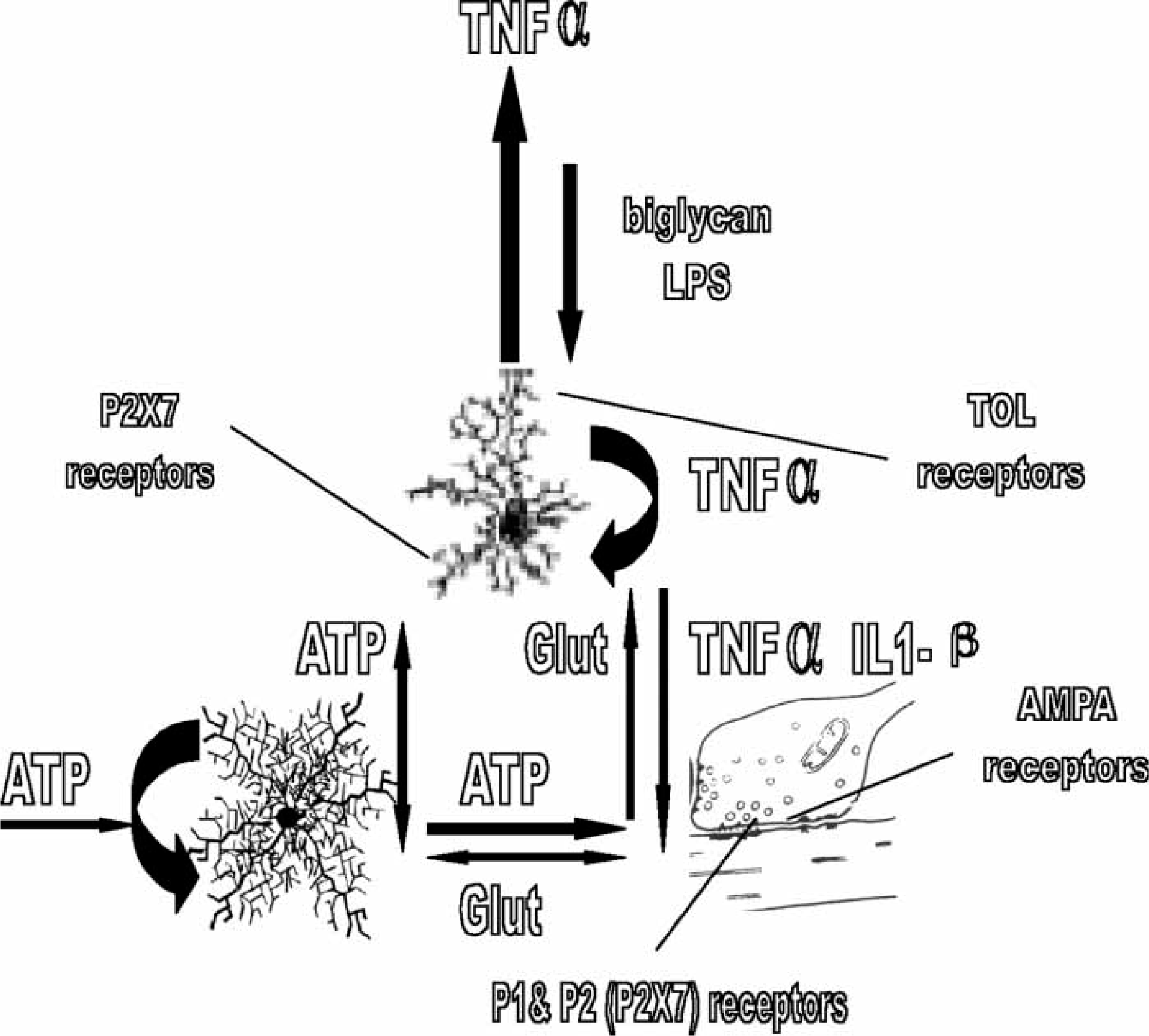
Of great interest is the recent observation that both hippocampus and cortex possess activated microglia releasing TNF-α some 0.5 h after systemic LPS injection (Figure 5), rising to approximately threefold that density in 3 h, and then maintaining it for at least a further 10 months after the LPS has been cleared from the circulation and all signs of elevated TNF-α release from peripheral organs has disappeared (Figure 5; [58]). The autocrine activation of microglia by TNF-α is almost certainly responsible for the high levels of TNF-α release by the activated microglia following infection [59]. This has the potential at the quadpartite synapse of providing a sustained pool of TNF-α, with autocrine action of this on microglia processes maintaining the pool after the initiating infective agent has passed (Figure 6). Such a maintained pool of TNF-α determines that the increase in post-synaptic AMPA receptors is sustained, resulting in permanent changes in neural network function. Not only exogenous agents such as LPS but also endogenous agents such as biglycan, released from astrocytes have the potential to initiate the TNF-α autocrine loop (Figure 6). Such high levels of TNF-α at quadpartite synapses can then diffuse to adjacent unaffected synapses, so propagating glial cell activation throughout the parenchyma (Figure 6; [60]). In addition, LPS or biglycan together with ATP release the cytokine interleukin (IL-1β), as a consequence of the action of ATP on P2X7 receptors (Figure 6; [61, 62]). This cytokine decreases the expression of AMPA receptors in post-synaptic membranes [63], see also [64]. In this way IL-1β provides a further means of altering the efficacy of synaptic transmission and hence the functioning of neural networks.
Transient infection can lead to sustained release of tumour necrosis factor-α (TNF-α) from microglia. Transverse sections through rodent cortex and hippocampus (CA1, CA2, CA3) showing sites of activated microglia releasing TNF-α (black dots). Upper panel is taken at 0.5 h after systemic injection of gram-negative bacterial endotoxin (lipopolysaccharide). Lower panel is taken at 3 h ond at 10 months after the injection when all peripheral signs of the infection had passed. These results are diagrammatic only. receptor activation together with infection leads to release of interleukin 1β (IL-1β) from microglia, and a decrease in glutamatergic receptors that competes with the increase due to tumour necrosis factor −α (TNF-α). Infection, mediated for instance by a Gram-negative bacterial component lipopolysaccharide (LPS), can trigger activation of toll-like (TOL) receptors on microglia and their autocatalytic release of TNF-α (large curved black arrow on the microglial cell). Large amounts of TNF-α, generated autocatalytically as well as by adenosine triphosphate (ATP) acting on the microglial P2X7 receptors, then diffuses from the synapse to activate microglia and their release of TNF-α at adjacent synapses (large vertical black arrow). Concomitant action of LPS, or an endogenous ligand for TOL receptors (biglycan), on synaptic microglia together with that of synaptic ATP on the microglia P2X7 receptors triggers the release of IL-1β from the microglia. TNF-α and IL-1β then compete to regulate the number of glutamatergic AMPA receptors in the post-synaptic process, either up or down. P2X7 receptors then play a central role in determining the efficacy of synaptic transmission, either directly through modulation of glutamate release or indirectly through modulation of the extent of AMPA receptors on which the glutamate acts.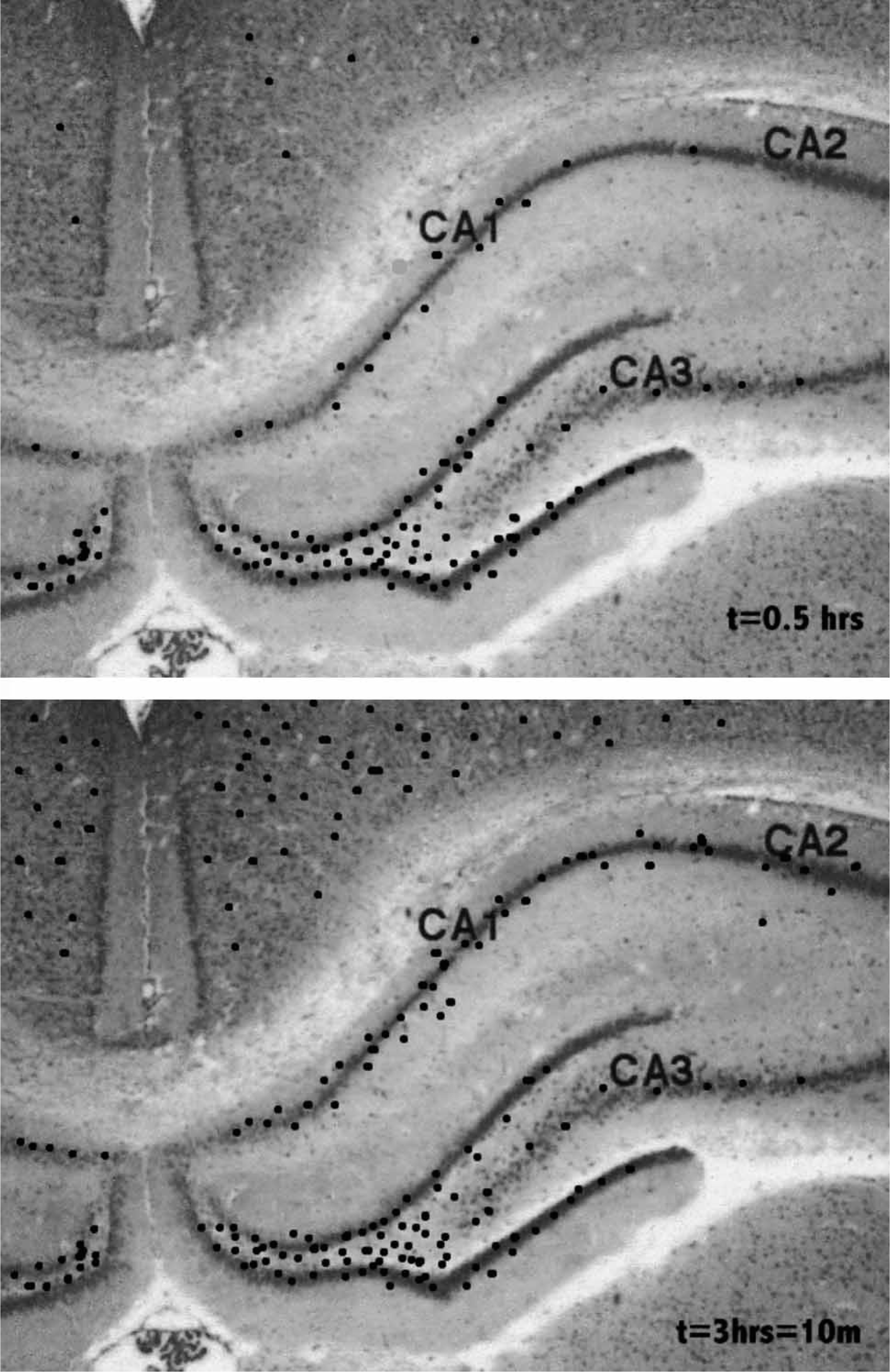
P2X7 receptors mediate genetic predisposition for depression
Recently, a polymorphism in P2X7 (rs2230912 resulting in a Gln640Arg single nucleotide polymorphism (SNP) has been found that is associated with bipolar-affective disorder [14], as well as major depressive illness [15]. This Gln460Arg polymorphism is located in the C-terminal domain of the P2X7 receptor, which is required for its normal function. What could be the relationship between altered P2X7 receptor function and depression? Identified polymorphisms in the P2X7 receptor of lymphocytes are known to produce a loss of function (the Glu-496b Ala SNP) or to alter trafficking of the receptor to the membrane surface, so decreasing its membrane expression (the Ile-568 to Asn SNP; [65]). The functional consequences for cytokine release of polymorphisms in the P2X7 receptor have been investigated for the case of 1513C polymorphism, which results in reduction in TNF-α release from LPS-stimulated leucocytes in the presence of ATP [66], as expected from the role of ATP on P2X7 receptors on microglia releasing TNF-α (Figure 4). The potential of a polymorphism in the P2X7 receptor to lead to altered synaptic function and therefore altered neural network performance is clear given the role of cytokines in regulating the efficacy of synaptic transmission in neural networks.
Major risk factor in psychotic disease: synaptic P2X7 receptors
Major depressive illness is associated with significant elevations in the density of microglia and in circulating levels of pro-inflammatory cytokines [67–73]. Given the role of P2X7 receptors in the release of cytokines it is perhaps not surprising that linkage studies have shown that the P2X7 gene is involved in both major depressive illness [15] and bipolar affective disorder [14]. The therapeutic exploitation of P2X7 receptors is now under way because of their potential role, not only in depressive illness, but in such disorders as Alzheimer's disease, spinal cord injury and sensory neuropathies [13], multiple sclerosis [74], as well as inflammatory/neuropathic pain [75].
Synaptic P2X7 regenerative loop hypothesis
This essay is prompted by the potential importance of the P2X7 receptor in depressive and other illnesses indicating the need for testable theories that link the function of this receptor to the neural networks in which it resides. The present hypothesis attempts to meet this need. It suggests that self-sustaining and interacting pools of ATP and TNF-α (as well as other cytokines) are important in determining the workings of neural networks. Specifically, in the absence of infection, synaptic glutamate has the potential to release sufficient ATP from astrocyte processes as to enhance the regenerative release of ATP, with this purine reaching levels that maintain presynaptic P2X7 receptors active, so fixing an elevated level of glutamate release, which in turn provides further ATP release in the glutamate-ATP loop. Polymorphisms in P2X7 receptors can lead to failure of this means of maintaining high levels of glutamate release. Second, if ATP acts on microglia P2X7 receptors and/or glutamate acts on the microglia they release TNF-α. This acts to maintain the level of AMPA receptors in the post-synaptic membrane for glutamatergic transmission. Polymorphisms in the P2X7 receptor can then indirectly lead to a loss of AMPA receptors in the post-synaptic membrane and hence a loss of synaptic efficacy. Finally, infective agents can activate synaptic microglia to release IL-1β if their P2X7 receptors are simultaneously activated by ATP. This cytokine then decreases AMPA receptors in the post-synaptic membrane. Polymorphisms in P2X7 can then enhance AMPA receptor numbers in the membrane. In summary, loss of P2X7 function can lead to a decrease in glutamate release and either a decrease or increase in AMPA receptors depending on the relative amounts of TNF-α and IL-1β at the synapse. Such changes, I speculate, are the basis for modifications in our abilities to express psychological capacities that are diagnosed as mood disorders.
