Abstract
The association between eating disorders (EDs) and affective disorders has been widely documented in the literature [1–4]. Major depressive disorder (MDD) and dysthymia are among the most commonly reported comorbid Axis I disorders in both individuals with bulimia nervosa (BN) and anorexia nervosa (AN) [5–8], with lifetime prevalences ranging from 20% to 83% [5, 9–12].
In individuals with EDs, comorbid affective disorders, most commonly identified as lifetime MDD, have been associated with diagnostic subtype [2, 6], greater substance and alcohol abuse [13], suicide attempts [12], and greater frequency of purging behaviours [14]. In contrast, among individuals with MDD, the prevalence of ED does not appear to be significantly elevated, with prevalences ranging from 4% to 10%, and does not differ significantly from the general population [15, 16]. In part, this could reflect the differential base rates of EDs and MDD, in that the likelihood of detecting a common disorder (MDD) among individuals with a less common disorder (EDs) is greater than that of detecting a less common disorder (ED) among individuals with a more common disorder (MDD).
Several studies suggest that eating and affective disorders may share common aetiological and phenomenological factors, such as temperament [17], haematological status [18], genetic factors [19], biochemical irregularities [20], low self-esteem [21], cognitive styles [22], eating symptoms [16], depressive symptoms [23], attachment style [24], childhood adversity [25], and psychosocial adjustment [26]. Previous family and twin studies have shown that relatives of individuals with EDs have a greater risk for depression than relatives of controls [1, 27]. However, whereas studies on AN showed that MDD is more likely to be transmitted by probands who are also depressed [28–30], studies on BN families indicated a cross-transmission of MDD and BN, showing elevated rates of MDD independent of the depression status of the proband [31–33]. Moreover, common familial aggregation has been described in both EDs and MDD [32].
In a study of juvenile twins, Silberg and Bulik explored the developmental association among symptoms of EDs, depression, overanxious disorder, and separation anxiety disorder during the course of development. They identified a common genetic factor influencing liability to symptoms of early and later overanxious disorder, depression, separation anxiety disorder, and eating symptoms as well as a shared environmental factor specifically influencing early depression and early ED symptoms [34]. This study indicates that there may be both shared genetic and shared environmental factors that influence risk to both EDs and depression.
Despite considerable research, the nature of the relation between MDD and EDs remains only partially understood. Heterogeneity of causal sequences is likely, because depressive symptoms may arise as a negative consequence of disordered eating and negative energy balance resulting from semistarvation [35], whereas onset of MDD before EDs [8, 10, 12, 36] implies that the latter can be secondary to affect disturbance [37]. In support of this assertion, negative affect and temperamental emotionality have been found to predict future onset of bulimic pathology [38], and premorbid MDD has been noted as a prospective risk factor for the onset of EDs [39, 40].
Few studies to date have assessed carefully the pattern of onset of EDs and MDD. Wildman
The objectives of the present study were (i) to investigate the temporal relation between EDs and MDD and to determine differences in the nature of depressive symptoms relative to the presence or absence of premorbid MDD; (ii) to determine whether pattern of onset differs by ED diagnostic subgroups; and (iii) to determine whether MDD prior to EDs is associated with greater suicidal attempts or ideation.
Methods
Participants
A final sample of 1371 individuals with EDs participated in the current study (AN restricting subtype, RAN, n = 375; AN purging subtype, PAN, n = 205; AN bingeing subtype, BAN, n = 129; BN purging subtype, PBN, n = 240; BN non-purging subtype, NPBN, n = 19; lifetime history of both AN and BN, ANBN, n = 342; and ED not otherwise specified, EDNOS, n = 61). Participants were from two of the multisite international Price Foundation Genetic Studies of Eating Disorders: BN Affected Relative Pair Study (BN ARP), and AN Trios. These studies were designed to identify susceptibility loci involved in risk for EDs. Participants were recruited through advertising for affected relative pairs and EDs treatment centers. Informed consent was obtained from all study participants, and all sites received approval from their local Institutional Review Board. Brief descriptions of each study are provided below. Full details on the BN study are available elsewhere [41].
BN Affected Relative Pair Study
The sample for this study included both probands and affected relatives. Probands met the following criteria: (i) modified DSM-IV [42] lifetime diagnosis of BN, purging type (purging must have included regular vomiting (with other means of purging allowed), and binging and vomiting must have occurred at least twice a week for a duration of at least 6 months); and (ii) age between 13 and 65 years. Affected relatives were biological family members who (i) were between the ages of 13 and 65 years; and (ii) had lifetime ED diagnoses of DSM-IV BN, purging type or non-purging type, modified DSM-IV AN (i.e. criterion D not required), or EDNOS (subthreshold AN, subthreshold BN, binge ED, or purging disorder). For the complete list of inclusion and exclusion criteria for probands and relatives see Kaye
AN Trios Study
The sample for this study included individuals with AN and their parents. Probands were required to meet the following criteria: (i) modified DSM-IV [42] lifetime diagnosis of AN, with or without amenorrhea; (ii) low weight that is/was less than 5th percentile of body mass index (BMI) for age and gender on the Hebebrand chart [43] of National Health and Nutrition Examination Survey (NHANES) epidemiological sample; (iii) onset prior to age 25; (iv) weight that is/was controlled through restricting and/or purging, which includes vomiting, use of laxatives, diuretics, enemas, suppositories, or ipecac; (v) age between 13 and 65; (vi) Caucasian ethnicity (one grandparent from another racial group is acceptable); and (vii) study diagnostic criteria met at least 3 years prior to study entry. Potential participants were excluded if they reported maximum BMI since puberty >27 kg/m2 for females and >27.8 kg/m2 for males.
The present study included participants from the BN ARP study and the AN Trios study with a diagnosis of EDs. For the present analysis, from a initial sample of 1488, the following individuals were excluded: (i) male (n = 28), because the number of male subjects with these diagnoses was too small for meaningful comparison; (ii) participants with either a DSM IV diagnosis of bipolar I or bipolar II (n = 46); and, (iii) individuals who were missing information to enable an MDD diagnosis (n = 43). For this third criterion, individuals were excluded if they did not answer or had missing values for any items in Module A of the Structured Clinical Interview for DSM-IV Axis I Disorders (SCID-I) that prohibited making either a negative or positive diagnosis [44]. The remaining 1371 ED participants were included in the analysis.
For the analyses comparing individuals with MDD prior to EDs to those with EDs first, participants were required to have a DSM IV diagnosis of MDD (the appetite symptom was excluded from the criteria) and to have data for both age of onset of MDD and of EDs (n = 963).
Measures and interviewers
Demographic and clinical variables
Data relative to age of onset and duration of both MDD and EDs were included in the analyses.
Eating disorder diagnoses
Lifetime histories of EDs in probands and affected relatives were assessed with the Structured Inventory of Anorexia Nervosa and Bulimic Syndromes (SIAB) [45]. Additional information regarding ED recovery status as well as the presence or absence of ED symptoms was obtained by an expanded version of Module H of the Structured Clinical Interview for DSM-IV Axis I Disorders (SCID-I) [44].
Major depression diagnosis and depressive symptoms
Lifetime major depression and level of depressive symptoms were assessed with the SCID-I [44].
Statistical analyses
Statistical analyses were conducted using the GENMOD procedure of SAS version 8.1 (Statistical Analysis System, Cary, NC, USA). We first explored rates of MDD across diagnostic subtypes, and compared age of onset of MDD and EDs. The sample was divided into two groups: those with MDD onset before the development of the ED, and those with MDD onset after or during the same year as the ED onset. Frequency of endorsement of depressive symptoms was compared in the two groups using logistic regression analysis with corrections for non-independence using generalized estimating equations (GEE) [46–48]. All significance tests were two-tailed (p < 0.05). Age at interview was entered into all models as a covariate. Because ED subtype was not significantly associated with order of onset of MDD (χ2=1.98, df = 6, p = 0.92), ED subtype was not used as a covariate in other analyses on order of onset. P-values were adjusted for multiple testing using the method of false discovery rate (FDR) [49].
Results
Prevalence of MDD and ED subtypes
In the 1371 individuals, the observed lifetime prevalence of MDD was 72.9%. Table 1 presents the prevalence of MDD by ED subtype. Logistic regression with GEE corrections and post-hoc pairwise comparisons indicated that MDD was significantly related to ED subtype (χ2=39.72, df = 6, p = 0.0001) and age (χ2=5.02, df = 1, p = 0.025). The post-hoc tests showed that the RAN group had significantly fewer individuals with MDD than PAN, BAN, PBN, NPBN, and ANBN. The NOS group also differed from the PAN, PBN, NPBN, and ANBN groups, but did not differ from the RAN and BAN groups.
Lifetime prevalence of MDD by eating disorder subtype
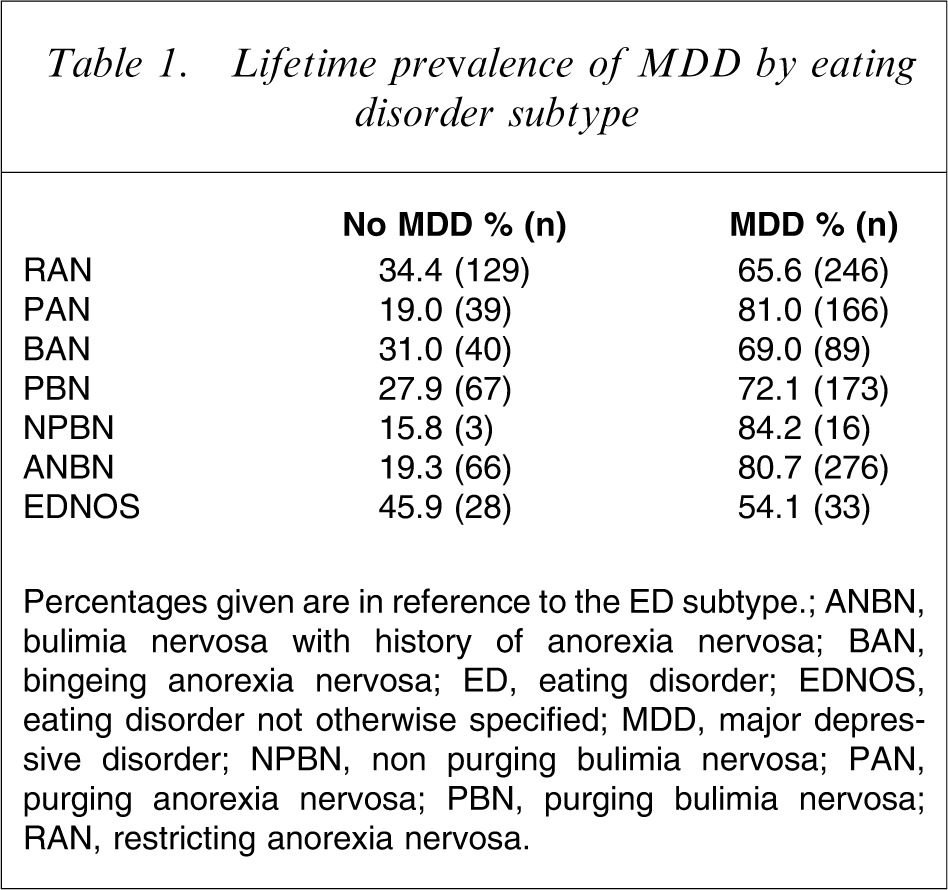
Percentages given are in reference to the ED subtype.
ANBN, bulimia nervosa with history of anorexia nervosa; BAN, bingeing anorexia nervosa; ED, eating disorder; EDNOS, eating disorder not otherwise specified; MDD, major depressive disorder; NPBN, non purging bulimia nervosa; PAN, purging anorexia nervosa; PBN, purging bulimia nervosa; RAN, restricting anorexia nervosa.
Onset of MDD by ED subtypes
Among those with lifetime MDD (n = 963), we explored the order of onset of EDs and MDD. As shown in Table 2, after applying logistic regressions with GEE corrections, order of onset was not significantly associated with ED subtype (χ2=1.98, df = 6, p = 0.92), nor with age (χ2=3.75, df = 1 p = 0.05). Of the individuals with lifetime MDD, 34.5% reported MDD onset before the onset of their ED.
Frequency of individuals who had lifetime MDD first and eating disorders first by eating disorder subtype
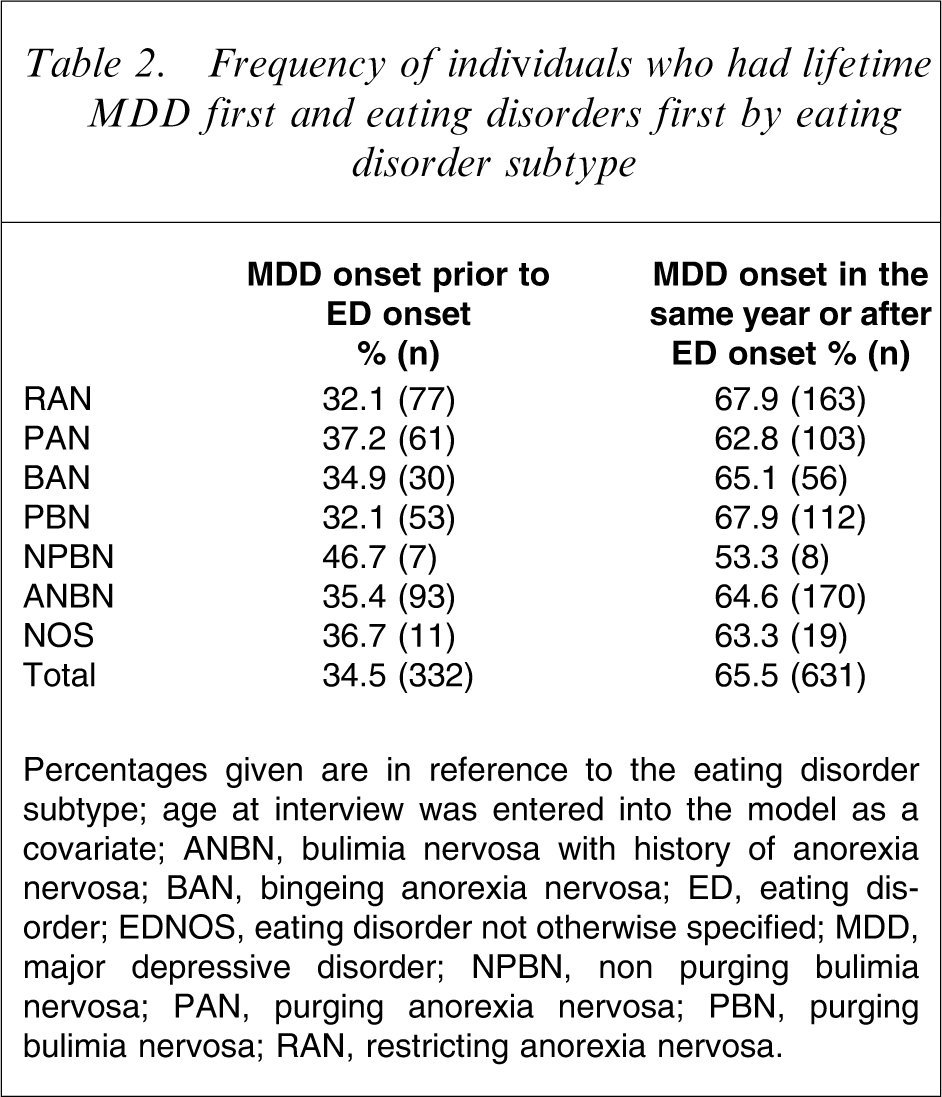
Percentages given are in reference to the eating disorder subtype; age at interview was entered into the model as a covariate.
ANBN, bulimia nervosa with history of anorexia nervosa; BAN, bingeing anorexia nervosa; ED, eating disorder; EDNOS, eating disorder not otherwise specified; MDD, major depressive disorder; NPBN, non purging bulimia nervosa; PAN, purging anorexia nervosa; PBN, purging bulimia nervosa; RAN, restricting anorexia nervosa.
Onset of MDD and depressive symptoms
We then explored whether depressive symptoms differed in those individuals who reported MDD onset before versus after ED onset (Table 3). We found significant differences between the groups on psychomotor agitation (OR = 1.53; 95%CI = 1.14–2.06) and thoughts of own death (OR = 1.73; 95%CI = 1.31–2.30). The MDD onset-first group reported greater psychomotor agitation and thoughts of own death (but not suicide attempts or ideation). As expected, the MDD-first group reported a longer mean duration of MDD (χ2=182.7, p < 0.001).
Frequency of endorsement of the depressive symptoms by order of onset
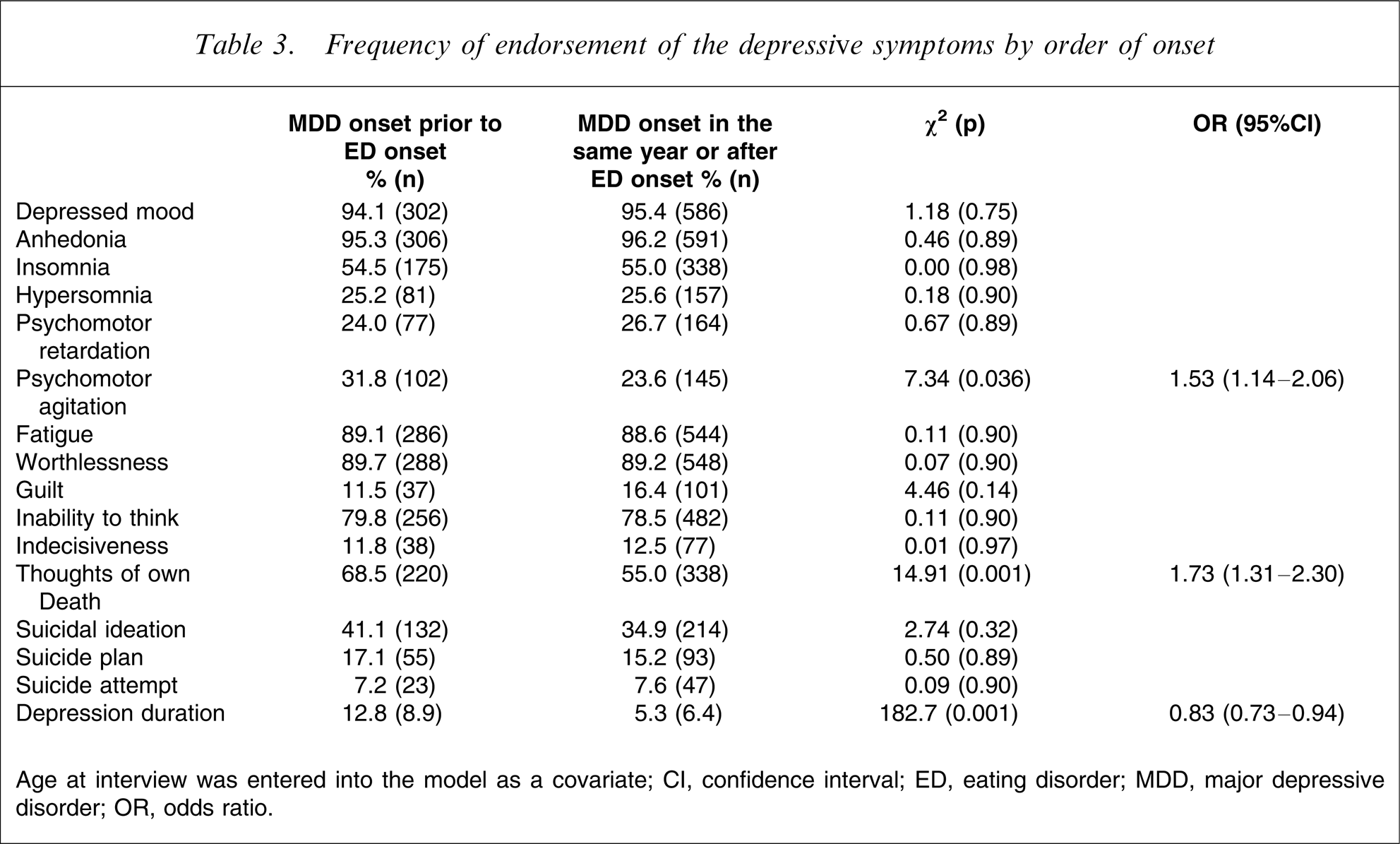
Age at interview was entered into the model as a covariate
CI, confidence interval; ED, eating disorder; MDD, major depressive disorder; OR, odds ratio.
Temporal proximity between MDD and ED diagnosis
As shown in Figure 1, attempting to identify the temporal proximity between the onset of MDD and the onset of EDs, we calculated the difference between these two ages of onset (age of onset of MDD–age of onset of ED). As calculated, negative values indicated those instances in which MDD came first. Figure 1 shows that among all individuals who had MDD before the ED (34.5% from the total ED sample who had a lifetime MDD), 26.5% had the MDD onset during the year previous to the onset of the ED. More than two-thirds (67%) of individuals experienced the onset of both disorders within the same 3 year window.
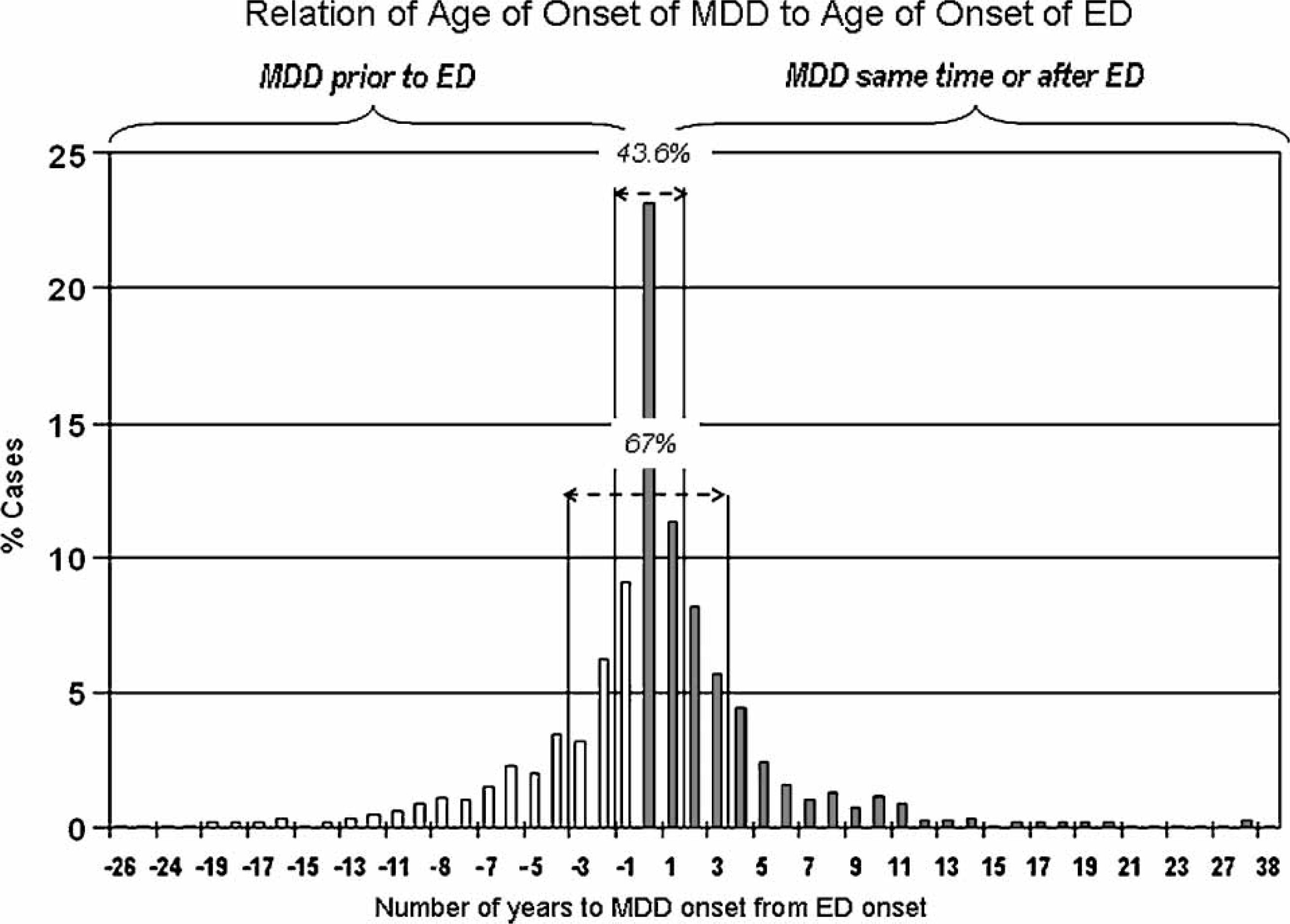
Relation of age of onset of MDD to age of onset of eating disorders. ED, eating disorder; MDD, major depressive disorder. Negative numbers indicate that MDD came first MDD occurred prior to ED; ▪ MDD occurred at the same time or after ED
Discussion
This study examined the prevalence of MDD across ED subtypes, the chronology of onset of both classes of disorders, and whether specific symptomatic and clinical factors distinguish individuals with EDs who experienced depression prior to their EDs from individuals who develop depression after an ED.
Confirming previous studies, we observed a high lifetime prevalence of MDD among individuals with EDs (72.9%) [6, 11–13]. Although previous research has shown that dysthmia may be more strongly associated EDs than MDD among adolescents [8, 50], we were not able to investigate this relationship in the present study, because dysthmia diagnoses were not available for all of the study samples.
Extending previous investigations, we observed statistically significant differences in the prevalence of lifetime MDD across diagnostic subgroups. Individuals with ED subtypes that included either binging and/or purging (PAN, BAN, PBN, NPBN, ANBN) reported greater lifetime MDD than individuals with the restricting subtype (RAN), consistent with the few existing studies, in which individuals with PBN were significantly more likely than those with RAN to be diagnosed with lifetime history of any mood disorder [2, 6]. Contradictory to a recent study [51], individuals with EDNOS reported the lowest lifetime MDD rate. A second main finding in the present study was that 34.5% of ED patients presented with MDD onset prior to ED onset. The remainder reported either contemporaneous onset or MDD after the ED had emerged. This result concurred with previous studies [12, 36]. We were particularly interested in whether the nature of the MDD differed in those individuals who experienced depression first. Indeed, those with MDD first reported longer duration of MDD, more frequent psychomotor agitation, and greater thoughts of their own death. In contrast to Wildman
In an analysis of the temporal pattern of onset of MDD and ED onset, we found that 43.6% of the onsets occurred within 1 year of each other and 67% within a 3 year window. There are several possible interpretations of this observation. First, it is possible that trigger events (i.e. puberty, trauma, life transitions) may activate processes that lead to the expression of both MDD and EDs in individuals who are genetically vulnerable to both disorders. Second, in instances where MDD emerges first, factors associated with MDD such as loss of appetite or low self-esteem may themselves activate underlying genetically mediated vulnerability to EDs. Third, in those who develop EDs first, aspects of the ED (e.g. starvation) may serve as triggers for an underlying latent vulnerability to MDD.
Although much more sophisticated study designs are required to clarify which of these interpretations is most accurate, nonetheless, the clinical implications of the findings are clear. The period of time bracketing the onset of an eating or affective disorder remains a high-risk time for developing additional psychopathology in at-risk individuals. Clinicians treating individuals with new-onset EDs or MDD should remain vigilant for the emergence of additional psychopathology, especially during the initial 3 year window following the onset of the first disorder. Careful attention should be paid to the emergence of mood symptoms in individuals with new-onset EDs. Similarly, individuals with new-onset MDD should also be screened for the onset of ED symptoms. Although this may not be universal, it may especially apply to those individuals in whom there is some indication of increased risk for EDs (e.g. family history of EDs).
The results of this study should be interpreted within the context of several limitations. First, given the nature of the SCID and SIAB, EDs and MDD onsets were assessed independently. We did not use a structured interview such as the Longitudinal Interval Follow-up Evaluation [52], which allows one to gauge more finely the relative onsets of various psychiatric disorders. Second, our samples included families that are enriched for EDs recruited through advertising and ED treatment centers. This may lead to higher prevalences of comorbidity and limit the generalizability of the present findings. Arguing against this, however, is the fact that the present comorbidity rates are on par with other clinical samples of individuals with EDs [11, 12], but somewhat higher than community samples of individuals with EDs [53]. Third, the small sample size of some groups (e.g. NPBN and NOS) did not allow a complete explication of patterns of onset in these groups. Finally, the odds ratios reported were modest, and by nature of our design we were forced to rely on retrospective self-report data, and recall biases may have been operative, although one can only speculate as to the direction of the recall bias. The onset of one psychiatric disorder may heighten awareness of the development of additional disorders. Alternatively, similar underlying mechanisms may trigger the onset of several disorders, which could account for the relatively narrow window of emergence of both EDs and MDD. Assessment of personality disorders in addition to Axis I disorders would have enabled a more comprehensive evaluation of the relation of mood instability and ED behaviours.
Further prospective studies should include more granular explorations of the temporal emergence of symptoms of MDD and EDs (i.e. not just full-scale diagnoses) in order to explain more fully the manner in which the emergence of some symptoms of one disorder influence the emergence of symptoms of the other.
Footnotes
Acknowledgements
The authors wish to thank the Price Foundation for the support of the clinical collection of participants and support of data analysis. The authors acknowledge the staff of the Price Foundation Collaborative Group for their efforts in participant screening and clinical assessments. The authors are indebted to the participating families for their contribution of time and effort in support of this study. This work was financially supported by the Price Foundation of Geneva, Switzerland. Andrea Poyastro Pinheiro received financial support from Conselho Nacional de Desenvolvimento Cientifico e Tecnologico (201093–2004/9) CNPQ–Brazil. This study was supported by grants MH-66117 from the National Institutes of Health, Bethesda, MD, USA
