Abstract
Articles on treatment
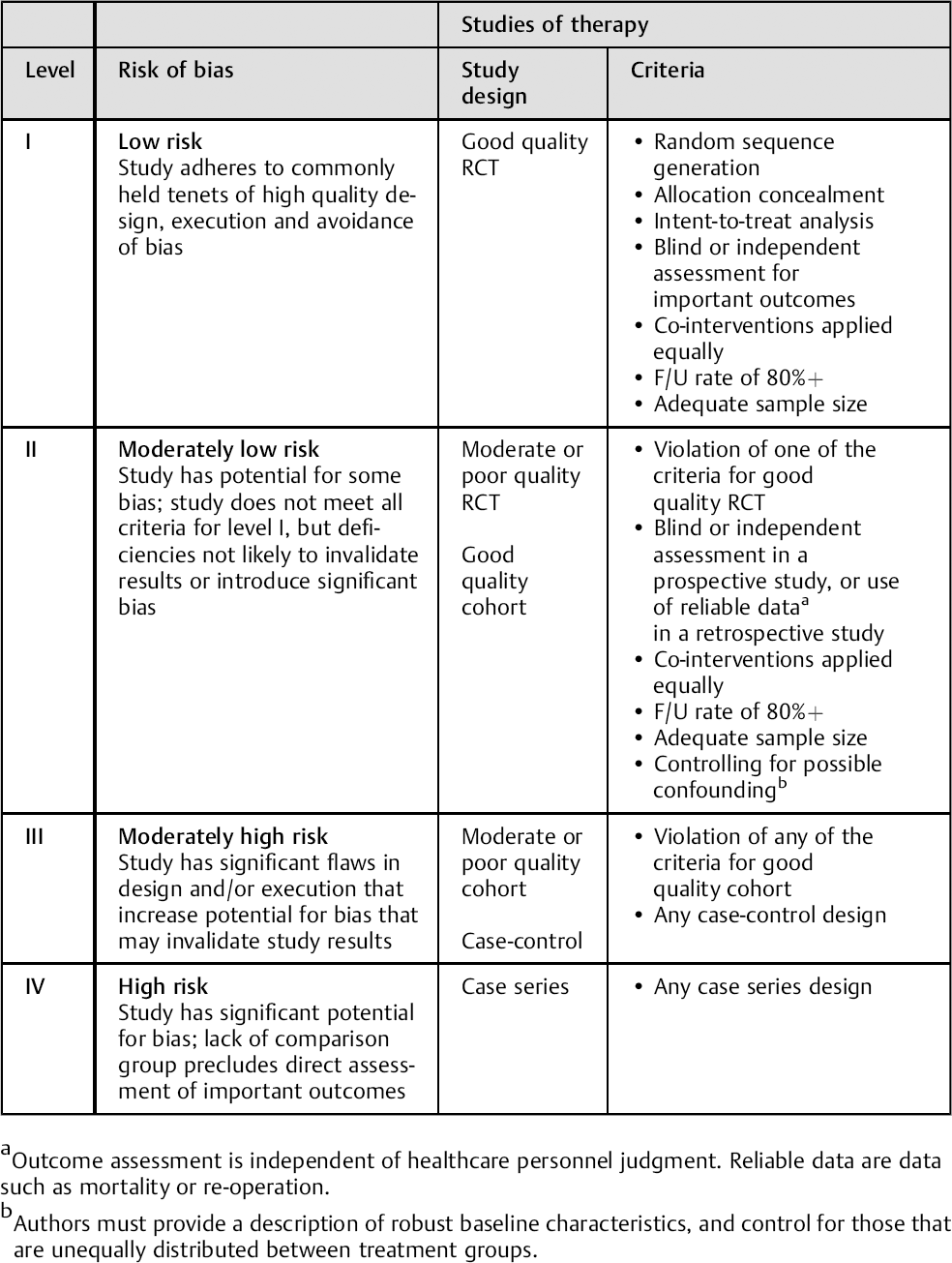
Outcome assessment is independent of healthcare personnel judgment. Reliable data are data such as mortality or re-operation.
Authors must provide a description of robust baseline characteristics, and control for those that are unequally distributed between treatment groups.
Determination of Overall Strength of Evidence (SoE)
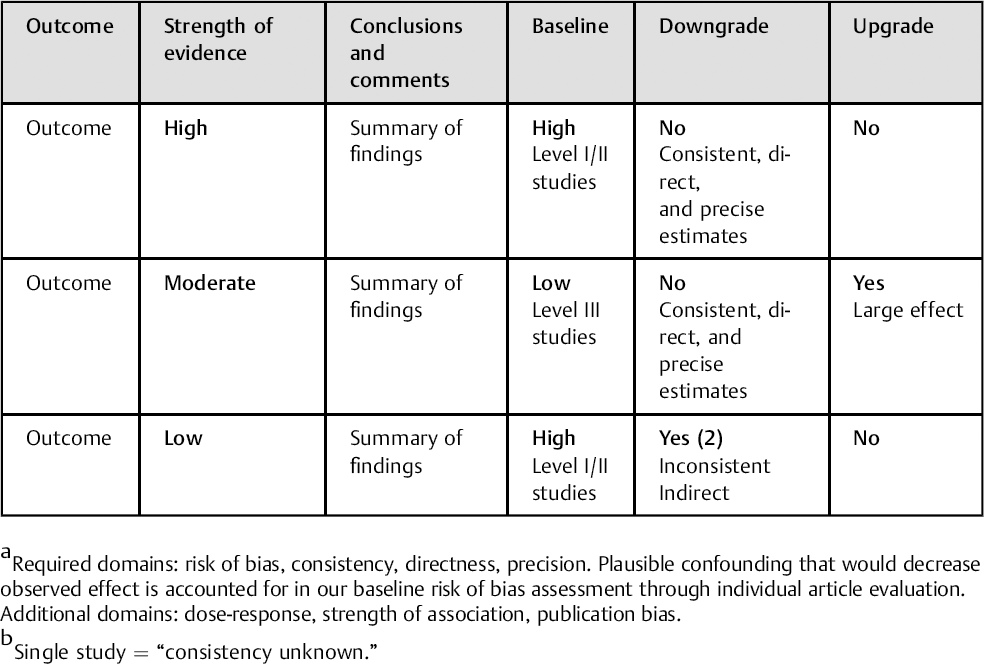
After individual article evaluation, the overall body of evidence with respect to each outcome is determined based on precepts outlined by the Grades of Recommendation Assessment, Development and Evaluation (GRADE) Working Group and recommendations made by the Agency for Healthcare Research and Quality (AHRQ). Qualitative analysis is performed considering the AHRQ required and additional domains. The table below provides an outline of the method used to determine the final SoE.
Strength of Evidence for Existing Systematic Reviews
Level of evidence ratings for Cochrane reviews and other systematic reviews are assigned a baseline score of HIGH if RCTs were used, LOW if observational studies were used. The rating can be upgraded or downgraded based on adherence to the core criteria for methods, qualitative, and quantitative analyses for systematic reviews (there is a reference/evaluation table for this).
The following four possible levels and their definition are reported:
All AHRQ “required” and “additional” domains a are assessed. Only those that influence the baseline grade are listed in table.
Required domains: risk of bias, consistency, directness, precision. Plausible confounding that would decrease observed effect is accounted for in our baseline risk of bias assessment through individual article evaluation. Additional domains: dose-response, strength of association, publication bias.
Baseline strength: Risk of bias (including control of confounding) is accounted for in the individual article evaluations. High = majority of articles level I/II; low = majority of articles level III/IV
Downgrade: Inconsistency b of results (1 or 2); Indirectness of evidence (1 or 2); Imprecision of effect estimates (1 or 2); Sub-group analyses not stated apriori and no test for interaction (2)
Single study = “consistency unknown.”
Upgrade: Large magnitude of effect (1 or 2); Dose response gradient (1)
Articles on prognosis or risk
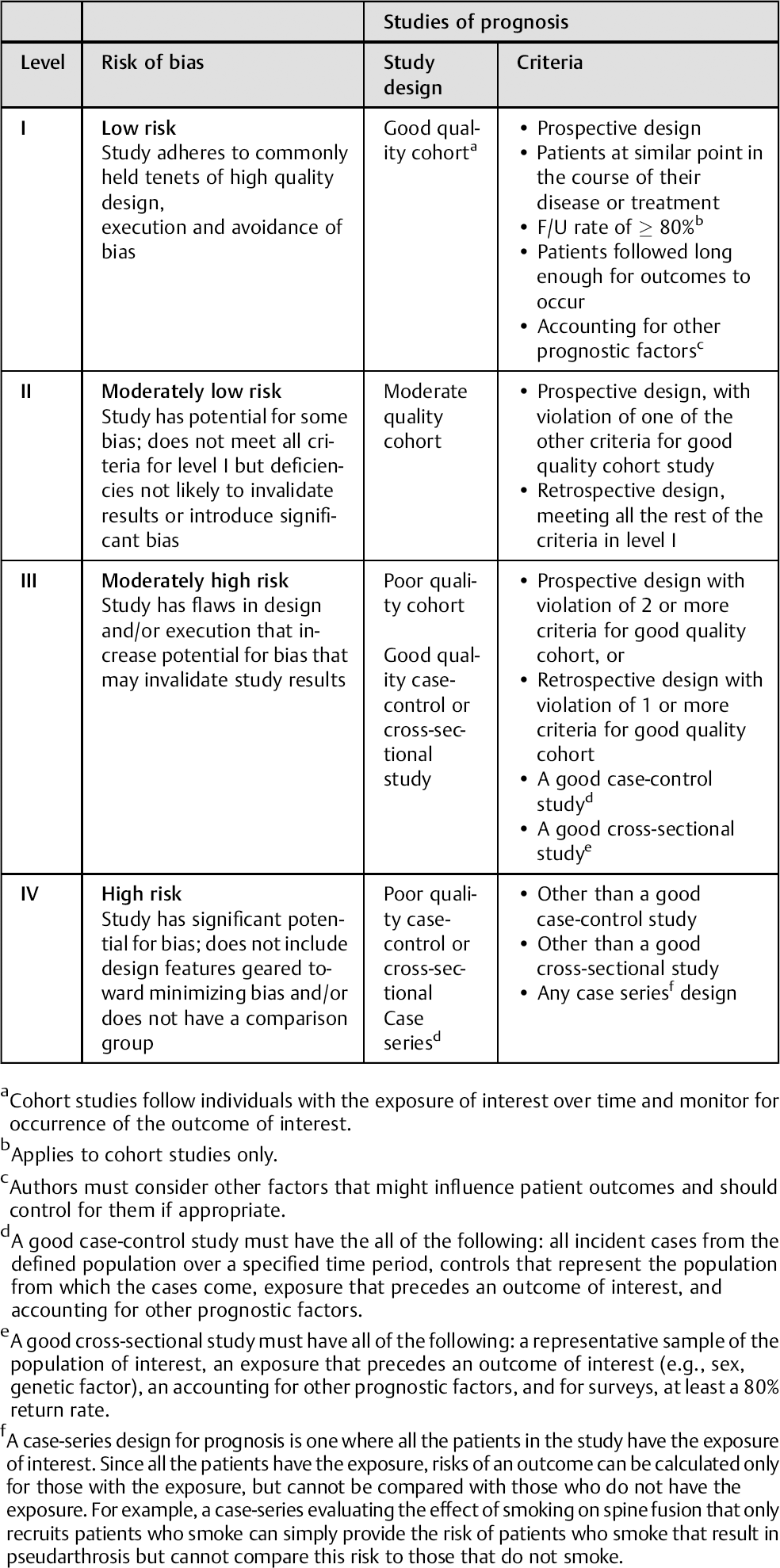
Cohort studies follow individuals with the exposure of interest over time and monitor for occurrence of the outcome of interest.
Applies to cohort studies only.
Authors must consider other factors that might influence patient outcomes and should control for them if appropriate.
A good case-control study must have the all of the following: all incident cases from the defined population over a specified time period, controls that represent the population from which the cases come, exposure that precedes an outcome of interest, and accounting for other prognostic factors.
A good cross-sectional study must have all of the following: a representative sample of the population of interest, an exposure that precedes an outcome of interest (e.g., sex, genetic factor), an accounting for other prognostic factors, and for surveys, at least a 80% return rate.
A case-series design for prognosis is one where all the patients in the study have the exposure of interest. Since all the patients have the exposure, risks of an outcome can be calculated only for those with the exposure, but cannot be compared with those who do not have the exposure. For example, a case-series evaluating the effect of smoking on spine fusion that only recruits patients who smoke can simply provide the risk of patients who smoke that result in pseudarthrosis but cannot compare this risk to those that do not smoke.
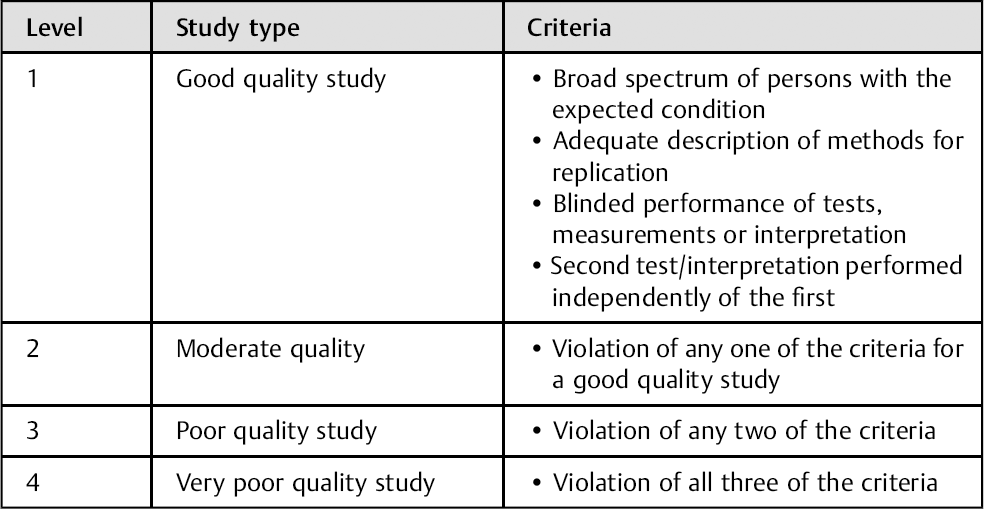
Definitions of the Different Levels of Evidence for Reliability Studies