Abstract
Study Design
Case report.
Objective
We report the youngest child diagnosed with upper cervical osteoblastoma and the first case operated on with our novel surgical approach.
Methods
Our patient underwent a two-stage surgery. During the first operation via a posterior approach, a subtotal resection of a C2 bony mass was performed. C3 was also subtotally resected due to tumor extension. Posterior fixation of C1–C5 was performed by C1 sublaminar hooks and C4 and C5 lateral mass screws. Ten days later, a total resection of the residual bony mass was performed through an anterior approach (between the sternocleidomastoid muscle and carotid sheath). Reconstruction of C1–C3 was performed with C1 anterior sublaminar wiring and an expandable titanium cage.
Results
Successful reconstruction of C2–C3 vertebral bodies was achieved. At 2-year follow-up, the child was symptom-free. Imaging studies revealed no recurrence of tumor or instability.
Conclusion
A novel technique for reconstruction of C2–C3 vertebral bodies is demonstrated for the youngest case (a 6-year-old boy) of osteoblastoma in the literature. We recommend this approach for cervical spine reconstruction in patients who have an intact C1 arc and resected lower bodies.
Introduction
Osteoblastomas are rare benign tumors. 1 , 2 Spine osteoblastoma has been diagnosed in children as young as 2 years of age. 3 To our knowledge, the youngest case of cervical spine osteoblastoma has been documented in a 9-year-old child. 4
Considering the rarity of pediatric cervical osteoblastomas 5 and the complexity between different structures present in the cervical region that becomes even more challenging in pediatric age groups, there are still questions to be answered regarding surgical approaches to resect these lesions in the pediatric population. 6 Anterior reconstruction of the upper cervical spine is a complex problem in this population.
To the best of our knowledge, we report the youngest child with upper cervical osteoblastoma reported in the literature so far. He is the first case operated on with our novel surgical technique.
Case Report
History
A 6-year-old boy was referred to our department by a neurosurgeon for a newly developed neck skew and aggravation of neck pain that had been bothering the patient for 2 years. Prior to his visit at our department, he was under a 2-year follow-up by several pediatricians for neck pain and pain on neck motion. Magnetic resonance imaging (MRI) was obtained at that time but did not show a significant finding (Fig. 1A, B). When we visited the patient, in addition to neck skew and pain, he complained of difficulty swallowing. Neck pain worsened at night with no significant response to aspirin. His past medical history and family history for malignancy was unremarkable.
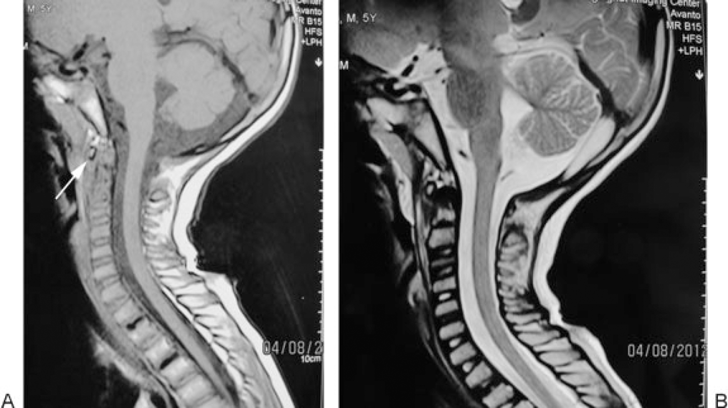
(A) Cervical magnetic resonance imaging (MRI) (T1-weighted): 10 months before the operation. Arrow points to the minimal structural and intensity change in odontoid process. (B) Cervical MRI (T2-weighted): 10 months before the operation.
Laboratory Workups
Laboratory workups did not show any abnormal finding.
Physical Examination
On physical examination, the patient had a severe limitation of neck range of motion. Loss of coordination of extremities and signs of myelopathy were detected. Local tenderness was not present. Movement of the neck produced pain. The right upper extremity muscle power (according to Medical Research Council Classification) evaluated by manual testing was 4/5 (the distal muscles were weaker than proximal ones). Lower extremity muscle power was intact. Upper and lower extremities deep tendon reflexes were 3/4. Pinprick test of the right C3–C5 dermatomes was impaired (paresthesia). The Hoffman test was positive. The patient's sense of vibration and position were intact. The rectal muscles were not affected. Impaired tandem gait, loss of dexterity, and gait imbalance were observed.
Imaging Studies
Chest X-ray was unremarkable. Cervical X-ray (Fig. 2) revealed an expansile lesion that eroded the vertebral body and posterior elements of C2–C3. Spiral axial computed tomography (CT) scan of the cervical column (Fig. 3A, B) depicted a large 36 × 33 × 27-mm calcified mass of C2 that caused deformity of C2 and also affected C3 with severe pressure erosion of the odontoid process. Secondary spinal canal narrowing was also noted (anteroposterior canal diameter: 8 mm). MRI (Fig. 4A, B) revealed an expansile, well-demarcated, inhomogeneous (iso- and hypointense in T1-weighted images and hypo- and hyperintense in T2-weighted images) mass that extended from the clivus to C4. Gadolinium-enhanced images (Fig. 4C) depicted destruction of the C2 and C3 vertebra (the odontoid process, right facet joints, and both vertebral bodies). Axial MRIs displayed encircling of right vertebral artery (Fig. 5).
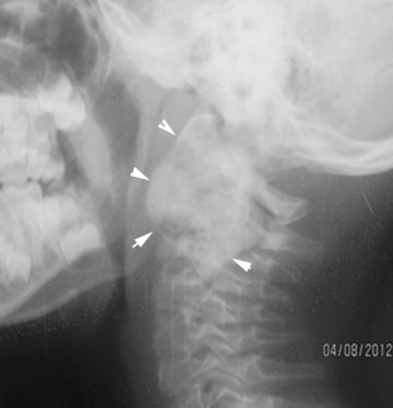
Cervical X-ray: Arrows show the erosion of vertebral body and posterior elements of C2–C3 by the expansile mass.
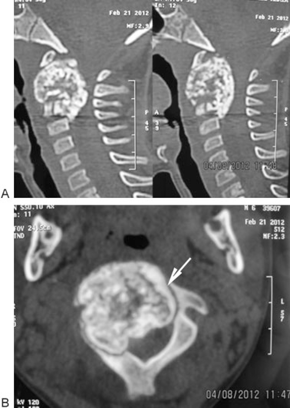
(A) Cervical computed tomography (CT), sagittal reformat: calcified mass of C2 that also affected C3. (B) Cervical CT, axial view: arrow points to the well demarcated calcified lesion involving body and right side posterior element of C2.
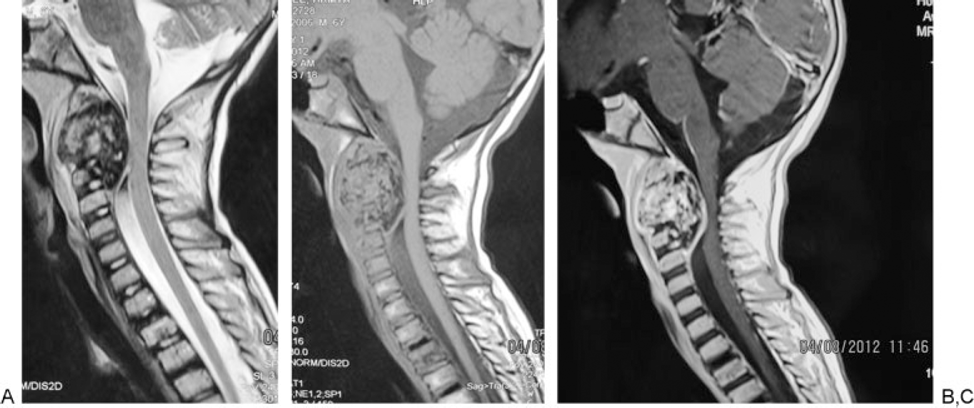
(A) Cervical magnetic resonance imaging (MRI; T2-weighted): inhomogeneous mass extending from clivus to C4. (B) Cervical MRI (T1-weighted). (C) Cervical MRI (gadolinium-enhanced): enhancing mass destructed C2 and C3 vertebra.
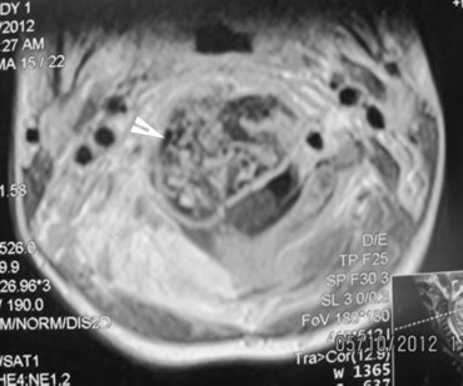
Cervical magnetic resonance imaging (axial images): arrow shows the right vertebral artery that is encompassed by the mass.
Multidetector CT angiogram of the neck and skull base (Fig. 6) showed right vertebral artery lateral displacement by a huge C2 lobulated mass that was abutting the right vertebral artery. Whole-body bone scan by Tc-99m-methylene diphosphonate was also performed, which showed an abnormally increased uptake of radioactive uptake in C2. Results of CT-guided C2 needle biopsy were highly suggestive of osteoblastoma.
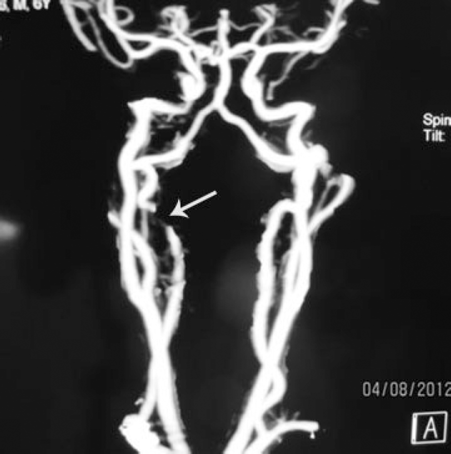
Multidetector computed tomography angiogram of neck and skull base: arrow shows right C2 segment vertebral artery occlusion.
Operation
The patient underwent a two-stage surgery. During the first operation via a posterior approach, a subtotal resection of the C2 bony mass was performed. C3 was also subtotally resected due to tumor extension. The posterior fixation of C1–C5 was performed by C1 sublaminar hooks and C4 and C5 lateral mass screws. Intraoperative images are shown in Fig. 7. Fig. 8A, B shows X-ray and CT after the first operation. After 10 days, total resection of residual bony mass was performed through an anterior approach (between the sternocleidomastoid muscle and carotid sheath; Fig. 9A, B). The vertebral artery was obliterated. Due to a thin C1 arch and the absence of good bony purchase for screws, we used anterior sublaminar wiring for C1 and reconstructed the C2–C3 bodies with an expandable cage. The lower end of the implant was screwed to C4. Fig. 10 displays the X-ray after the second operation.
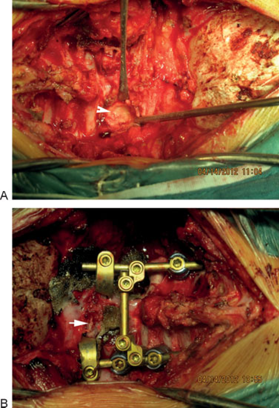
(A) First operation: arrow points to the bony texture of posterior part of the tumor that required drilling. (B) First operation: final posterior fixation. Arrow shows posterior C1 arc.
(A) First operation, postoperative plain X-rays. (B) First operation, postoperative computed tomography scan. Arrow points to the remnant of the tumor in left part of spinal canal.
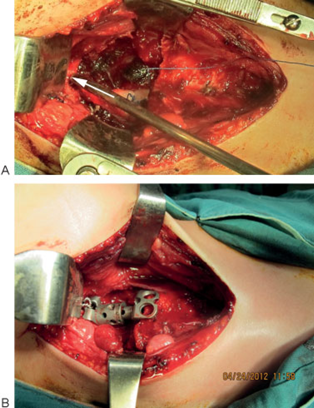
(A) Second operation: arrow shows anterior arc of C1. (B) Second operation: implant fixed to C1 with sublaminar wiring technique.
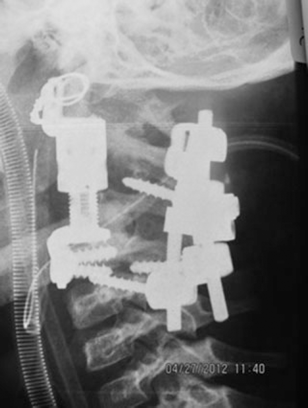
Second operation: postoperative X-ray.
On gross examination, the pinkish gray mass had a mixed consistency with soft and hard parts with no septations.
Pathologic Examination
The pathologic examination (Fig. 11) showed osteoblasts suggestive of osteoblastoma.
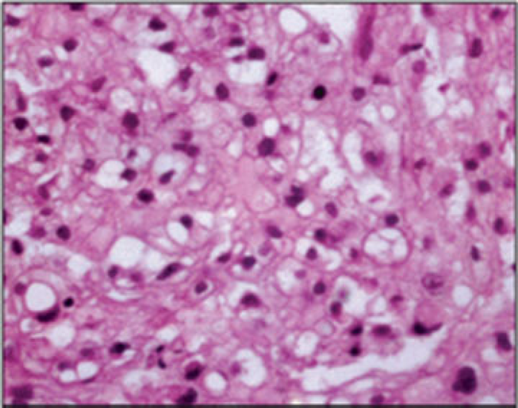
Excised mass hematoxylin and eosin histopathology.
Follow-up
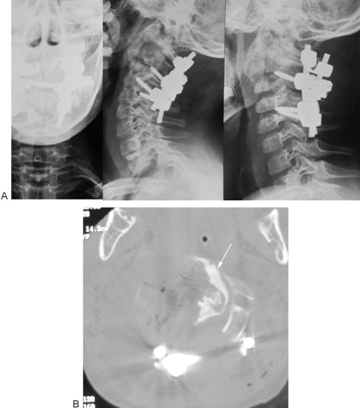
Fig. 12A, B, C demonstrates plain X-rays. Fig. 12D shows MRI taken at 2-year follow-up. No tumor recurrence or instability is noted.
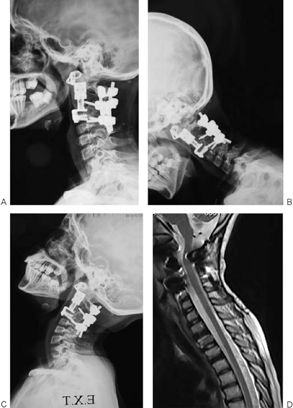
(A) Thirty-month follow-up plain X-ray. (B) Thirty-month follow-up plain X-ray (flexion view). (C) Thirty-month follow-up plain X-ray (extension view). (D) Two-year follow-up magnetic resonance imaging.
Discussion
Osteoblastomas are rare benign tumors, 1 , 2 which represent 3 to 5% of all benign bone tumors. 5 , 7 They may behave in a locally aggressive manner. 8 , 9 , 10 , 11 , 12 , 13 The spine is involved in ∼40 to 50% of cases. 14 , 15 The incidence of cervical spine osteoblastomas has been reported as 9 to 40%. 10 , 12 , 14 , 16 In the pediatric population, ∼5 to 10% of central nervous system tumors are primary spinal tumors. 17 Primary spinal tumors with a benign nature are more common than malignant ones in children and adolescents. 5 Osteoblastomas account for 5% of benign bone tumors that affect the vertebral column of children and adolescents. 5 An age range of 6 months to 75 years has been reported for 90% of osteoblastoma occurrences. 18 Spine osteoblastoma has been diagnosed in children as young as 2 years of age. 3 To our knowledge, the youngest occurrence for cervical spine osteoblastoma has been documented in a 9-year-old child. 4 Presenting symptoms can be neck pain and stiffness, torticollis, scoliosis, gait problems, and even spastic quadriparesis. 9 , 12 , 14 , 16 , 19 , 20 , 21 Nonspecific symptoms on presentation coupled with the rarity of this tumor in the pediatric age group can lead to a delay in diagnosis. 22 , 23 , 24 , 25 , 26
The chance of malignant transformation has been reported 5 , 8 , 11 , 12 that ranged from 12 to 25%. 27 As illustrated in imaging studies of our case obtained 10 months prior to admission, the mass significantly increased in size. Rapid growth over months is described for aggressive osteoblastomas. 28 Pochaczevsky et al reported significant growth of an osteoblastoma over 8 months. 29
To achieve relief of symptoms, stop the aggressive behavior that may destruct neighboring vital neurovascular structures, and prevent the chance of malignant transformation, surgery is the treatment of choice. The type of surgical approach for resection of spinal tumors is selected based on a host of factors that include the type of tumor and its location within the spinal column, the presence or absence of neural compression and the portion of spinal cord involved, 30 patients’ individual characteristics, the stability of the spine, 31 and suspicion for malignancy. 11
Considering the rarity of pediatric cervical osteoblastomas 5 and the complexity between different structures present in the cervical region that becomes even more challenging in pediatric age groups, there are still questions to be answered regarding the surgical approaches to resect these lesions in the pediatric population. 6
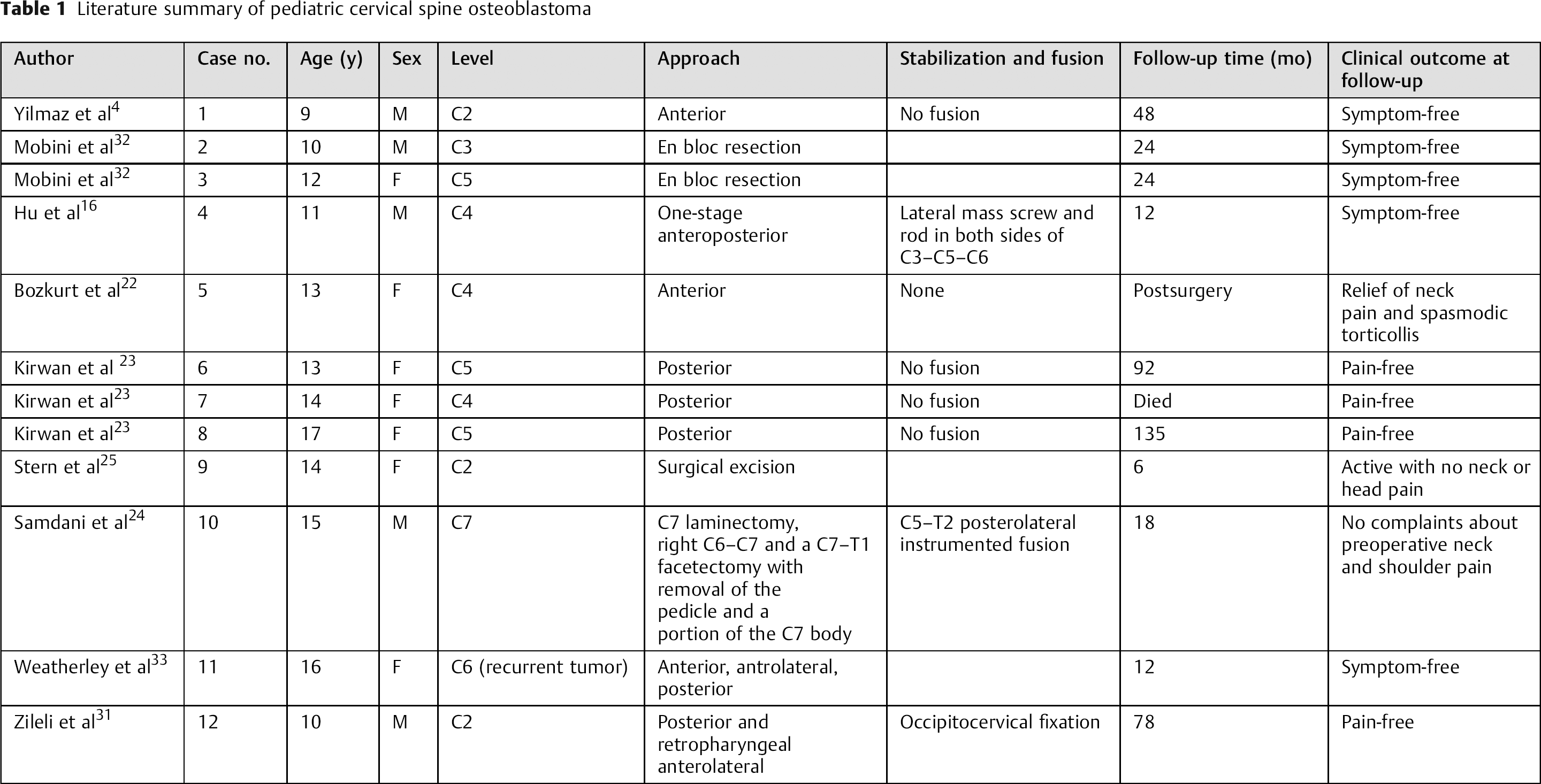
Cervical osteoblastoma in pediatric age group has been presented by several authors. 16 , 19 , 22 , 23 , 24 , 25 To our knowledge, the youngest reported case is a 9-year-old boy with a C2 corpus tumor. He was operated on through an anterior approach. No fusion was performed due to limited bone drilling and undamaged structures that are important in stability. The child was symptom-free with sclerosis of C2 corpus on CT at 4-year follow-up. 4 Mobini et al reported a 10-year-old boy and a 12-year-old girl with C3 and C5 osteoblastoma, respectively. 32 En bloc resection was performed. At 2-year follow-up, both patients were pain-free with no recurrences. In the case reported by Hu et al, 16 an 11-year-old boy had a destructive osteoblastoma with secondary aneurysmal bone cyst of the cervical vertebra. One-stage anteroposterior tumor resection was performed. The cervical spine was stabilized using a lateral mass screw and rod in both sides of C3–C5–C6 in a posterior approach. Length titanium cage filled with shattered bone from the left crista iliaca and fixed with a titanium plate was used to restore the C4 vertebral body defect. At 1-year follow-up, the patient was symptom-free with no evidence of recurrence or instability in his imaging studies. Bozkurt et al presented a C4 vertebral body osteoblastoma in a 13-year-old girl who was surgically managed via an anterior approach. 22 Stabilization and fusion procedures were not needed. Postoperatively, she was relieved from neck pain and spasmodic torticollis with no neurologic deficits. CT showed total excision. In the series by Kirwan et al, which presented 18 cases with osteoid osteoma and osteoblastoma, 23 three cases with cervical osteoblastomas were reported (13-, 14-, and 17-year-old females with C5, C4, and C5 tumors, respectively). A posterior approach was used to access the laminas, spinous processes, and pedicles that where the sites of tumor involvement. Spinal fusion was not required. A C2 osteoblastoma was diagnosed in a 14-year-old girl and was excised. Six months after the operation, the patient was active with no neck or head pain and had full range of motion. 25 Samdani et al reported a 15-year-old boy with C7 osteoblastoma. 24 He was managed with preoperative embolization that was followed a day later by C7 laminectomy, right C6–C7, and C7–T1 facetectomy with removal of the pedicle and a portion of the C7 body. C5–T2 posterolateral instrumented fusion was done. At 18-month follow-up, he no longer complained of his preoperative symptoms (neck and shoulder pain), and CT showed solid fusion with no evidence of recurrence. A 16-year-old girl with recurrent C6 osteoblastoma was reported by Weatherley et al. 33 On the primary surgery, through a posterior approach, tumor excision was attempted followed by posterior fusion from C5 to C7. Three years after the operation, the patient had pain recurrence, and imaging studies revealed a much more extensive involvement of the pedicle and lateral mass. On the second operation, a combined anterior, anterolateral, and posterior approach was used. Radical excision was confirmed by radiographs. At 1-year follow-up, the patient was reported to be pain-free. Zileli et al, in their retrospective study looking at 35 cases of cervical spine tumors, reported a case of C2 osteoblastoma in a 10-year-old boy that was managed with posterior and retropharyngeal anterolateral approaches. 31 Occipitocervical fixation was done. At 78-month follow-up, they reported no evidence of disease. Table 1 summarizes the literature review of pediatric cervical spine osteoblastoma.
Literature summary of pediatric cervical spine osteoblastoma
A two-stage surgical approach for resection of C6 osteoblastoma has been reported by Shikata et al in a 31-year-old man. 9 They performed anterior spinal fusion from C5 to C7 with a bone graft that was secured with an alumina-ceramic screw. Good bone fusion and no tumor recurrence were reported in 1-year and 6-month follow-up.
Decreasing intraoperative blood loss, reducing the chance of tumor recurrence and postoperative complications, facilitating total tumor resection, and improving patients outcomes have been the rationale behind utilizing preoperative embolization for this vascular tumor. 24 , 34 , 35 , 36 Facilities to perform preoperative embolization are not available at our center for individuals younger than 15 years of age.
Pediatric spine fusion is a challenging issue because the surgeon must take into account the small anatomical space, the ongoing spine growth, and mobility. Posterior sublaminar wiring has been reported by Crostelli et al in the pediatric age group. 6 Wire rupture was reported 1 and 2 years postoperatively, although the rupture did not result in any consequences.
We introduce the anterior C1 sublaminar wiring technique. This novel approach can be used in anterior reconstruction of the cervical spine for children. At 2-year follow-up, our patient was symptom-free and showed no evidence of instability; results of this technique are promising.
Conclusion
With the successful reconstruction of C2–C3 vertebral bodies presented herein, this novel approach could be considered in future cervical spine reconstruction in patients who have an intact C1 arc and resected lower bodies.
Disclosures
Ali Haghnegahdar, none
Mahsa Sedighi, none
