Abstract
Introduction
One surgical treatment modality for the management of degenerative disc disease causing back and/or leg pain is interbody fusion. Nonunion is associated with poor outcomes. The gold standard bone graft for augmenting and achieving fusion is iliac crest autograft but significant donor morbidity is associated with its harvest. Allografts require processing, to prevent transmission of infections, which impairs their osteogenic and osteoinductive properties. An alternative synthetic bone graft that avoids the morbidity of autograft is safe, and provides reliable fusion and is therefore desired. In our institution, we have used three synthetic bone grafts, Actifuse (Baxter, USA), i-FACTOR (Cerapedics, USA) and Vitoss BA (Stryker, USA), which all report to be bioactive and to enhance bone fusion in transforaminal lumbar interbody fusion (TLIF) procedures. They were all used in combination with morselized local autograft.
Materials and Methods
Patients undergoing TLIF surgery with one of the mentioned bone grafts were identified from hospital records. Postoperative radiographs were reviewed by an independent blinded radiologist for evidence of spanning trabeculae on orthogonal radiographs.
Results
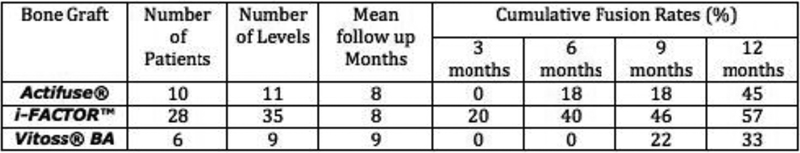
A total of 40 patients (55 levels) were included. The number of patients, operated levels, follow-up, and cumulative and overall fusion rates for each bone graft group are demonstrated in Table 1.
Conclusion
Patients who underwent TLIF with a mixture of i-FACTOR combined morselized local autograft had a higher rate of fusion with a similar mean follow-up and higher cumulative fusion rates over the period studied than Actifuse and Vitoss BA. The smaller patient numbers in the Actifuse and Vitoss BA groups does limit comparisons.
None declared
