Abstract
Introduction
Intervertebral disc herniation afflicts 1% of adults causing painful radiculopathy by mechanical deformation and biochemical irritation of apposed neural structures. Most patients improve with conservative or surgical management,1,2 but nearly 5% experience a chronic neuropathic pain phenotype termed failed back surgery syndrome. This pain is refractory to medical therapy and occurs without surgically correctable structural pathology. In experimental models, noncompressive disc herniation elicits inflammatory histopathology and behavioral findings of mechanical allodynia and thermal hyperalgesia.3-6 In this work, we investigate chronic sensitization upon resolution of pathological inflammation and acute inflammatory pain. We further examine mechanisms underlying this sensitization, demonstrating heightened extent, distribution, and activity of TRPV1 receptors.
Materials and Methods
Procedure
C57BL/6 mice were divided into Sham and nucleus pulposus (NP) groups, undergoing mid-thigh sciatic nerve dissection, either exposure alone (Sham) or placement of littermate tail NP proximal to the nerve trifurcation. Over 3 weeks, animals were evaluated for behavior as defined below. Thereafter, they underwent intraplantar injection of subthreshold capsaicin (0.1 mg, 10 mL) or vehicle alone, testing behavior over 24 hours. Steps following animal sacrifice are detailed below.
Behavior
Mechanical sensitivity was tested by hindpaw Von Frey filament stimulation, measuring 50% paw withdrawal force. Heat sensitivity was tested by hindpaw stimulation using a radiant light source, recording latency to paw withdrawal. Cold sensitivity was tested by hindpaw stimulation with acetone, recording duration of nursing the paw over the following minute.
Immunohistochemistry (IHC)
Following perfusion and fixation, spinal cords, dorsal root ganglia (DRG), and sciatic nerves were sectioned and stained for inflammatory and immune cell markers, cytokines, and cation channels. Imaging was by fluorescence microscopy, with blinded evaluation of fluorescence uniformity and intensity.
DRG Explants
Following perfusion and incubation, DRG explants were immersed in an ionic assay buffer, stimulated with 2.5 mM capsaicin in the presence of 1 mM cobalt chloride. DRGs were fixed, sectioned, and silver stained. Light microscopy was performed and the fractional stained area was computed.
Statistical Analysis
Behavioral outcomes were analyzed by ANOVA. IHC was analyzed by Student t-test.
Results
Behavior
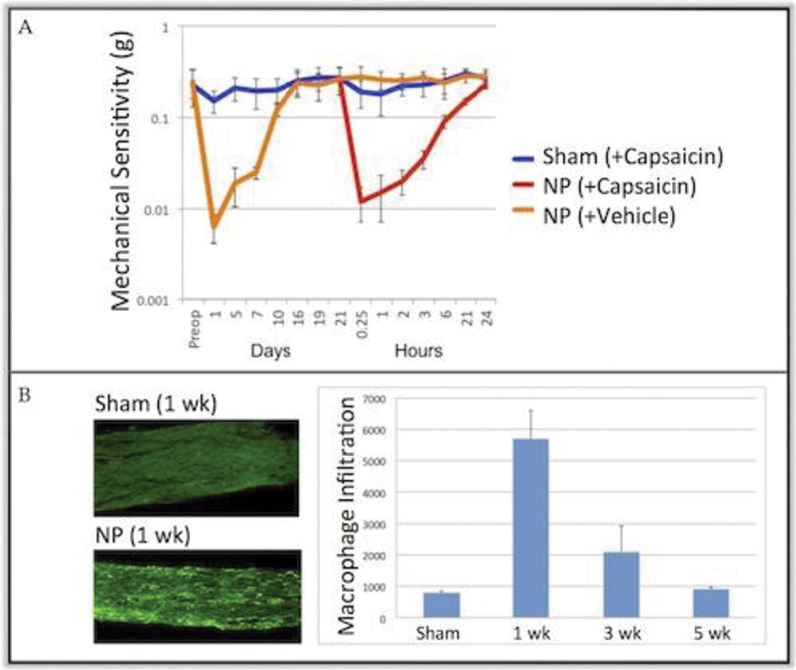
NP mice exhibited an acute painful phenotype, with early onset of mechanical allodynia, thermal hyperalgesia, and cold allodynia compared with controls. Von Frey filament testing for the limb ipsilateral to surgical NP injury demonstrated a lower 50% withdrawal threshold than sham animals (Fig. 1A, p < 0.02, NP [orange] and sham [blue]). Following recovery of the painful phenotype, subthreshold intraplantar capsaicin regenerated a profound allodynic response compared with sham animals and with vehicle-injected NP animals (Fig. 1A, p < 0.01, NP + capsaicin [red] vs. NP + vehicle [orange] and sham + capsaicin [blue]).
Immunohistochemistry
Sciatic nerve sections from NP animals show perineural autoreactive lymphocyte infiltration (not shown) and intraneural macrophage infiltration (Fig. 1B, p < 0.01). This was most profound 1 week after surgery, normalizing by 5 weeks. DRG analysis for ATF3, a marker of neuronal death, demonstrated no positivity compared with transection nerve injury eliciting substantial positivity. NP-treated animal demonstrated greater DRG expression of TRPV1, with distribution in both small- and medium-sized neurons. This noninjurious response was recapitulated in the spinal cord where no significant microglial proliferation (Iba1) was observed. Conversely, at 5 weeks, higher levels of dorsal horn TRPV1 expression was noted.
Conclusion
Heterotopic NP on mouse sciatic nerve generates an acute inflammatory neuritis, with a transient and resolving behavioral phenotype of mechanical allodynia, thermal hyperalgesia, and cold allodynia. Concomitant is an early immune-related infiltration of macrophages. Following both behavioral and histopathological resolution, these animals remain hypersensitive to peripheral stimulation with TRPV1-agonist capsaicin. This sensitivity can be explained by the heightened expression of TRPV1 at the DRG and in the spinal cord dorsal horn. DRG explants also have a greater ex vivo response to capsaicin stimulation. Together, the heightened sensitivity to noxious stimulation despite resolution of the acute behavioral phenotype coupled with heightened expression and activity of signal transduction cation channel TRPV1 suggests structural encoding of hypersensitivity following acute radiculopathy. This work provides insight into the pathophysiology of neuropathic pain after disc herniation.
None declared
Weinstein JN, Lurie JD, Tosteson TD, et al. Surgical vs nonoperative treatment for lumbar disk herniation: the Spine Patient Outcomes Research Trial (SPORT) observational cohort. JAMA 2006;296(20):2451–2459
Weinstein JN, Tosteson TD, Lurie JD, et al. Surgical vs nonoperative treatment for lumbar disk herniation: the Spine Patient Outcomes Research Trial (SPORT): a randomized trial. JAMA 2006;296(20):2441–2450
Olmarker K, Iwabuchi M, Larsson K, Rydevik B. Walking analysis of rats subjected to experimental disc herniation. Eur Spine J 1998;7(5):394–399
Otani K, Arai I, Mao GP, Konno S, Olmarker K, Kikuchi S. Nucleus pulposus-induced nerve root injury: relationship between blood flow and motor nerve conduction velocity. Neurosurgery 1999;45(3):614-619, discussion 619–620
Shamji MF, Allen KD, So S, et al. Gait abnormalities and inflammatory cytokines in an autologous nucleus pulposus model of radiculopathy. Spine 2009;34(7):648–654
Shamji MF, Jing L, Chen J, et al. Treatment of neuroinflammation by soluble tumor necrosis factor receptor Type II fused to a thermally responsive carrier. J Neurosurg Spine 2008;9(2):221–228
