Abstract
The nucleus pulposus of the intervertebral disk contains high amounts of the proteoglycan aggrecan, which confers the disk with a remarkable ability to resist compression. Other molecules such as collagens and noncollagenous proteins in the extracellular matrix are also essential for function. During disk degeneration, aggrecan and other molecules are lost due to proteolysis. This can result in loss of disk height, which can ultimately lead to pain. Biological therapy of intervertebral disk degeneration aims at preventing or restoring primarily aggrecan content and other molecules using therapeutic molecules. The purpose of the article is to review recent advances in biological repair of degenerate disks and pain.
Eighty percent of the population will experience some form of back pain over their lifetime, with ∼10% of sufferers becoming chronically disabled. Although there are numerous causes of back pain, it is strongly associated with disk degeneration. 1 The treatment strategies employed currently can be nonsurgical or surgical and are aimed at symptomatic relief rather than repairing or reversing damage to the intervertebral disk (IVD). In fact surgery involving spinal fusion may actually accelerate disk degeneration at adjacent levels.
Normal Disk Function
It is possible to bend and twist the spine while resisting compression from gravity and muscle action because of the IVDs. 2 They are formed of the peripheral collagen-rich annulus fibrosus (AF) surrounding the proteoglycan-rich central nucleus pulposus (NP), sandwiched between cartilaginous end plates (Fig. 1). 3 The function of the disk is to resist compression and permit limited movements in which loading forces are spread evenly on the vertebral bodies. The AF consists primarily of collagen type I fibers and is responsible for resisting tensile forces, and the NP consists mainly of loosely assembled collagen type II fibers and proteoglycan, mostly aggrecan. The cell density in the human adult NP is 4 × 106 cells/cm3, and the AF has 9 × 106 cells/cm3, which is very low when compared with other tissues such as cartilage. 4 Most of the disk nutrition is through the cartilage end plates, but with age the vascularity decreases and the end plates become calcified, which limit the disk's nutrient supply.
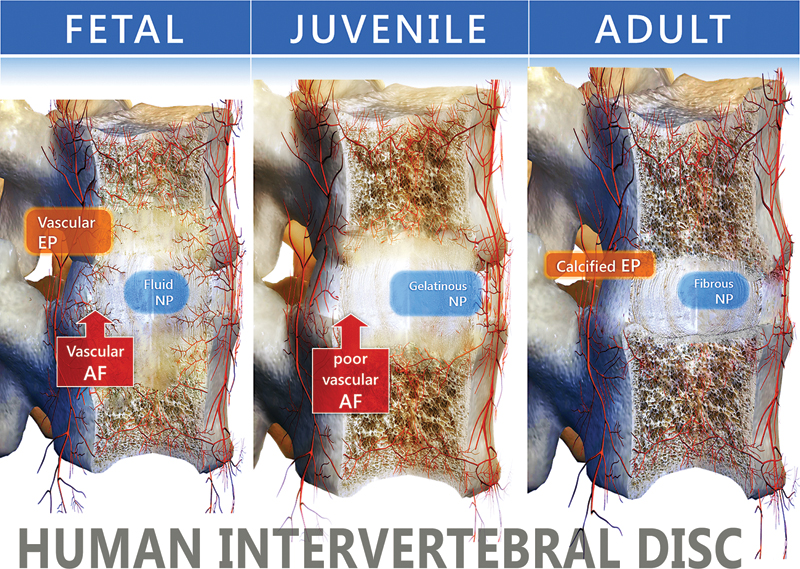
Schematic views of the human intervertebral disk, vertebrae, and vasculature in the fetal (26 weeks), juvenile (10 years), and adult (50 years) human. The figure shows the disk with the nucleus pulposus (NP) surrounded by the annulus fibrosus (AF) and separated from the vertebral bodies by the end plate (EP). The thickness and diameter of the end plate, the vascularity of the disk, and the abundance of notochordal cells in the nucleus pulposus decline with age. Mesenchymal cells gradually replace notochordal cells in the nucleus pulposus, and the consistency of the nucleus pulposus changes from fluid to more gelatinous because of decreased proteoglycan content.
Thus a sparse number of cells maintain homeostasis in an abundant extracellular matrix (ECM) by balancing synthesis and degradation (Fig. 2). This is orchestrated through complex anabolic (bone morphogenetic proteins [BMPs], transforming growth factor-β [TGF-β], and insulin-like growth factor [IGF]) and catabolic (matrix metalloproteinases, aggrecanases, and inflammatory cytokines like interleukin 1 [IL-1] and tumor necrosis factor-α [TNF-α]) processes, in addition to tissue inhibitors of matrix metalloproteinases (TIMPs), which control catabolic activities. The adult disk is the largest avascular tissue in the body, which is a major determinant of its metabolic functions. The main route for disk nutrition is through the cartilaginous end plate and is adapted to anaerobic metabolism because of a low oxygen tension. 5 , 6 This results in high concentrations of lactic acid and low pH, 7 conditions that are not conducive to high levels of protein synthesis required during tissue repair or regeneration. It is possible that an authentic disk cell might be the only cell capable of functioning under these adverse conditions.

The disk cell is a master of its destiny. The figure depicts the disk cell responsible for synthesizing matrix molecules such as aggrecan and collagen as well as degrading the matrix through the action of proteases. In a healthy disk there is a balance between synthesis and degradation.
Aging and Disk Degeneration
Mechanisms that contribute to degenerative changes in the disk include reduction in nutrient supply, diminished cell viability, loss of notochordal cells, genetics, cell senescence, and loading history. 8 , 9 These lead to biochemical alterations in composition and structure of the ECM due to both depleted synthesis and increased degradation, with aggrecan being particularly susceptible to proteolytic damage and loss. Aging of the IVD is associated with increased degeneration. 10 , 11 The disk has no endogenous repair activity. However, induced repair of disk tissue may be possible, as chymopapain treatment to degrade the degenerate NP stimulates new matrix formation, although not consistently. 12 , 13
During disk degeneration, the NP changes from a gelatinous structure to a more fibrous texture as fissures appear in the NP and the AF. The proteoglycan-to-collagen ratio (measured as GAG-to-hydroxyproline ratio) is highest in patients with grade 2 disks (∼ 23:1) and lowest in those with Thomson grade 4 disks (∼ 5:1). 11 In healthy juveniles (2 to 5 years old), the GAG-to-hydroxyproline ratio in the NP is ∼ 25:1. Similarly, in young adult disk with no degeneration (15 to 25 years), the GAG-to-hydroxyproline ratio within the NP is ∼ 27:1. With increasing age there is a marked drop in the GAG-to-hydroxyproline ratio, which declines to ∼ 5:1 in the elderly (60 to 80 years). These processes may be the results of a marked reduction of nutritional supply, due to regression of blood vessels and calcification of the end plates. 14 , 15
The focus of molecular therapy is to restore the juvenile disk composition to degenerative disks by stimulating the production of proteoglycan and other matrix components. This article reviews the research on using molecular therapy to treat disk degeneration and subsequently pain.
Molecular Therapy
Concepts in Tissue Engineering of IVD
There is growing evidence that laboratory-grown tissues or organs can be used to support the function of defective or injured body parts, and these tissue-engineering procedures have recently been applied to the disk. 16 , 17 , 18 , 19 Such advances in regenerative medicine have led to promising new approaches for the biological treatment of disk degeneration. Disk repair can be facilitated by the injection of growth factors, cell supplementation, gene therapy, and the introduction of biomaterials. Several recent investigations showed in rabbit models or in cell culture models of differentiation that it may be possible to use mesenchymal stem cells for IVD repair and regeneration, avoiding the need to harvest autologous disk cells. 20 , 21
Growth Factors
Growth factors are usually polypeptides that are involved in stimulation and maintenance of the integrity of the disk ECM. Typically, they bind to receptors resulting in signaling cascades between cells and communicate their effects through endocrine, paracrine, and autocrine mechanisms. The endocrine signaling mechanism involves the release of a growth factor from a specialized group of cells in the kidney, liver, heart, or other organs into the circulation and acting on a distant target tissue. In a paracrine mechanism, growth factor communication occurs between neighboring cells that have specific receptors within a tissue or organ. Autocrine signals, on the other hand, are those in which a growth factor acts on the same cell, and an intracrine signal is produced by growth factor acting within the same cell.
Growth factors, such as IGFs, TGF-β, epidermal growth factors (EGFs), and fibroblast growth factor (FGF), are known to stimulate new disk matrix formation (Fig. 3). 22 , 23 Other therapeutic molecules, such as anticatabolics (TIMP-1 and TIMP-2) and intracellular regulators (Smad proteins, Sox 9), are being used (reviewed by Yoon and Patel 24 ; Fig. 4).
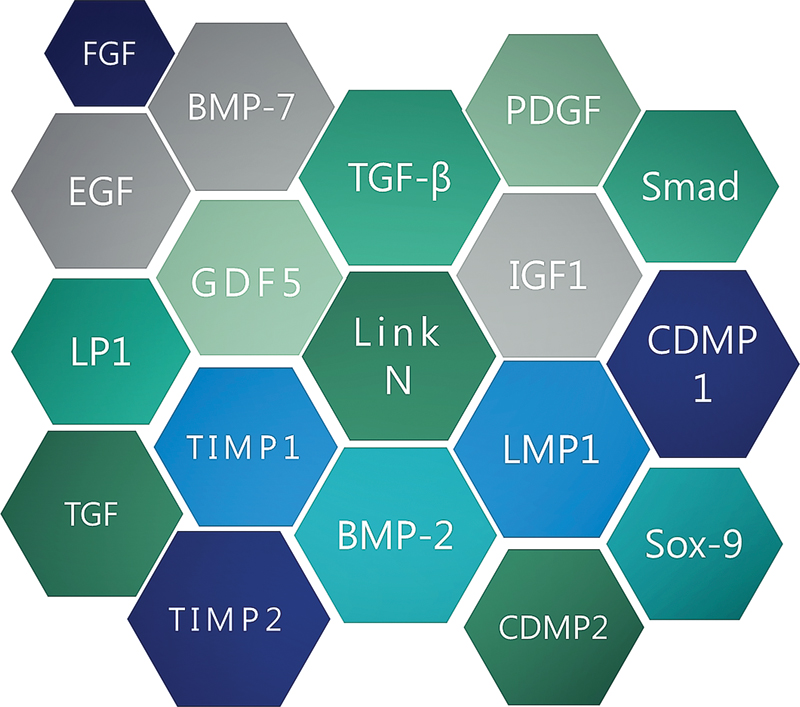
Therapeutic molecules used in disk repair: transforming growth factor (TGF), bone morphogenetic protein (BMP), growth differentiation factor (GDF), cartilage-derived morphogenetic protein (CDMP), Sma-Mad (Smad) proteins, Sox 9, LIM mineralization protein-1 (LMP-1), tissue inhibitor of matrix metalloproteinase (TIMP), insulin-like growth factor-1 (IGF-1), platelet-derived growth factor-1 (PDGF), epidermal growth factor (EGF), fibroblast growth factor (FGF), and Link N.
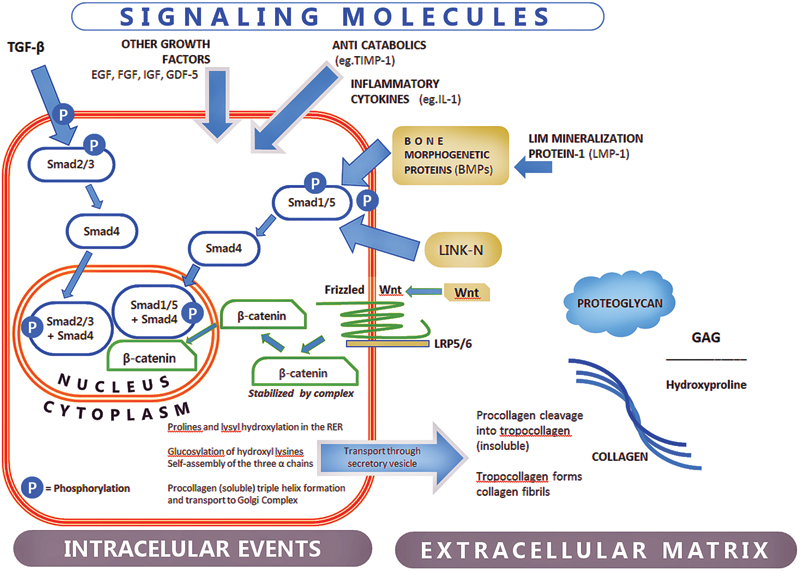
Signal transduction pathways in the disk cell by the TGF-β superfamily of ligands, Link N and Wnt. TGF-β superfamily includes BMPs. Typically, TGF-β superfamily activates its type II receptor, which recruits and phosphorylates a type I receptor. TGF-ßs are mediated by SMAD2 and SMAD3 that form complexes with SMAD4 and translocate to the nucleus, and BMPs and Link N are mediated by SMAD1 and SMAD5. The canonical Wnt pathways are initiated when Wnt protein bind to receptors of the Frizzled family and the LRP5/6 co-receptor. The signal is further mediated by Dishevelled family of proteins and leads to β − catenin cytoplasm accumulation followed by entering in the nucleus where it modulates gene expression. Abbreviations: BMP, bone morphogenetic protein; EGF, epidermal growth factor; FGF, fibroblast growth factor; GDF-5, growth differentiation factor 5; IGF, insulin-like growth factor; IL-1, interleukin-1; LMP-1, LIM mineralization protein-1; TGF, transforming growth factor; TIMP-1, tissue inhibitors of matrix metalloproteinase-1; Wnt, Wnt/b-catenin.
Insulin-Like Growth Factors
IGFs (IGF-1 and IGF-2) are polypeptides whose sequence is similar to insulin, and IGFs bind their receptors, IGF1R and IGF2R, forming a complex system termed the IGF axis. IGF-1 controls matrix synthesis and mitotic activity. It stimulated proteoglycan synthesis in cells of the bovine NP. 25 Further, Osada et al demonstrated the expression of IGF-1 mRNA and IGF-1 in cultured bovine IVD cells and proposed an autocrine/paracrine loop in the IVD. The effect of various growth factors on protein and proteoglycan synthesis on canine disk cells was dependent on the region of the disk from where the cells were derived. 23 TGF-β1 and EGF produced better responses than FGF, and IGF-I had no effect on cells from the AF. 22 However, IGF-I increased proteoglycan synthesis in monolayer cultures of bovine disk cells. 11 In rat disks, during the late stages of aging, IGF-1 receptors are downregulated in addition to an increase in IGF binding proteins, leading to a decreased IGF-1 response. 26 In a free-swelling explant culture model, IGF-I produced marginal synthetic response in the NP and none in the AF. 27
Fibroblast Growth Factor
There are 22 members of the FGF family in humans. Although FGF-1 is acidic, FGF-2 is basic and is the most studied. FGFs from 1 to 10 all bind to FGF receptors. Others such as FGF11 to FGF14 do not, and apart from possessing similar sequence homology, they function differently to the FGFs. FGF-3 receptors are expressed in disk cells. 28 Adding FGF-2 during culture expansion of NP cells in monolayer was found to sustain a differentiated cell phenotype by maintaining responsiveness to TGF-β1. 29 However, FGF-1 supplementation to degenerated murine caudal disks had little or no effect on aggrecan and type II collagen mRNA expression. 30 The authors concluded that AF cells in degenerated disks are responsive to some growth factors like TGF-β and not others in vivo.
Epidermal Growth Factor
EGF is a low-molecular-weight polypeptide that stimulates cell proliferation, growth, and differentiation of cells by binding to its receptor and stimulating protein tyrosine kinase activity. As discussed previously, EGF stimulated proteoglycan synthesis in the organ culture of dog IVD tissues. 31
TGF-β Superfamily
All immune cell lineages, including B cells, T cells, and dendritic cells as well as macrophages, secrete TGF-β, which negatively regulates their proliferation, differentiation, and activation by other cytokines. Thus, TGF-β is a potent immunosuppressant, and perturbation of TGF-β signaling is linked to autoimmunity, inflammation, and cancer. 32
TGF-β pathway starts at the GS domain where TGF-β is activated and the ligand binding to the type II receptor (TGF- ßRII) leads to recruitment of type I receptor (TGF-ßRI). Then it phosphorylates the SMAD2 and SMAD3 family of signal transducers. 33
With SMAD4 they form hetero-oligomeric complexes (also phosphorylation) and translocate to the nucleus, where they interact at the promoter with other transcription factors at DNA sequence-specific binding. 33 TGF-β regulates cell proliferation and ECM synthesis. TGF-β is stored in a latent form and very little is active. Disk cell proliferation and proteoglycan synthesis is stimulated by several growth factors that including TGF-β, IGF-1, platelet-derived growth factor, BMP-2, EGF, and BMP-7, also referred to as OP-1. 25 , 31 , 34 , 35 These data suggest that using growth factors or growth factor genes may improve the success of a cellular approach to disk tissue engineering. TGF-β exists in three isoforms and is part of a superfamily that includes BMPs. TGF-β was shown to be better at stimulating proteoglycan synthesis in disk cells than EGF, IGF-1, platelet-derived growth factor, and FGF. 31 Using adenovirus-mediated transfer of the TGF-β gene into normal rabbit disks in vivo, enhanced proteoglycan synthesis was demonstrated. 36 Others have showed increased collagen and proteoglycan synthesis by cells from degenerated human disks. 37
The signaling nodes, while repressing Smad, may be also responsible for interference in multiple pathways, which in turn will produce various pathophysiologic outcomes and may be also be linked to pain.
Bone Morphogenetic Proteins
BMPs were originally discovered by their ability to induce the formation of bone and cartilage, and the BMP pathway is paramount in the development of the dorsoventral axis in both vertebrates and invertebrates. Like TGF-β, BMPs are normally present in low amounts in healthy disk where they play a role in repair and maintenance of disk integrity. The receptors of BMP-2 and -4 were first found in hyaline cartilaginous cells within the end plate of young mice. The important functioning of BMP signals in disk degeneration was suggested by the fact that with age these BMPs and their receptors moved to the annulus and to the calcified cartilage at the site of enthesis. 38 Recombinant BMP-2 can stimulate proteoglycan synthesis without expression of osteogenic phenotype intervertebral disk cells. 39 Others have found that TGF-β and BMP-2 can induced disk calcification. 40 Finally, LIM mineralization protein-1 can stimulate proteoglycan synthesis through its action on BMP. BMP pathway consists of BMP dimers binding to a membrane complex composed of BMP receptors 1 and 2 (serine/threonine kinases). Regulatory Smad1/Smad5 through phosphorylation with Smad 4 (co-Smad) form a Smad1/5 + 4 complex that enters the nucleus. Inside, the nucleus regulates gene expression after it associates with transcription factors. Nakase et al reported the localization of transcripts for BMP-4, -6, and growth differentiation factor-5 as well as BMP receptors in the outer layer of the anterior annulus at an early stage of experimental cervical spondylosis, suggesting that BMPs are involved in chondrogenesis in spondylosis. 41
Recombinant human BMP-7 (OP-1), a member of the TGF-β family of proteins, stimulated the synthesis of proteoglycans and collagens when added to rabbit disk cells cultured in alginate beads, after depletion of the matrix by IL-1 or chondroitinase ABC. 42 , 43 To expand on these in vitro findings, the effects of BMP-7 were determined in vivo in a rabbit model of intervertebral disk degeneration. 44 , 45 BMP-7 injection increased proteoglycan and collagen content in the disk, reversing the decrease in disk height, which led to restoration of the biomechanical properties. These studies showed that BMP-7 could promote repair in disk degeneration.
N-Terminus of Link Protein as an Endogenous Growth Factor
Human articular cartilage aging is associated with proteolytic degradation of its constituent proteoglycan aggregates. 46 Link protein is found in aggrecan/hyaluronan aggregates, where it stabilizes the interaction between the two. The peptide DHLSDNYTLDHDRAIH (Link N), cleaved by stromelysin from the N-terminus of the Link protein, can act as a growth factor and stimulate synthesis of proteoglycans and collagens in articular cartilage. 47 , 48 , 49 , 50 Link N is conserved between rabbits and humans. It could represent an endogenous growth factor in the disk as it can stimulate the synthesis of both proteoglycan and collagen by disk cells in vitro, 51 , 52 increase proteoglycan levels in vivo 53 in a rabbit model of disk degeneration, and downregulate hypertrophic and osteogenic differentiation of human mesenchymal stem cells. 54 We also showed that the effects of this peptide could last for 12 weeks in the rabbit and for more than a week in human explant cultures (Gawri et al, in press, European Cells and Materials). We have recently shown that Link N activates Smad1/5 signaling similar to that used by BMPs (Fig. 4), Thus, Link N could represent a potential growth factor able to stimulate the production of an NP-like phenotype.
Gene Therapy
One of the limitations of growth factors is that they have short half-lives, transiently increasing matrix synthesis. Thus, a method is needed that can provide sustained growth factor concentrations within the IVD. Gene therapy offers that potential as it can direct a target cell to synthesize a desired substance by delivering a corresponding DNA or RNA using a viral or nonviral vector. Viral vectors and biocompatible materials, such as cationic liposomes, DNA–ligand complexes, and gene gun techniques, have been developed as vehicles to introduce potentially therapeutic genes into mammalian cells. Wehling et al first demonstrated gene transfer to the IVD. 55 Later, the efficacy of cDNA for TGF-β, 56 sox9, 57 LIM mineralization protein-1, 58 and TIMP 59 to upregulate synthesis of matrix into the disk was shown. However, gene therapy, although very promising for the treatment of disk degeneration, is still in its infancy.
Wnt Signaling
Wnt/b-catenin (hereafter called Wnt) signaling is involved in development, degeneration, and regeneration of the IVD. 60 , 61 , 62 The signaling cascade is initiated at the cell membrane by interaction between Wnt and the Frizzled receptors plus LRP5/6 co-receptors and is not depending on phosphorylation (Fig. 4). Canonical Wnt signaling stabilizes cytoplasmic β-catenin and its translocation into the nucleus, to regulate expression of Wnt-target genes. 63 Noncanonical Wnt signaling is independent of β-catenin signaling. They involve the activation of protein kinase C, calmodulin-dependent kinase II, and c-Jun N-terminal kinase. Wnt signaling has also been associated with degenerative joint disease. 64 Wnt signaling suppresses proliferation of NP cells and induces cell senescence of the IVDs. 60 , 61 Upregulation of matrix metalloproteinases by Wnt signaling causes dedifferentiation of NP cells, promoting cellular senescence and possibly leading to IVD degeneration. 60 , 61 Members of the TGF-β superfamily and Wnt signaling cascades have been shown to physically interact in different tissues, suggesting new targets for therapy. 65 , 66
Pain and Innervations
Mechanisms of Axial Low Back Pain
Although disk degeneration does not necessarily correlate with actual pain, disk degeneration can be found in most patients suffering from back pain. 1 Axial low back pain is characterized by spontaneous or movement-evoked pain or soreness. It is confined to the spine and low back region and can be induced by disk degeneration. IVDs have a sensory component that normally penetrates only the outer layers of the disk. 67 In patients with chronic low back pain, increased nerve fibers and blood vessels that express substance P into the AF and even penetrating the NP were observed, 68 which may be facilitated due to the loss of aggrecan and hydrostatic pressure. 9 , 69 , 70 Nociceptive nerve ingrowth into painful IVD is linked with nerve growth factor production by blood vessels growing into the IVDs, from adjacent vertebral bodies, 71 which can be upregulated in disk cells by the proinflammatory cytokines IL-1β and TNF-α (Fig. 5). 72
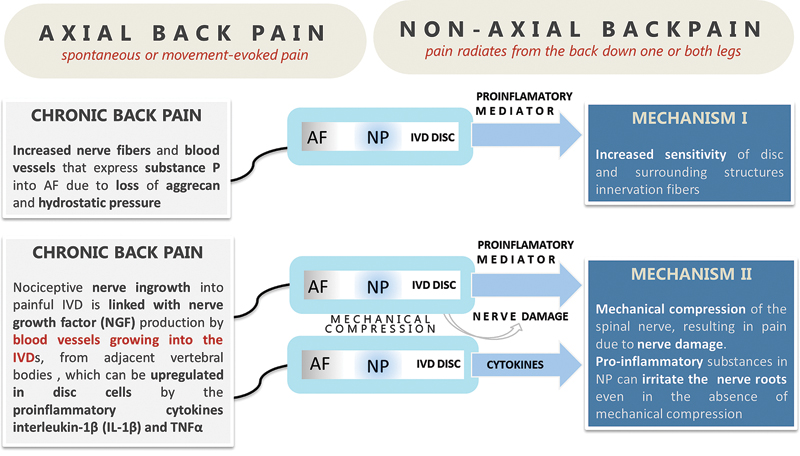
Axial and nonaxial back pain. Back pain is an intricate amalgam of conditions that includes both axial and radicular (nonaxial) pain. Axial back pain is defined as spontaneous or movement- evoked pain or discomfort localized to the spine and low back region. Nonaxial, referred to as radicular pain or sciatica, usually follows the trajectory of the sciatic nerve. Abbreviations: AF, annulus fibrosus; IL-1β, interleukin-1β; IVD, intervertebral disk; NGF, nerve growth factor; NP, nucleus pulposus; TNFα, tumor necrosis factor-α.
Mechanisms of Nonaxial Pain
Nonaxial pain (radicular pain or sciatica) in which the pain radiates from the back down one or both legs following the course of the sciatic nerve is thought to have two primary mechanisms. The first mechanism is that proinflammatory and pronociceptive mediators released from degenerating disks may sensitize fibers that innervate the disk and surrounding structures. The second mechanism is that severe disk degeneration and herniation can result in mechanical compression of the spinal nerve, resulting in pain due to nerve damage. Proinflammatory substances secreted by the NP may be involved in the etiology of nonaxial low back pain by irritation of nerve roots even in the absence of mechanical compression. 73 Local epidural application of NP or some of its constituent cytokines produces increased spinal nerve root excitability and hypersensitivity to painful stimuli. 74 Annular tears during disk degeneration can therefore release its contents and lead to nerve fiber irritation and back pain. Disk herniation can also result in compression of spinal nerve roots or dorsal root ganglia neurons. 75
Pain Receptors
Pain receptors are found on nerve endings located in the disk. Sensory fibers and their endings in the outer AF of lumbar intervertebral disks were first demonstrated in a rat. 32 The AF of the adult human intervertebral disk is sparsely innervated, with substance P localized on the endothelium of small blood vessels in the AF of disks removed during anterior fusion for back pain. 33 Once there is a pain stimulus, which may be due to tissue damage, activation of pain receptors takes place, releasing neurotransmitters that send information to the spinal cord and the brain. This process of pain transmission is called nociception and the pain receptors are called nociceptors. NK1 receptors through which vascular effects are mediated were found on vessels in the AF, suggesting a role for substance P in discogenic pain. Mechanoreceptors and nociceptors have been documented in the spine, 76 , 77 and they are more extensive in degenerated disks where nerve fibers were found in the anterior longitudinal ligament and in the outer region of the disk. 78 Apart from substance P-immunoreactive nerve fibers found in the anterior longitudinal ligament and the outer zone of the AF, fibers were also found in the inner parts of the disk. Finally, pain is an extremely complex process. Identification of the receptors and processes that are involved in the transmission of pain should lead to improved therapeutics in pain management.
Conclusion
Current biological repair approaches are exploring a combination of molecular, cellular, and tissue-engineering techniques to restore the integrity of the intervertebral disk or at least retard the further degeneration of NP. The target cohort of patients is dependent on the stage of disk degeneration. During early degeneration, when the AF remains intact, injection of therapeutic molecules such as growth factors, anabolic peptides (as an alternative to recombinant growth factors), or gene therapy (for prolonged delivery) to stimulate increased matrix synthesis is feasible. Supplementing the disk with more cells and/or scaffolds may be necessary in later stages of degeneration. However, a way to improve disk nutrition is essential. Maintaining disk height and improving the biological integrity of the intervertebral disk may lead to relief in pain. However, more research is needed to further establish the effects of these potential therapeutic agents on pain.
Disclosures
Fackson Mwale, None
Footnotes
Acknowledgments
I thank AO Spine, Canadian Institute of Health Research (CIHR), and North American Spine Society (NASS). Dr. Ovidiu Ciobanu generated the figures.
