Abstract
Introduction
Disk degeneration is characterized by the inability of the resident cells to maintain disk tissue healthy due to a change of their phenotype and their decreasing number.1 Cell therapies have mainly focused on delivering adult mesenchymal stem cells (MSCs) to counteract this process. Though able to inhibit it, these cells could not restore the disk to a healthy state2,3 and complementary methods should be explored. The disk originally contains two cell populations: the nucleus pulposus cells (NPCs) and the notochordal cells (NCs). As NCs are not present in adult humans, their potential for disk regeneration has been poorly explored. We hypothesized that recreating the original cell combination of a developing disk (when the largest amount of matrix is produced) should induce discogenic cell phenotype and promote disk matrix formation. In the present study, NCs were combined with MSCs (used to complement the decreasing disk cell population) and to NPCs (representative of the native cell population) in alginate beads and their effects on matrix production and cell phenotype were evaluated after 28 days of culture.
Materials and Methods
MSCs were isolated from bone marrow of calves and expanded up to P3 (n = 4). NPCs were extracted from caudal disks of cows (n = 12; 3 donors pooled per repeat) and NCs from lumbar and thoracic disks of young pigs (n = 8; 2 donors pooled per repeat). To distinguish the different cell types, NCs were stained with CFDA-SE and NPCs with orange CMTMR cell trackers before seeding. MSCs remained unstained. MSCs, NPCs, and NCs were mixed as follows (100% referring to a final concentration of 3 million cells/mL): (i) “NC”: 0% MSCs, 0% NPCs, 50% NCs; (ii) “MSC + NC”: 100% MSCs, 0% NPCs, 50% NCs; (iii) “NPC + NC”: 0% MSCs, 100% NPCs, 50% NCs; (iv) “Mix”: 50% MSCs, 50% NPCs, 0% NCs; (v) “Mix + NC25”: 50% MSCs, 50% NPCs, 25% NCs; (vi) “Mix + NC50”: 50% MSCs, 50% NPCs, 50% NCs (n = 4 for each cell combination). For all combinations, porcine skin fibroblasts (SFs) were used to control for specific effects of NCs. Cells were encapsulated in alginate beads and cultured in high glucose DMEM with 10% FBS and 1% penicillin/streptomycin at 2% oxygen for 28 days. MSCs alone (100%) were cultured under chondrogenic conditions (Ch + ; serum free medium supplemented with 10 ng/mL TGFß3 and 100 nM dexamethasone) to confirm the chondrogenic potential of the MSCs. At day 1 and 28, cell combinations were assessed by FACS; cell viability by confocal imaging (calcein blue staining); extracellular matrix composition by biochemical assays (DMMB, Hoechst dye and Chloramin-T assays for glycosaminoglycan (GAG), DNA and hydroxyproline (HYP) content, respectively) and histological staining (Alcian blue); and cell phenotype by RT-qPCR (for collagen types I, II and X, aggrecan, cytokeratins 8 and 19, vimentin).
Results
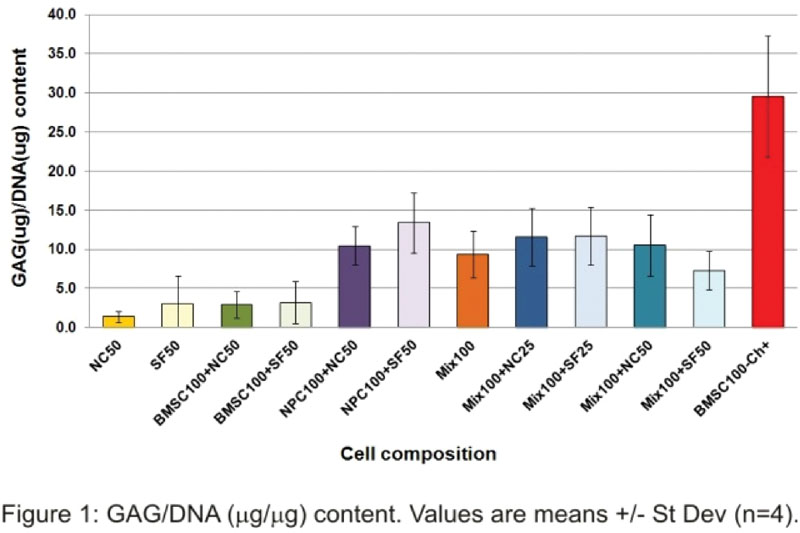
Ratios of the different cell combinations were validated by FACS and confocal imaging at day 1. Chondrogenic potential of MSCs was confirmed by GAG and collagen (HYP) production by MSCs cultured under chondrogenic conditions. At day 28, cells were viable in all combinations (confocal imaging) and proliferated in all conditions, except for MSC + NC and MSC + SF (DNA assay). Orange CMTMR staining however could not be detected at day 28, most likely due to substantial NPC proliferation (×2.5 to ×5.5 the initial cell number). For all cell combinations, GAG was produced at day 28 (Fig.1) but to a very limited amount for NCs only. Adding NCs to NPCs or MSCs + NPCs (but not just MSCs) increased GAG production. However the same trend was observed when adding SFs. Moreover, co-culturing NCs with NPCs + MSCs may have a synergistic effect as the Mix + NC25 group contained more GAG than the expected values [0.5 × MSC + NC + 0.5× NPC + NC]. Collagen production (HYP) was only detected for MSCs cultured in chondrogenic conditions. Influence of the different co-cultures on cell phenotype is currently being checked by RT-qPCR analysis.
Conclusion
These results show that NCs can positively affect disk matrix production by MSCs + NPCs and that they may act more as regulators than progenitors as they do not produce GAG when cultured alone. These positive effects, however, are only modest at the protein level (gene expression ongoing), suggesting that NCs do not provide a strong simulative signal, contrary to what has been reported for NCs co-cultured with MSCs4,5 or NPCs.6 These studies, however, used NCs cultured for maximum 4 days against 4 weeks in the present study. Early disappearance of NC vacuoles (observed with confocal imaging) may indicate NC dedifferentiation and less powerful stimulative signal in the long term. This work was supported by an AOSpine International Hans Joerg Wyss Foundation Research Award grant No HJW2011-SU12.
Yes
Freemont. Rheumatology 2009;48(1)
Sakai, et al. Biomaterials 2006;27(3)
Yang, et al. Molecular Therapy 2009;17(11)
Aguiar, et al. Exp Cell Res 1999;246(1)
Erwin, et al. Spine 2006;31(10)
Korecki, et al. Stem Cell Research and Therapy 2010;1(2)
