Abstract
Introduction
Confluent fibrosis, manifested as over-accumulation of fibrous matrix during tissue repair or remodeling, leads to scar formation and often organ failure. Studies suggest that 68% of protruded intervertebral disks and 44% of prolapsed IVD exhibit signs of fibrosis in the nucleus pulposus (NP) and that disk scarring indicates advanced stages of degeneration. It is not clear how fibrosis contributes to the initiation or progression of disk degeneration, and conversely if the prevention of fibrotic events plays a role in IVD repair or regeneration. Mesenchymal stem cells (MSCs) may arrest IVD degeneration and cross-talk with IVD cells via cell-cell contact or long-range signaling, implying MSCs may modulate disk microenvironment and function indirectly. Lupine is a reliable model for IVD degeneration studies as their lumbar spine bears biological relevance to humans regarding mechanics and age-related changes. Studies showed that puncturing to the annulus could transform the NP into a fibrocartilaginous phenotype, recapitulating the fibrotic features of human disk degeneration. We here tested if MSCs can rectify IVD degeneration and its function through modulation of fibrotic events, and investigated the pathways of the action.
Materials and Methods
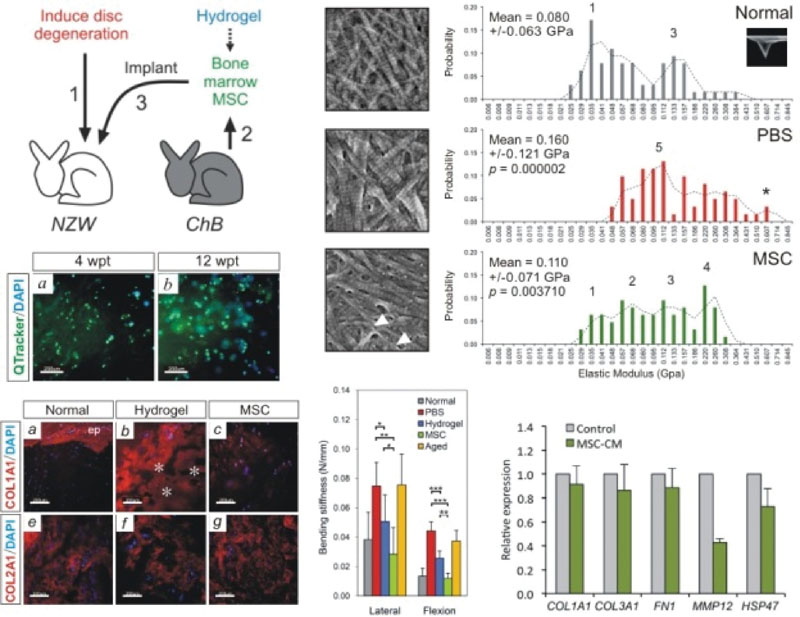
Total 6-month-old NZW rabbits with puncture-induced lumbar disk degeneration were intradiscally implanted with Chinchilla bone marrow MSCs, labeled by Qtracker and encapsulated in oligopeptide-hydrogel at 5 x 10E3 cells per disk (n = 8). Saline or hydrogel alone was injected as control. Treated disk levels were monitored by 3T MRI and radiographs up to 1 year. At 4 to 12 weeks posttreatment, the mechanical integrity of NP and joint segment were investigated by confined compression and motion stiffness tests. The collagen fibril diameter (n = 120) and the porosity of the fibril meshwork in the NP were analyzed in situ using scanning electron microscopy and morphometric analyzer. The elastic modulus of collagen fibrils (n = 75) in the NP was measured by nanoindentation using atomic force microscopy. Collagen I and II content was assessed by immunofluorescence. Human bone marrow MSC-conditioned media (MSC-CM) was collected and used to culture alginate-encapsulated NP cells harvested from patients with Pfirrmann grade III/IV disk degeneration. After 7 days, expression of fibrotic genes was studied using real-time PCR (n = 3).
Results
T2-weighted MRI and disk height analysis validated that MSCs arrested the degeneration. Motion segment stiffness was significantly attenuated in MSC-treated disks, indicating MSCs alleviated ankylosis caused by the degeneration and restored the spinal segment function. MSCs regenerated the swelling pressure and compressive strength of the NP, implying that MSCs regenerated joint function through preserving the biphasic tissue properties. Collagen molecules in the degenerated NP aggregated into gigantic fibrils, about 2-fold larger in diameter, resulting in fewer fibers and larger interfibrillar space. However, MSCs inhibited the deregulated higher-order assembly of fibrils. MSCs significantly reduced collagen I deposition and prevented the increase of fibril elastic modulus in the NP associated with degeneration. These suggest that the abnormal collagen fibrils are not solely resulted from fibril fusion but also a heterotypic change of collagen composition, and that the positive effects of MSCs are coupled to a regulation of fibrillogenesis in the NP matrix. MSC-CM repressed the expression of fibrosis-related genes MMP12 and HSP47 in degenerative human NP cells.
Conclusion
MSC transplantation can alleviate fibrotic diseases including pulmonary, renal, and cardiac fibrosis through pathways independent of MSC differentiation. Our findings imply that MSCs also rescue disk degeneration and recover disk function by suppressing fibrotic events. The action of MSCs is elicited through a regulation of fibrillogenesis in part via paracrine signaling to the degenerative NP cells. We propose a model in which MSCs reinforce the mechanical integrity of NP by normalizing the interplay between collagen meshwork and proteoglycan. Disk repair by augmenting the antifibrotic function of MSCs or use of other antifibrotic agents warrants further investigation.
This study is supported by the Research Grant Council of Hong Kong and HKU Foundation.
Yes
None declared
