Abstract
A case of feline cutaneous phaeohyphomycosis due to Cladophyalophora bantiana is described. The cat was presented with breathing difficulty and a swollen, ulcerated nodule on the dorsal nose and left nostril. Histological examination of the nodule revealed a cystic granulomatous dermatitis characterised by neutrophils, macrophages and giant cells. Pigmented, yeast-like fungus cells and hyphal elements were easily identified in haematoxylin-eosin stained tissue sections. Cladophyalophora bantiana was isolated from a tissue specimen. This organism, primarily known to cause cerebral infection in humans and cats, only rarely causes cutaneous infection. Despite anti-fungal chemotherapy two relapses occurred. The cat was feline immunodeficiency virus - and feline leukemia virus-negative and even if the owner was unaware of trauma, the hypothesis of wound contamination is the most likely.
Phaeoid (brown-pigmented) fungi with mycelial morphology in tissues cause phaeohyphomycoses in humans, mammals, and lower animals. The morphological aspect of the causative agent separates phaeohyphomycoses from other clinical types of disease involving brown pigmented fungi: chromoblastomycosis, characterised by spherical fungus cells in tissues (sclerotic bodies) and the eumycotic mycetoma, with tissue formation of ‘black granules or grains’ (Salfelder 1990). The terms phaeohyphomycosis, chromoblastomycosis, and eumycotic mycetoma have sometimes been used improperly to describe infections by pigmented fungi, resulting in confusion over proper terminology (Matsumoto & Ajello 1998).
Phaeohyphomycosis occurs worldwide, usually the infection follows the traumatic implantation of fungal cells from contaminated soil, thorns or wood splinters. The ingestion or inhalation of spores might also cause deep infection (Dixon et al 1987, Matsumoto & Ajello, 1998).
The isolated fungi belong to several taxonomic groups, with the common characteristic to form pigmented hyphae (Matsumoto & Ajello 1998, Scott et al 2001). The number of fungi listed by Ajello (1981) in 1981 (32 species in 18 genera) increased in 1994 to 101 species in 57 genera (Matsumoto et al 1994), currently involves 109 species in 60 genera (Matsumoto & Ajello 1998) and is expected to further increase in the future (De Hoog et al 2000).
The taxonomy of Cladophyalophora bantiana has been controversial for years. Saccardo attempted the first description as Torula bantiana in 1912 from an isolate of a human case of cerebral phaeohyphomycosis (Banti 1911). In 1960 T bantiana was transferred to the genus Cladosporium (Borelli 1960), but McGinnis et al (1986) classified in detail the morphological and physiological differences between C bantiana and some Cladosporium and reclassified the fungus in the genus Xylohypha. Recently, de Hoog et al (1995) placed Xylohypha bantiana in the genus Cladophyalophora. This genus accounts for five pathogenic species each causing a characteristic mycosis, distinct from that caused by Cladosporium species, only occasionally found in man as opportunists (De Hoog 1983). The five pathogenic species include C arxii, C bantiana, C boppii, C carronii and C devriesii.
Cladophialophora bantiana is responsible for phaeohyphomycosis primarily involving the central nervous system and integument (Dixon et al 1989). In humans and cats this species is highly neurotropic (Jang et al 1977, Hill et al 1978, Bennett et al 1979, Shinwari et al 1985) and the multiple granulomatous lesions, which are found in the brain and often accompanied by chronic meningitis, can be lethal. Neurotropism is related strictly to the presence of introns in the 18S rDNA subunit of the fungus (Gerrits van den Ende & de Hoog 1999). Even if the route of entry is not always obvious, the introduction through inhalation is suspected. Phaeohyphomycosis due to C bantiana can rarely produce cutaneous and subcutaneous infection in humans (Jacyk et al 1997, Patterson et al 1999). In the cat fungi such as Alternaria alternata (Dhein et al 1988, Outerbridge et al 1995, McKay et al 2001), Alternaria infectoria (Roosje et al 1993), Drechslera spicifera (Muller et al 1975, Chermette et al 1997), Exophiala jeanselmei (Bostock & Coloe 1982), Exophiala spinifera (Kettlewell et al 1989), Fonsecaea pedrosoi (Fondati et al 2001), Moniliella suaveolens (McKenzie et al 1984), Phialophora verrucosa (Dion et al 1982), Scolecobasidium humi-cola (Van Stenhouse et al 1988), Staphylotrichium coccosporum (Fuchs et al 1996), Stemphylium spp (Sousa et al 1984) and others, can cause cutaneous and subcutaneous phaeohyphomycoses. Cases of feline subcutaneous phaeohyphomycosis were described in which Cladosporium sp and Torula bantiana were identified (Sousa et al 1984, Van den Broek & Thoday 1987). Since several modifications have been made on taxonomy of phaeohyphomycosis these should be considered cases due to C bantiana.
In the present study we report a case of phaeohyphomycosis in a cat in which fungal culture yielded C bantiana as the causative agent and histopathology showed typical pigmented fungi. Surgical excision of the deep-seated mass was not possible and chemotherapy is discussed.
Clinical history
A 5-year-old male European domestic short-haired cat was presented with a history of breathing difficulty due to nasal swelling. At dermatological examination a clear nasal discharge from the left nostril, an oedematous swelling on the dorsum of the nose and on the left side of the muzzle, which also affected the nostril, were seen. At physical examination the cat was otherwise healthy. A CBC and serum biochemical profile were within normal limits. Differential diagnosis included deep mycosis, bacterial granuloma, foreign body granuloma, squamous cell carcinoma and melanoma. Due to pigmentation of the nodule, deep mycosis and melanoma were considered as the most likely. Since the client declined most of the diagnostic plan suggested due to the expense, surgery was indicated. In case of deep mycosis the excision of the mass could not ensure complete removal of all the fungal cells and the cat was given anti-fungal therapy (itraconazole, 5 mg/kg BID per os). A slight improvement of clinical signs was seen in a few days followed by relapse and reactivation of the cutaneous lesion (Fig 1) and severe gastrointestinal signs.

Cat. Swollen, oedematous and dark pigmented nodules on the dorsum of the nose and left nostril.
Material and methods
A biopsy from the dorsum of the nose was performed and a tissue specimen from the nodule was taken, after removal of overlying epidermis, for mycological investigation. The skin sample was fixed in a 10% buffered formalin solution and routinely embedded in paraffin. Five-micrometer-thick sections were stained with haematoxylin and eosin for histological evaluation, additional 5-μm-thick sections were stained with periodic acid Schiff and Grocott's methenamine silver (GMS) stains for fungi. The tissue specimen was cultured on Sabouraud's dextrose agar (Difco, Detroit, Mi, USA) at 25°C.
Results
Histopathological examination of the nodule presented an intradermal and subcutaneous granulomatous dermatitis with a cystic centre. The lesion was encapsulated by a layer of dense collagenous connective tissue and consisted mainly of neutrophils, macrophages and multi-nucleated giant cells. Brown pigmented fungi were readily detected in the haematoxylin-eosin stained infiltrate and were positively stained by the Grocott method (Fig 2). The fungi showed a variety of morphology ranging from isolated spherical cells, rarely dividing by separation in a single plane, to dark walled, short, septated mostly unbranched hyphal elements (Fig 3), to catenulate spherical cells, the so called toruloid or moniliform hyphae. Rounded cells ranged from 2.6 to 7.1 μm in diameter, hyphae were approximately 3–4 μm in width.

Skin from the dorsum of the nose. Several yeast-like fungus cells and hyphal fungal elements stain positively with Grocott method (Grocott stain, scale bar=20 μm).
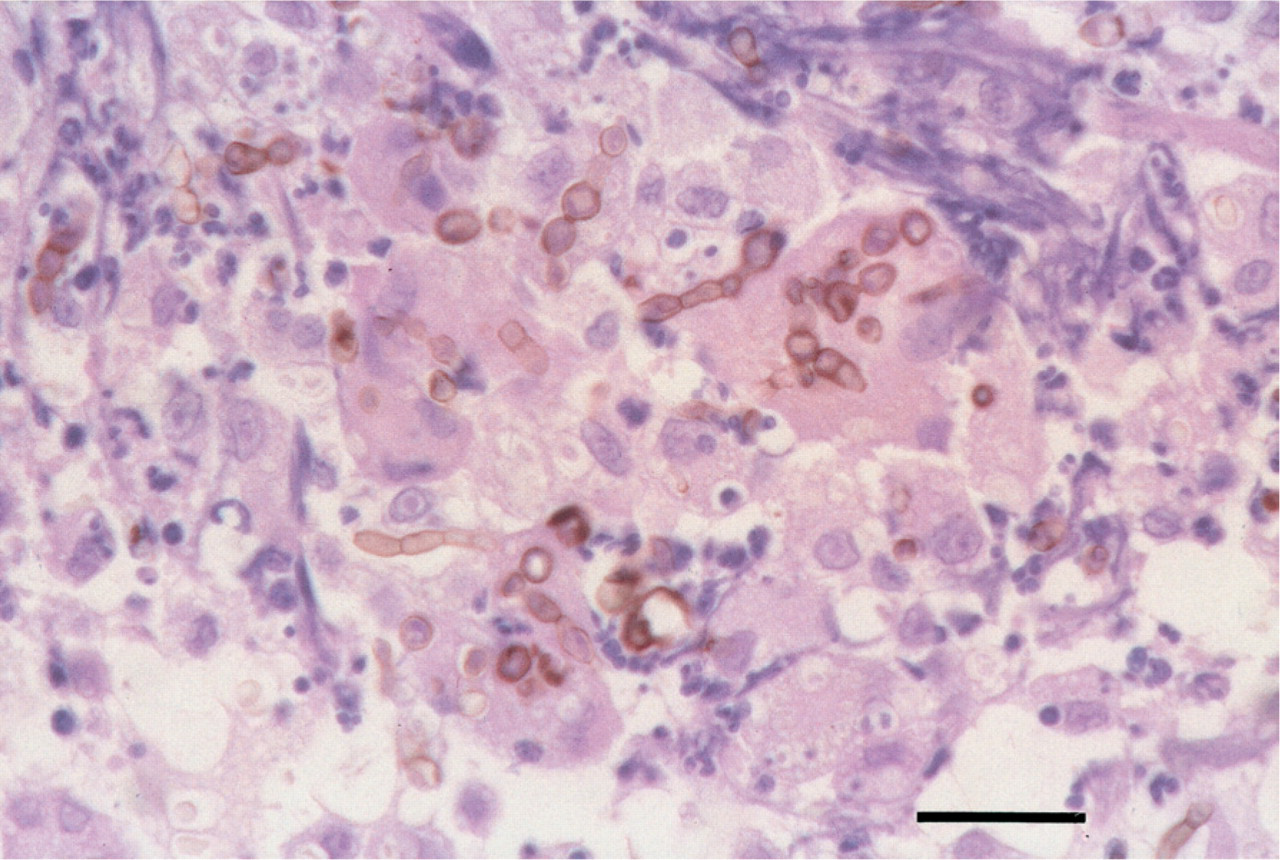
Skin from the dorsum of the nose. Brown pigmented, short unbranched hyphal elements and yeast-like fungus cells inside macrophages and giant cells (H-E, scale bar = 20 μm).
The culture showed several slow growing, small, deep green, velvety colonies at the 6th day. Microscopic features of the isolate consisted of fusiform conidia in moderately long chains and scarcely differentiated hyphae (Fig 4). Based upon macroscopic and microscopic features of the isolate the fungus was identified as C bantiana. The owner refused therapy with amphotericin B and the cat was given fluconazole 2.5 mg/kg BID for five days, followed by 5 mg/kg per os BID. Response to therapy was obtained in three months with return to clinical normality. At this time the cat showed moderate loss of appetite and the owner chose to discontinue the therapy. Relapse of both cutaneous and respiratory signs occurred in 15 days. The cat was serologically investigated for FIV and FeLV with negative results. A CBC, serum biochemical profile and liver enzymes (ALT 48 U/l, AST 32 U/l) were unremarkable and the same therapy (fluconazole) at the same schedule was given to the cat, which is still clinically normal after six months.
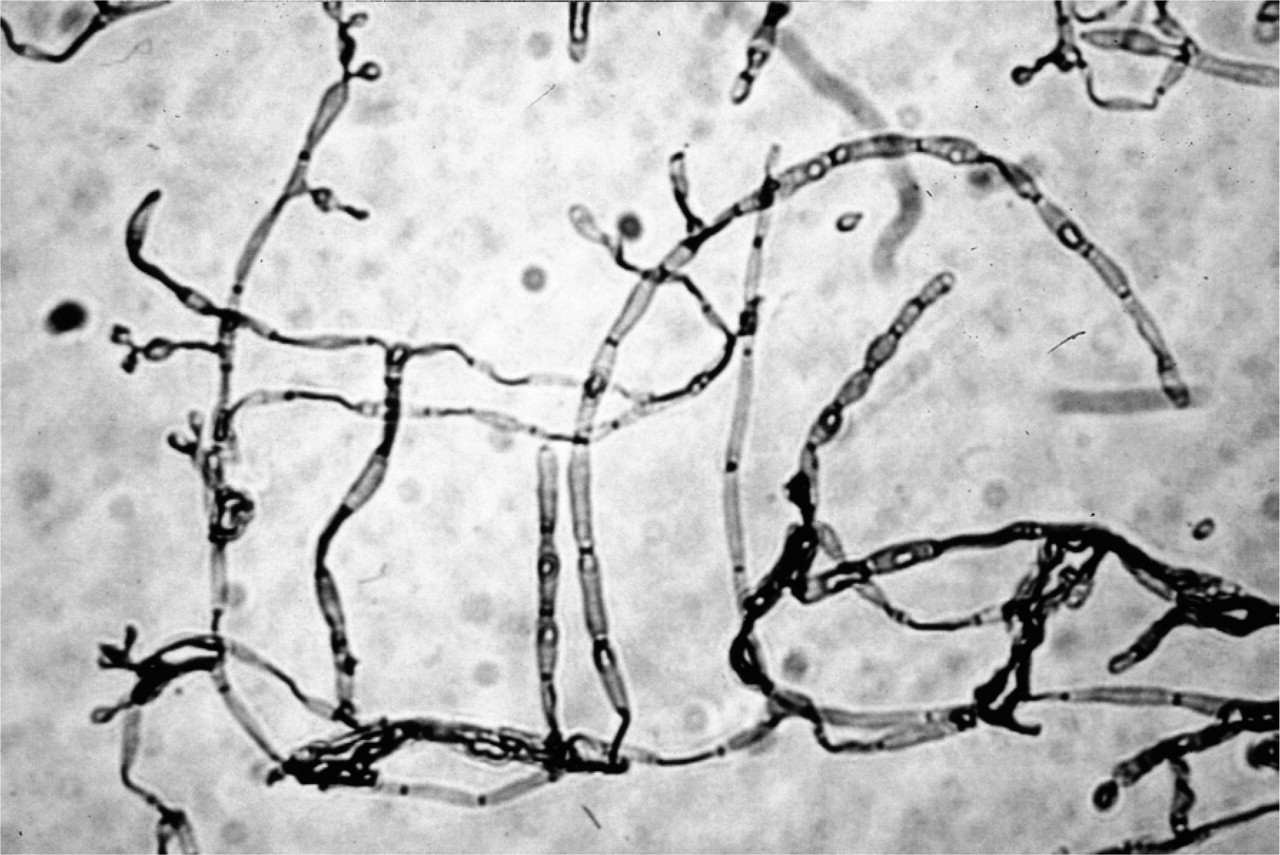
Unstained preparation from culture. Moderately long chains of mostly fusiform conidia arising from scarcely differentiated hyphae.
Discussion
Cutaneous and subcutaneous phaeohyphomycoses have been documented in cats. In Table 1 the fungi isolated from phaeohyphomycosis reported in the literature are listed along with the corresponding anatomical location. Some of the cases are listed with the general definition of phaeohyphomycosis since isolation of the fungus was not performed (Hill et al 1978, Sisk & Chandler 1982). Many of the nodules were located on the nose, as in our case, or on distal limbs. Besides the anatomical location, the history might help in directing the diagnosis. Trauma occurring before initial examination should suggest a penetrating injury, which is often responsible for the implantation of the infectious agent. This assessment was not possible in our case. Even if the anatomical location and history are suggestive of the disease, both mycology and histopathology, must be performed to achieve an aetiological diagnosis. Histopathology is confirmatory and allows differentiation from other lesions which often affect the dorsum of the nose in the cat, these include bacterial granuloma, foreign body granuloma, squamous cell carcinoma and melanoma. A diagnosis of phaeohyphomycosis could be made on the basis of the pigmented mycelium found in the lesion. In tissue, the loose aggregates of either individual hyphae or yeast-like forms, often intracellular, cannot be confused with the compact mycelia aggregates of eumycotic black grain mycetomas. Moreover, the features of chromoblastomycosis are sufficiently distinctive to be separated from phaeohyphomycosis due to the presence in the former of several brown pigmented planate-dividing, rounded sclerotic bodies. In the case reported only one or two planate-dividing lievitiform cells were detected.
Fungal species and anatomical location of feline cutaneous phaeohyphomycosis

Histology must always be performed even if culture has yielded the fungus. In fact, if the culture has been obtained by a non-sterile specimen, the positivity should be interpreted as environmental airborne contamination and needs to be supported by direct microscopic evidence to be significant. Mycology is the only reliable means of distinguishing the several species of fungi involved in phaeohyphomycosis. The conidial morphology, the arrangement of conidia and the morphology of the conidiogenous cells are especially useful. Unfortunately, not all cases described in literature were supported by culture isolation. In our case the culture yielded C bantiana, one of the pathogenic species belonging to phaeohyphomycosis. Among the genus Chladophyalophora, C bantiana shows a marked neurotropism if compared to other fungi. In humans the cerebral form of phaeohyphomycosis due to C bantiana seems to derive from spore inhalation. Nasal localisation therefore should represent the entrance route for severe and often fatal cerebral infections.
Phaeohyphomycoses are characterised by a recrudescent clinical course and are refractory to many anti-fungal drugs. For these reasons, the aetiological diagnosis is necessary. Successful therapy relies on wide surgical excision of affected areas or anti-fungal chemotherapy in non-resectable cases, nevertheless recurrence at the same or new site(s) are common. Chemotherapy alone may yield variable results even within strains of a single species; moreover in vitro susceptibility tests have no predictable value (Matsumoto & Ajello 1998). In the case reported, the use of oral itraconazole was promising as it was effective in reducing the size of the mass but it had to be withdrawn because of gastrointestinal toxicity. Side-effects have been reported in cats, they seem to be dose-dependent and are usually resolved after drug withdrawal (Scott et al 2001). The second relapsing event, due to discontinuing drug administration underlines the importance of long-term therapy in avoiding re-occurrences. Identification of the fungus is relevant for cases managed by chemotherapy, but is also useful in cases treated by surgery since the type of the fungus, mostly in immunocompromised subjects, will influence the prognosis. Several treatment regimens have been employed for phaeohyphomycosis in animals by different authors. Recently, de Hoog et al (2000) classified human chromoblastomycosis according to morphology and severity of the lesions and found that only 44% of cases graded as severe were cured clinically and mycologically with itraconazole, while most of the patients having mild and moderate lesions achieved long-lasting cures with the same drug. No extensive clinical trials are available in veterinary medicine allowing us to correlate clinical and histological parameters with the response to therapy. Extension of the lesion, fibrosis and oedema might be considered as factors influencing anti-fungal therapy.
In the last decade phaeohyphomycosis has becoming one of the emerging human infections since fungi never previously associated with infection were found unexpectedly as pathogenic (Matsumoto & Ajello 1998). Despite the fact that most of the reported cases of human cerebral phaeohyphomycosis have occurred in immunocompetent patients, immunocompromise is considered by other authors as a risk factor (Jacyk et al 1997, Osiyemi et al 2001). In veterinary literature cases of phaeohyphonycosis are reported in cats in which the infection have been supported by immunocompromise (McKenzie et al 1984, Fuchs et al 1996). In the case described herein the cat was FIV - and FeLV-negative, immune function was not tested and other systemic diseases were not apparent, therefore the hypothesis of wound contamination is the most likely.
