Abstract
Histological examination was performed in 123 cats with primary nasal and paranasal sinus tumours; 117 had undergone surgical biopsy and six necropsy. Special stains and immunohistochemistry were performed on poorly differentiated cases. Ninety-two percent (113/123) of the tumours were malignant. There was an increased risk for old cats (mean age of 10.9 years), and a male predilection (59% males). Clinical signs and breeds varied with the histological type of tumour. Thirty-nine percent (48/123) of the cases presented with nasal discharge, 21% (26/123) with dyspnea, 20% (24/123) with facial swelling, and 15% (19/123) with epistaxis. Forty-three percent (53/123) of the tumours were of epithelial origin. Adenocarcinomas (18/53) and squamous cell carcinomas (17/53) were the most common epithelial tumours. Fifty percent (26/53) of the epithelial tumours originated from the pseudo-stratified respiratory epithelium of the nasal cavity, 28% (15/53) from the stratified squamous epithelium of the vestibule, 9% (5/53) from olfactory epithelium, 9% (5/53) from submucosal glands and 4% (2/53) from minor salivary glands. Malignant lymphoma (35/123) was the most common tumour. Seventy-one percent (25/35) of the malignant lymphomas were B-cell tumours and 29% (10/35) were T-cell tumours. Six cases of malignant lymphomas were proved to be epitheliotropic T-cell lymphomas. This is the first report of a primary nasal epitheliotropic T-cell lymphoma in cats.
Tumours of the nasal cavity and sinuses are rare in cats. Early studies indicated that feline nasal and paranasal tumours comprised approximately 1% of all feline tumours reported (Moulton 1990). A more recent study indicated an increase in the number to about 8.4% (Cox et al 1991). The increased percentage of these tumours may be due to inclusion of tumours of the nasal planum, or simply an increase in the number of these tumours. Up to 90% of the feline nasal and paranasal sinus tumours have been reported to be malignant (Cox et al 1991). Most of the tumours are locally invasive, and distant metastasis is rare (Legendre et al 1983, Moulton 1990).
Studies on clinicopathological features, histomorphological description and classification of feline nasal and paranasal sinus tumours are very limited. In literature, mostly case reports are described, and classifications are based mainly on systems used for dogs (Patnaik 1983). The merits of these classification systems are questionable, and this has caused some inconsistencies, because most reports were based on a few cases (Cox et al 1991, O'Brein et al 1996, Patnaik 1983). Very little effort has been expended on diagnostic immunohistochemistry of feline nasal and paranasal tumours.
In most studies tumours of epithelial origin, and specifically adenocarcinomas predominated. Malignant lymphoma was the most predominant non-epithelial tumour, and is more common in cats than in dogs (Levy & Ford 1994). Diagnosis of feline nasal and sinus tumours is difficult because of the location. A presumptive diagnosis can be reached by blood count, chemical profile, urinalysis, coagulation disorder screening and radiography on animals suspected of nasal and paranasal sinus tumours from clinical signs. However, for a definitive diagnosis, histopathological examination and diagnostic immunohistochemistry of biopsy specimens obtained from nasal cavity is needed.
The aim of the present study was to establish the clinicopathological features of feline nasal and paranasal sinus tumours, and give a descriptive histomorphology and diagnostic immunohistochemistry of the tumours.
Materials and methods
Case material
One hundred and twenty three cases with diagnosis of tumours of the nasal cavity and para-nasal sinuses were retrieved from computerised data of the Department of Pathology, Faculty of Veterinary Medicine, University of Utrecht, between 1993 and 2000. Age, sex, breed, clinical signs, feline leukaemia virus infection status and gross pathological descriptions were obtained from the records. Tumours arising externally from the epithelium of the nasal planum were not included.
Histopathology
Two pathologists re-evaluated haematoxylin and eosin stained sections by routine microscopy to confirm the diagnosis, and gave a morphological description. The distribution patterns, and cytological features of cells were recorded. Special stains like periodic acid-Schiff (PAS), toluidine blue and alkaline Congo red were done when required. Histological classification was based on the major component in each tumour. The World Health Organisation (WHO) system of classification for tumours of the respiratory system of domestic animals (Dungworth et al 1999) was used for classifying these tumours in cats. Immunohistochemistry was done on poorly differentiated tumours, all round cell tumours and spindle cell tumours to confirm the diagnosis.
Immunohistochemistry
Unstained sections of the tumours were deparaffinised and stained using the avidin-biotin complex method described before (Hsu et al 1981). Table 1 lists the antibodies used in this study, their cell specificity, tumours stained and the specific dilutions. Negative controls included substituting the primary antibody with non-related sera. Known positive controls were included for each marker. Endogenous peroxidase activity was blocked by immersing the slides for 20 min in 3% solution of H2O2 made in methanol. The slides were preincubated with 10% normal horse serum for 30 min before incubation with primary antibody. The biotinylated horse antimouse or horse antirabbit secondary antisera and avidin-biotin-peroxidase were incubated for 30 min. The immunolabelling were revealed by an enzyme-substrate reaction in which 3, 3′ diaminobenzidine was used as a chromogen. Finally, Mayer's haematoxylin was used as a counterstain.
Antibodies, cell types and tumour types evaluated in feline nasal and paranasal sinus tumours

Results
Clinical signs recorded in 123 cats with nasal and paranasal sinus tumours are shown in Table 2. The most common clinical signs were nasal discharge (48/123, 39%), dyspnea (26/123, 21%), facial swelling (24/123, 20%), and epistaxis (19/123, 15%). Epistaxis was more common in epithelial tumours. Seizures were not characteristic of any other tumours besides olfactory neuroblastomas. Both cases of olfactory neuroblastomas encountered had seizures, probably resulting from tumour extension into the brain. Submandibular lymph node swelling was seen in 21 cases, but metastasis was not evident in these lymph nodes.
Clinical signs based on morphology in 123 cases feline sinus and paranasal tumours

Histomorphological classification, sex and age distribution of the 123 cases of feline nasal and paranasal sinus tumours is shown in Table 3. Histopathological findings showed that 91.9% (113/123) of the tumours were malignant. Tumours of epithelial origin composed 43.1% (53/123) of the tumours. Adenocarcinomas (18/53) and squamous cell carcinomas (17/123) were the most common epithelial tumours. The origin of the different types of epithelial tumours is given in Table 4. One-half of the epithelial tumours (26/53, 49.1%) originated from the pseudo-stratified respiratory epithelium and 28.3% (15/53) were from the stratified squamous epithelium of the vestibule, 9.4% (5/53) from the olfactory epithelium. The remaining epithelial tumours were from submucosal glands (5/53, 9.4%), and minor salivary (2/53, 3.8%) with no evidence of surface epithelium involvement. Tumours of non-epithelial origin composed of 56.9% (70/123) of the tumours. One-half of the non-epithelial tumours (35/70, 50%) were malignant lymphomas.
Histomorphological classification, sex and age distribution in 123 cases of feline nasal and paranasal sinus tumours

Feline nasal and paranasal epithelial tumours: origin

The mean age of cats with tumours according to the histological types ranged from 8.7 to 15.2 years with an overall mean of 10.9 years. Tumours of epithelial origin had a higher mean (12.8 years) than those of non-epithelial origin (8.8) years. One cat with a fibroma was 16 weeks old at presentation. There was sex predominance with 59.3% (73/12) males affected. Male cats predominated in both epithelial (32/53, 60%) and non-epithelial (41/70, 58.5) tumours. Breed distribution of 123 cats with nasal and paranasal sinus tumours is given in Table 5. As can be observed from this table, there seemed to be a variation in number of epithelial vs non-epithelial tumours according to breed, but because of small numbers cats of each breed, these findings may not be of significance.
Breed distribution based on histomorphology in 123 cats with nasal and paranasal sinus tumours.
Epithelial tumours
Adenoma
These cases represented 19.4% (5/53) of the epithelial tumours. They consisted of a proliferation of serous or mucous glands. In all the cases, the tumour mass was well demarcated and encapsulated, showing expansive growth. In one case the glandular elements were set in PAS positive mucoid stromal material, with this material forming the bulk of the tumour mass. The surface respiratory epithelium was intact, and these tumours seemed to arise from the submucosal glands.
Basal cell tumours
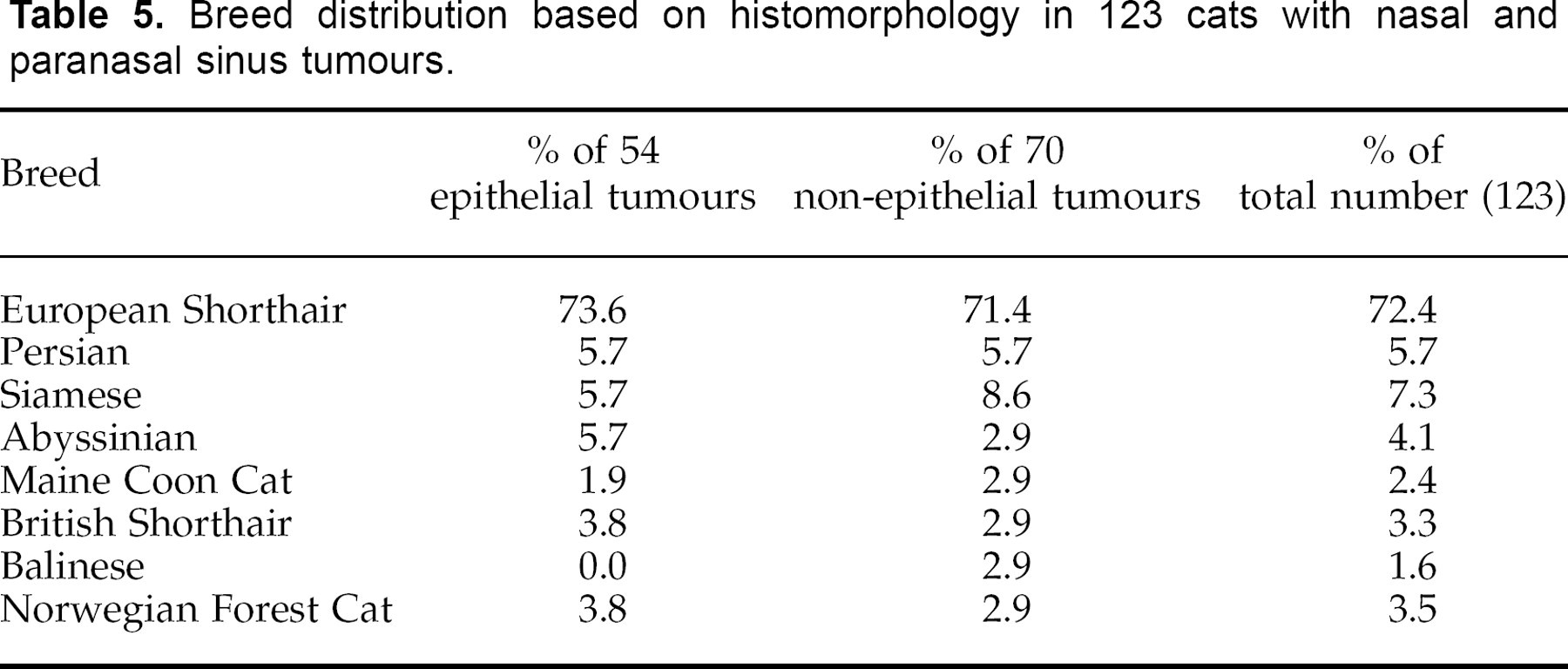
Basal cell tumours (two cases) comprised 1.6% of the nasal and paranasal sinus tumours. Both cases were from the vestibule, and of the solid type. In both cases, the tumour mass was well circumscribed, but not encapsulated. The tumour cells formed multiple, solid nests of tightly packed moderately anaplastic and epithelial cells, separated by a fine fibrovascular stroma (Fig 1). The tumour cells had round nuclei, finely clumped chromatin and eosinophilic cytoplasm. In spite of poorly differentiated and moderately anaplastic appearance of the basal cells, feline basal cell tumours are considered benign (Diters & Walsh 1984).

Nasal cavity. Solid basal cell tumour with multiple nests of epithelial cells separated by a fine fibrovascular stroma. HE×200.
Adenocarcinoma
Adenocarcinomas represented 34.0% (18/53) of the epithelial tumours. Diagnosis of adenocarcinoma was reached when there was invasion in the submucosa or blood vessels, and surrounding soft and skeletal tissues. Different morphological types of adenocarcinomas were observed: acinous, cystic, mucinous and papillary.
Acinous adenocarcinoma (three cases): in all the cases, the tumour consisted of moderately cellular masses forming acini and ducts similar to minor salivary glands. In two cases the conchal bone was invaded. Some of the glandular elements were dilated, and contained mucoid material positive with PAS staining. The tumour cells were columnar or cuboidal with distinct cell borders. The nuclei were centrally or basally located, with single prominent nucleoli. In one case there was anisocytosis and karyomegaly. There was a diffuse infiltration by lymphocytes. In all the cases the surface epithelium was intact, and these tumours seem to arise from the minor salivary gland of this area.
Cystic adenocarcinomas (three cases): these tumours consisted of dilated tubular and variously sized cystic structures containing PAS positive mucin. Tumour cells lining the tubulo-cystic structures were cuboidal or low-columnar with distinct cell borders and eosinophilic cytoplasm. Mitotic figures were rare. There was a diffuse infiltration by neutrophils in all the cases. In the three cases there was an overlapping layer of normal epithelium, indicating a submucosal glandular origin.
Mucinous adenocarcinoma (four cases): this tumour consisted of a moderately cellular infiltrating growth composed of columnar cells arranged in tubules, and occasionally papillary structures and ducts separated by a thin connective tissue stroma. The tubulopapillary structures contained varying amounts of intra- and extracellular mucin. The tumour cells were well-differentiated, cuboidal cells with eosinophilic cytoplasm. In two cases there was no transition from the surface epithelium, suggesting submucosal glands as the site of origin. In two other cases, the tumours were arising from the adjoining surface olfactory epithelium.
Papillary adenocarcinomas (eight cases): these tumours were characterised by a proliferation of columnar cells forming papillary foldings separated by a thin fibrovascular stroma. The cells were often ciliated. The nuclei were large and vesiculated. Mitotic figures were uncommon. In all the cases of papillary adenocarcinoma, the tumour was arising from the surface respiratory epithelium.
Squamous cell carcinoma
Squamous cell carcinoma accounted 17 of the 53 epithelial tumours. Fifteen of the tumours were mostly located in the nasal vestibule, but also involving deeper structures and two were located in the caudal part of the nasal cavity. Two different morphological types of squamous cell carcinomas were observed: the keratinizing squamous cell carcinoma and the non-keratinizing squamous cell carcinoma. The latter has been referred as transitional cell carcinoma. Twelve cases were well-differentiated keratinizing squamous cell carcinomas and five were poorly differentiated non-keratinizing squamous cell carcinomas. The keratinizing tumour consisted of solid cellular infiltrative masses, composed of polygonal to spindle shaped cells arranged in lobules, or cords separated by thin connective tissue stroma. In the centre of some lobules keratin pearls were observed. The tumour cells had variously distinct cell borders, and were occasionally joined by intercellular bridges. The cells had a moderate amount of cytoplasm, and some contained intracellular keratin. Mitotic rate was variable, from 1–6 per high power field. The non-keratinizing form consisted of nests of cells composed of polygonal or spindle shaped cells. Cellular and nuclear pleomorphism was present in addition to several mitotic figures. In two cases transition from the normal respiratory epithelium was distinct, suggesting origin from this area.
Undifferentiated carcinoma
Nine of the 53 cases of the epithelial tumours were undifferentiated carcinomas. The tumours consisted of highly cellular solid masses of round to pleomorphic cells without evidence of glandular or squamous differentiation. The neoplastic cells had scant, pale eosinophilic vacuolated cytoplasm, and large oval nuclei with single nucleoli. These tumours had a high mitotic rate, about 5 per high power field, and extensive necrosis, indicating high malignancy.
Olfactory neuroblastoma
Two cases were olfactory neuroblastomas. The initial diagnosis for both tumours was adenocarcinoma because of the tubule-like formations. Tumour cells were arranged in lobules separated by fibrovascular stroma. There was palisading of tumour cells around fibrovascular stroma (Fig 2), with formation of pseudorosettes (Fig 3). The tumour cells were small, round or oval with scant cytoplasm. Sometimes, a mixture of small and large columnar cells was present. Mitotic figures were rare. Immunohistochemical staining of both tumours showed positive S-100 staining of many cells within the tumour (Fig 4). Immunostaining for NSE showed positive staining of few scattered cells within the tumour. Staining for cytokeratin was positive in some of the cells forming rosettes. Immunostaining for chromogranin and synaptophysin was not done.

Nasal cavity. Olfactory neuroblastoma. Tumour cells are arranged in a palisading fashion around a central blood vessel. HE×200.
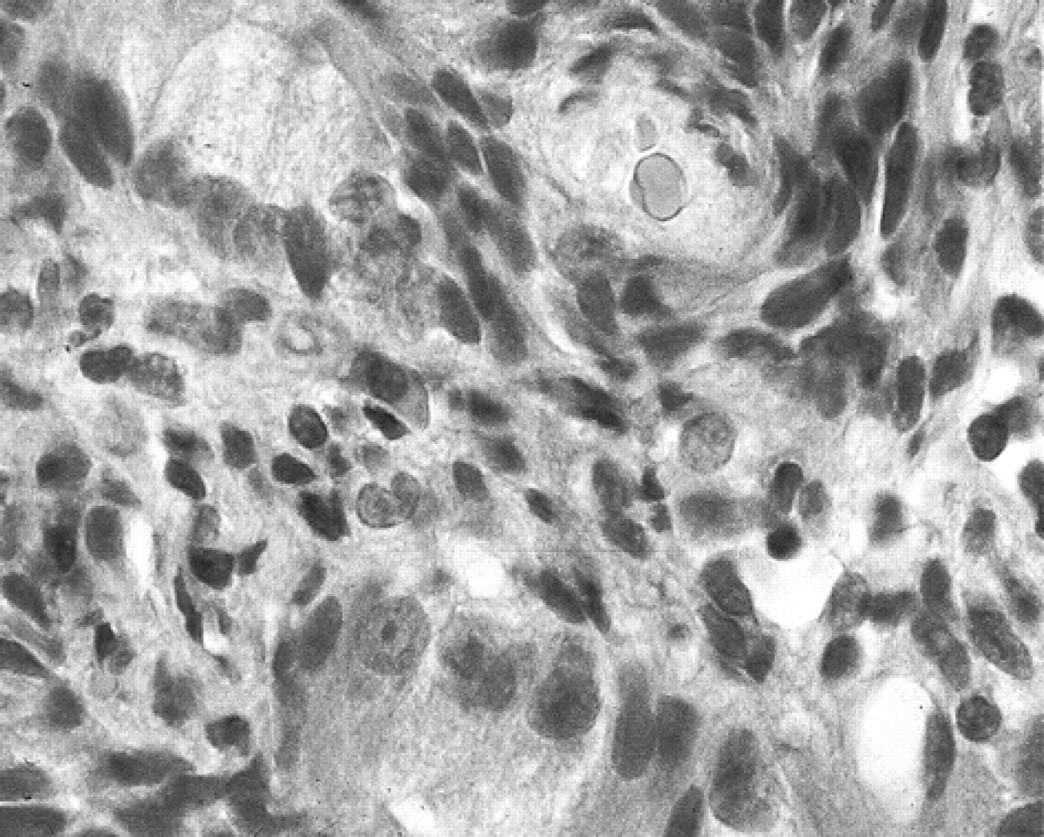
Nasal cavity. Olfactory neuroblastoma. Tumour cells forming pseudorosettes. HE×400

Nasal cavity. Olfactory neuroblastoma. S-100 immmunostaining, showing positive staining of some cells within the tumour. Avidin-biotin peroxidase method, diaminobenzidine chromogen, Mayer's haematoxylin counter. ×200.
Non-epithelial tumours
Malignant lymphoma
Malignant lymphoma (35 cases) was the most common nasal and paranasal sinus tumour, and was present in 28.5% of all the cases, and 50% of the non-epithelial tumours. In all the cases, there was no record of feline leukaemia virus infection. Overall, 25/35 cases proved to be B-cell lymphomas in which the cells consistently expressed BLA and 10/35 cases were T-cell lymphomas in which the cells expressed CD3. In 33/35 cases, there was a diffuse infiltration of the submucosa by monomorphic sheets of atypical round cells. In 2/35 cases there was follicular proliferation rather than diffuse infiltration. The tumour cells were small to medium sized with hyperchromatic nuclei, or large with histiocytic appearance. Occasionally, an inflammatory infiltrate composed of mainly neutrophils was present within the tumour.
In 6/35 cases of the malignant lymphomas the lymphoid infiltrate had tropism for the respiratory epithelium, and stratified squamous epithelium of the vestibule. Epitheliotropic malignant lymphocytes were diffusely distributed within the epithelium. Immunohistochemical staining of the six epitheliotropic lymphomas demonstrated that the neoplastic lymphocytes were negative for the B-cell marker, BLA, and positive for the T-cell marker, CD-3 (Fig 5), indicating a T-cell lineage.
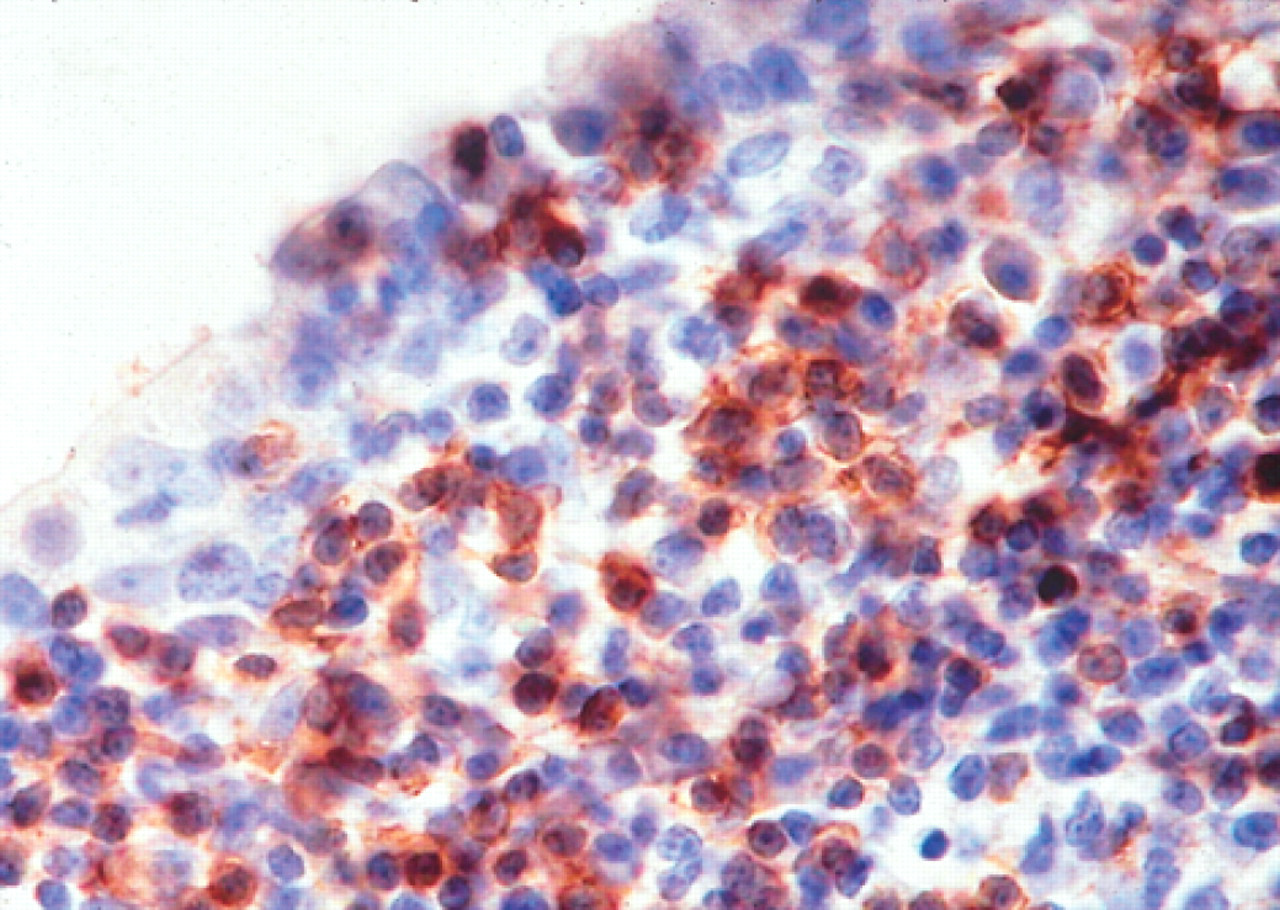
Nasal cavity. Epitheliotropic malignant lymphoma. Neoplastic lymphocytes, with positive immunoreaction to CD-3 antigen, infiltrating the respiratory epithelium. Note the intraepithelial lymphocytes. Avidin-biotin peroxidase method, diaminobenzidine chromogen, Mayer's haematoxylin counter stain. ×400.
Plasmacytoma
Five cases of plasmacytomas accounted for 4.1% of the tumours. The tumour cells were uniform and round, with eccentric nuclei and abundant cytoplasm. These tumours were positive for lambda light chain and negative for CD-3 when immunohistochemistry was performed.
Mastocytoma
Three (2.4%) mastocytomas were diagnosed. The three cases consisted of round cells with centrally located nuclei and abundant granules in the cytoplasm. The granules were demonstrated with toluidine blue stain. The tumour cells were negative for CD-3, BLA, and lambda light chain.
Fibrosarcoma and fibroma
Fibrosarcoma (10 cases) was the second most common non-epithelial tumour and was present in 8.1% of the cases. The tumours consisted of highly cellular infiltrative masses composed of poorly defined spindle or ovoid cells. Tumour cells consisted of large elongated nuclei with one or more nucleoli. The cytoplasm was difficult to distinguish from stroma. Only one fibroma was recorded. This tumour was well demarcated by a fibrous capsule and consisted of whorls and bundles of fibroblasts and collagen fibres.
Osteosarcoma and chondrosarcoma
Two osteosarcomas and three chondrosarcomas were diagnosed. Tumours were classified as osteosarcomas when osteoid or bone production was evident to differentiate them from chondrosarcoma. The osteosarcomas were composed of anaplastic osteoblasts producing bone matrix and numerous tumour giant cells. The tumour cells had large hyperchromatic nuclei. The mitotic rate was high. The chondrosarcomas were composed of irregular lobules of hyaline cartilage consisting of chondrocytes with a lot of fibrous stroma. Multinucleated cells were also present.
Haemangiosarcoma
Only one case of haemangiosarcoma was recorded. This tumour consisted of endothelial cells forming small vascular clefts, some containing blood cells. The cells varied in shape, mostly elongated and ovoid. Mitotic figures were very common. The tumour was positive for factor VIII-related antigen when immunohistochemistry was performed.
Melanoma
Five melanomas (4.0%) occurred in the vestibule. Three of the tumours were of the epitheloid cell type, and two were of the spindle cell type. The epitheloid type consisted of large polygonal cells forming clusters (Fig 6). The cytoplasm was pinkish-brown from the presence of melanin granules. The spindle cell type consisted of spindle shaped cells with much less cytoplasm than the epitheloid cells. Scattered deposits of melanin were present. Immunostaining for S-100 and NSE was positive for all the cases.

Nasal cavity. Melanoma. HE×200.
Discussion
This study comprises the largest feline nasal and paranasal tumour case series to be characterised histomorphologically and immunohistochemically. Previous reports of such studies were based on very small numbers of cases, and diagnostic immunohistochemistry was not done to confirm the diagnosis (Bright & Bojrab 1976, Evans & Hendrick 1989, Patnaik 1983).
Tumours of the nasal and paranasal sinuses can basically arise from any of the cells found in that region. Many different types of cells can be involved and before discussing these tumours it is worth reviewing the normal cell components of the region. The rostral cutaneous part of the nasal cavity (vestibule) is lined by a thick keratinised squamous epithelium. At mid-vestibule the epithelium is thinner and non-keratinised. The submucosa of the vestibule contains vessels, numerous free cells including mast cells, plasma cells, lymphocytes, macrophages and granulo-cytes. Bundles of collagen fibres, large blood vessels and serous glands are located deep in the submucosa. The caudal two thirds of the nasal cavity, and paranasal sinuses, except the olfactory region are lined by a pseudo-stratified epithelium, inter-pressed with goblet cells. In older animals there is metaplasia of some areas to squamous epithelium (Patnaik 1983). The olfactory epithelium comprises the dorso-caudal portion of the nasal cavity, and contains neurosecretory cells. The supporting tissue, bone and cartilage must also be considered as sites of origin of nasal and paranasal sinus tumours.
The mean age of the cats with tumours was 10.9 years. In previous studies of feline nasal and paranasal sinus tumours the mean age ranged from 8 to 10 years (Cox et al 1991, O'Brein et al 1996). In this study a fibroma in a 16 weeks old cat was recorded. In previous studies tumours in cats as young as 2 months have been reported (Cox & Powers 1989, O'Brein et al 1996). A male predominance (1.48:1) was observed in the present study, but no correction was made for sex distribution of total cases. However, castrated male dogs have been previously reported to be predisposed to intranasal tumours (Cox et al 1991), suggesting the male sex hormones to play a role in development of these tumours.
Nasal discharge, dyspnea, facial swelling and epistaxis were the most common signs recorded in this study. These signs are not specific to tumours because nasal and paranasal sinus inflammatory conditions can give the same signs. A definitive diagnosis requires histological or cytological examination. Clinical signs seen in cats in this study are similar to those described in the dog (Patnaik 1989). In this study nasal discharge (39%) was the most common clinical sign. In dogs the most common clinical signs were epistaxis (40%), and facial swelling (40%) (Patnaik 1989). The clinical signs varied according to histological type of tumour. For example, epistaxis was common in dogs with epithelial tumours, and seizures were confined to two cats with olfactory neuroblastomas.
The predominance of specific types of tumours varies in different studies. This may be due to the fact that some researchers discuss all types of nasal and paranasal sinus tumours together (Madewell et al 1976, Moulton 1990), and others separate tumours arising externally from epithelium of nasal planum from those arising from intranasal structures (nasal cavity and paranasal sinuses) (Carlisle & Gould 1982, Evans & Hendrick 1989, Legendre et al 1983). Prevalence of epithelial vs non-epithelial tumours varies in different studies (Bright & Bojrab 1976, Levy & Ford 1994, Madewell et al 1976). In dogs, epithelial tumours predominate (Patnaik 1989).
Feline basal cell tumours are benign cutaneous tumours, which occur with equal frequency in any body region (Diters & Walsh 1984). They originate from uncommitted basal reserve cells of the epidermis and adnexia. In this study, both cases were from the vestibule. Basal cell tumours have not been mentioned in previous reports of nasal and paranasal sinus tumours.
Squamous cell carcinomas are locally invasive and 11% of these tumours can give metastatic spreading (Moulton 1990). By the time of evaluation the tumour might be very extensive that it is difficult to determine by histopathology its origin (Patnaik 1983). After excluding tumours that appeared to arise from the nasal planum, the occurrence of these tumours was 10% of all nasal tumours, which is high compared to previous studies (Cox et al 1991). The difference might be due to inclusion of tumours arising from the nasal vestibule, which were considered as part of the nasal cavity in the present study. Since the vestibule is lined by a stratified squamous epithelium, squamous cell carcinomas can arise from this site. In addition, with age areas of squamous epithelium replace the normal pseudo-stratified ciliated epithelium of the respiratory cavity (Patnaik 1983), suggesting that squamous cell carcinomas can arise from these areas as well.
Adenocarcinomas had the highest occurrence of the tumours of epithelial origin. This is in agreement with previous studies (Patnaik 1983). These tumours seem to have a high occurrence because they can arise from surface ciliated respiratory epithelium covering the whole nasal cavity, as evidenced by a case of a tubulopapillary adenocarcinoma with ciliated neoplastic cells. They can also arise from submucosal glandular elements or minor salivary glands, as seen from many different morphological types: acinous, cystic, mucinous and papillary.
Olfactory neuroblastoma is an uncommon malignant tumour of neural crest origin arising from the olfactory sensory epithelium of the nasal cavity. Cases of this tumour have been reported for a dog (Dahme et al 1985), a horse (Loupal & Mikula 1985), and a heifer (Anderson & Cordy 1981). In cats, two cases have been reported on revaluation of 32 tumours (Cox & Powers 1989). Histologically the tumour is difficult to differentiate from other small, round cell tumours of the nasal cavity, such as anaplastic carcinoma, lymphosarcoma, melanoma, or poorly differentiated adenocarcinoma. The two previously reported cases of olfactory neuroblastomas in cats (Cox & Powers 1989) were initially diagnosed as anaplastic carcinoma and adenocarcinoma. The initial diagnosis for both of the tumours reported here was adenocarcinoma. In both tumours reported, all the histological and immunohistochemistry features consistent with olfactory neuroblastoma were present. These results suggest that most olfactory neuroblastomas are usually missed in the initial diagnosis. Both tumours showed local invasion into paranasal cavity and surrounding structures. However, no metastases were seen. In humans, olfactory neuroblastomas metastasise widely by both haematogenous and lymphatic routes, with the neck being the most common site. No other tumour showed this aggressive behaviour. Therefore, proper diagnosis of olfactory neuroblastoma is necessary since this tumour appears to be more aggressive than anaplastic carcinomas and adenocarcinomas.
Malignant lymphoma is the most common haemopoetic tumour of the cat, accounting up to 50% of all feline tumours (Hardy 1981). In our study malignant lymphomas had the highest occurrence of all nasal and paranasal sinus tumours. This agrees with previous studies, although carried out on few cases (O'Brein et al 1996, Patnaik 1983). In our study, the proportion of B-cell lymphomas exceeded that of T-cell lymphomas. In previous studies no attempts were made to characterise the phenotype of nasal and paranasal malignant lymphomas. However, our findings are in good agreement with a feline case series concerning alimentary tract lymphosarcoma, where the B-cell phenotype predominated (Jackson et al 1996). Six cases of malignant lymphoma were epitheliotropic. Epitheliotropism refers to localisation of malignant lymphocytes in the epithelium. Cutaneous epitheliotropic lymphomas in humans are of T-cell origin. Epitheliotropism in lymphomas, mimicking mycosis fungoides in humans has been reported in cutaneous lymphomas in the cat (Caciolo et al 1984) and dog (DeBoer et al 1990). Mucosal associated lymphoid tissue epitheliotropic lymphomas are very rare in animals. Epitheliotropism has been described in canine primary gastrointestinal lymphomas (French et al 1996, Steinberg et al 1995), and urinary bladder lymphoma (Maiolino & DeVico 2000). Epitheliotropic gastrointestinal tract lymphoma in a cat has only recently been reported (Krecic & Black 2000). To our knowledge, epitheliotropism in primary nasal and paranasal tumours in cats has not been reported previously. The demonstration of T-cell phenotype using anti-CD-3 allowed for definitive diagnosis of primary, nasal epitheliotropic T-cell lymphoma.
Melanomas are common in dogs, grey horses and in some lines of miniature swine, and rare in cats. Melanomas are usually cutaneous tumours, but can occur whenever melanocytes are present. In this study, there was a high occurrence of melanomas (4.0%) compared to an occurrence of 0.8% of feline cutaneous tumours. In a previous report, feline melanomas were found in the lateral abdominal skin, and shoulder (Miller et al 1981). No cases of melanomas in the vestibule have been reported before.
In conclusion, our results based on a large case pool, indicates that cats are subject to a variety of nasal and paranasal sinus tumours, but only five tumours (adenocarcinoma, squamous cell carcinoma, anaplastic carcinoma, malignant lymphoma and fibrosarcoma) are common. Definitive diagnosis of most of tumours requires immunohistochemical staining. Correct diagnosis of nasal and paranasal sinus tumours from biopsy samples is important, since most tumours appear to have different prognosis. In particular, olfactory neuroblastomas must be differentiated from adenocarcinomas and anaplastic carcinomas, as these seem to be more aggressive than other tumours. Nasal and paranasal sinus tumours are mainly a problem of old cats, with a male predilection.
Footnotes
Acknowledgements
We wish to thank Mr F van Mil for his technical assistance.
