Abstract
Chylothorax is a complex disease with many identified underlying causes including cardiac disease, mediastinal masses, heartworm disease and trauma. Management of this disease should be directed at identifying the cause, if possible, and treating the underlying disorder. In cats with idiopathic chylothorax, medical management is recommended initially because the condition may resolve spontaneously. Owners should be made aware of the potential development of fibrosing pleuritis in affected cats. When medical management is impractical or unsuccessful, surgical intervention should be considered. Surgical options include mesenteric lymphangiography and thoracic duct ligation, pericardiectomy, omentalisation, passive pleuroperitoneal shunting, active pleuroperitoneal or pleurovenous shunting, and pleurodesis. Of these, only thoracic duct ligation and pericardiectomy are preferred by the author because, if successful, the result is complete resolution of the chylothorax, thereby reducing the risk of developing fibrosing pleuritis. Omentalisation may be beneficial in some animals as adjuvant therapy, but this procedure may still allow fibrosing pleuritis to occur. Until the aetiology of the effusion in cats with idiopathic chylothorax is understood, the treatment success rate will be less than ideal. Future research needs to be directed at determining the pathophysiologic mechanisms underlying this disease in cats.
Management of animals with chylothorax has been greatly refined since the initial report of its surgical treatment in three dogs and one cat in 1958 (Patterson & Munson 1958). However, our ability to treat many affected animals effectively has been hindered by a lack of understanding of the aetiology of this devastating disease. As appropriate treatment of affected animals depends foremost on defining the aetiology, a discussion of the various underlying conditions associated with chylothorax is warranted. This will be followed by a discussion of the surgical management of this disease in cats.
Aetiology
Chylothorax was previously thought to be due to thoracic duct rupture secondary to trauma; however, this is now known to be a rare cause of chylothorax in animals. Other causes include mediastinal lymphosarcoma (Forrester et al 1991), cardiomyopathy (Birchard et al 1986) (particularly secondary to hyperthyroidism), pericardial effusion, congenital cardiac or lymphatic abnormalities (Suter & Green 1971, Fossum et al 1994), and heartworm infection (Donahoe et al 1974, Birchard & Bilbrey 1990). In the majority of cats, despite extensive diagnostic work-ups, the underlying aetiology is undermined (idiopathic chylothorax) (Fossum et al 1986, 1991, Fossum 1993). As the treatment of this disease varies considerably depending on the underlying aetiology, it is imperative that clinicians identify concurrent disease processes prior to instituting definitive therapy.
Any disease that results in high venous pressures may cause chylothorax; cardiomyopathy, pericardial effusion, congenital cardiac abnormalities and heartworm disease have been associated with chylothorax in cats. Thus, a complete cardiac work-up is warranted in all cats with confirmed chylothorax. Treatment of cats with cardiomyopathy and chylothorax should be based primarily on palliation when necessary with thoracentesis, and improving cardiac output and decreasing venous pressures with appropriate drug therapy. Although heartworm infection is uncommon in cats, experimental infection with Dirofilaria immitis has been shown to result in chylothorax in a small number of cases (Donahoe et al 1974). Naturally occurring heart-worm disease has also been associated with chylothorax in a cat (Birchard & Bilbrey 1990). It is recommended that cats with chylothorax be screened for heartworm infection. If pericardial effusion is diagnosed, the underlying aetiology should be determined and pericardiectomy performed, if indicated.
If any anterior mediastinal mass is identified, a fine needle aspirate may be performed to determine the tumour or tissue type. Specific therapy (ie radiation therapy, chemotherapy, antifungal therapy, surgery) should then be instituted according to findings. In these animals, the chylous effusion is probably secondary to compression of the cranial vena cava by the mass, and shrinkage of the mass may result in resolution of the pleural fluid. For prognostic purposes, it is prudent to assess FeLV and FIV status in affected cats. Traumatic chylothorax probably occurs more commonly in animals with chylothorax than one would expect based on reported cases. However, in most of these animals, the thoracic duct heals spontaneously (Hodges et al 1993).
When no obvious underlying disorder can be found, the term ‘idiopathic’ chylothorax is used. Unfortunately, management of cats with idiopathic chylothorax is difficult because no highly effective treatment exists. Until the aetiology of chylothorax in these animals is understood, therapy will remain palliative and less than optimal in many instances. One possibility is that these animals have increased volumes of lymph being transported through the thoracic duct. These increased flows may occur secondary to abnormal right-sided venous pressures that cause much of the lymph, that would normally be transported from the liver into the venous system, to be shunted into the lymphatic system. Possibly, minimally elevated venous pressures, in association with other unknown factors, may be sufficient to substantially elevate lymphatic flows through the thoracic duct.
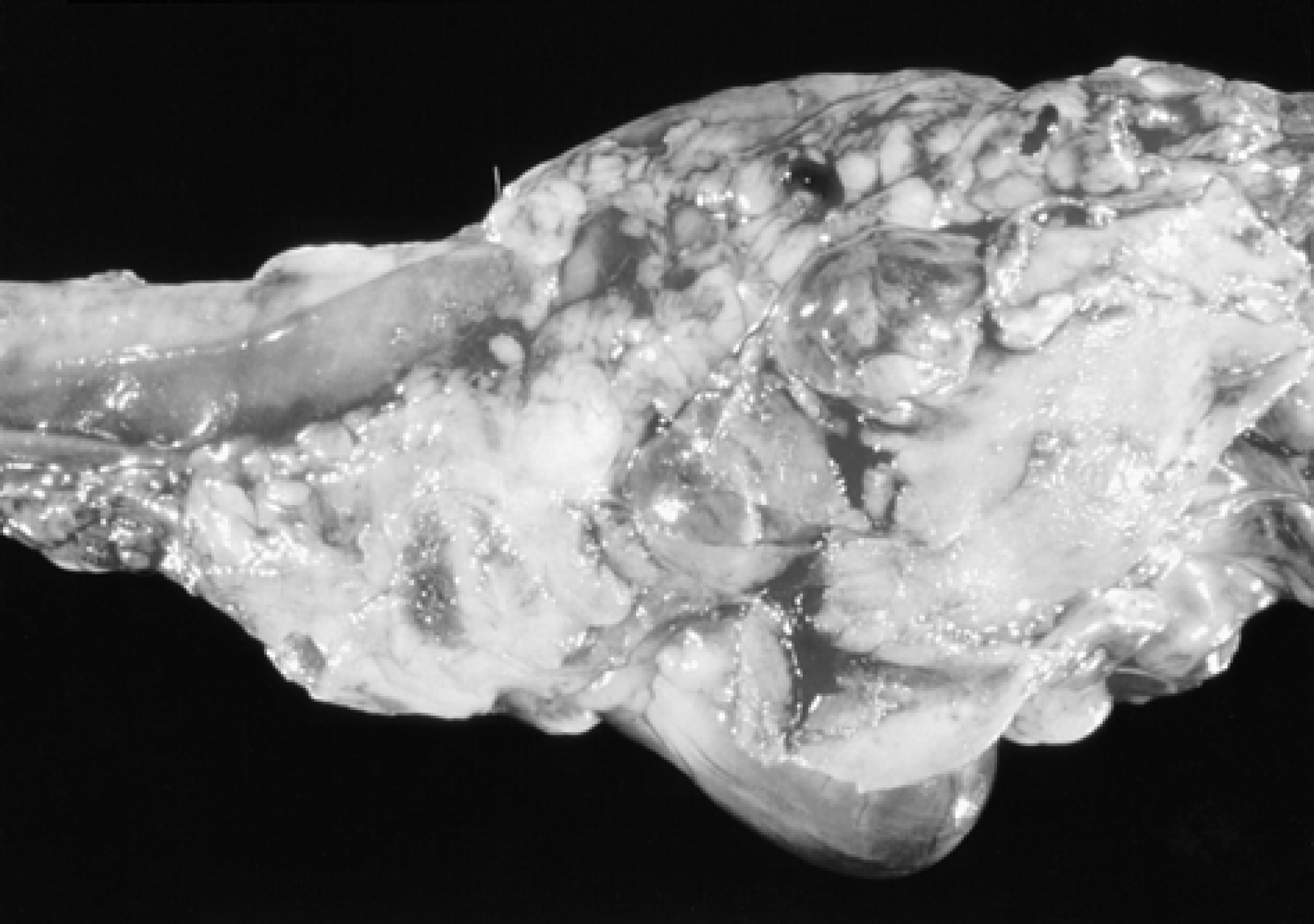
As the surgical options for treating this condition require a high level of expertise to be successful, and because the condition may spontaneously resolve with time, many cats are initially treated medically. This involves feeding a low-fat diet, performing needle thoracentesis as needed to relieve dyspnoea (not on a regular basis), and supplementing the cat with a benzopyrone (50–100 mg/kg, po, tid) (Thompson et al 1999). Owners should be warned of the potential development and grave prognosis associated with severe fibrosing pleuritis in cats with chronic chylothorax (Glennon et al 1987). Although the cause of the fibrosis is unknown, it apparently can develop subsequent to any prolonged exudative (ie chylothorax, pyothorax) or blood-stained effusion (haemothorax). In animals with fibrosing pleuritis, the pleura is thickened by diffuse fibrous tissue that restricts normal pulmonary expansion (Figs 1 and 2). Animals with fibrosing pleuritis may be misdiagnosed as having large amounts of pleural effusion, when in fact the pleuritis effusion is minimal (Fossum et al 1992). Radiographic evidence of pulmonary parenchyma that fails to re-expand after removal of pleural fluid should be considered possible evidence of atelectasis with associated fibrosis. Fibrosing pleuritis should also be considered in animals with persistent dyspnoea, yet minimal pleural fluid. Animals with fibrosing pleuritis are less likely to respond to medical or surgical treatments of chylothorax.

Photomicrograph of the lungs of a cat with fibrosing pleuritis associated with chylothorax. Notice the thickened pleura and the rounded appearance of the lung lobes.
Lateral view of lungs of a cat with chronic chylothorax. Notice the severe atelectasis and rounded appearance of the cranial lung lobes.
If medical management is being attempted, it is important to provide for the patient's nutritional and metabolic needs until the effusion resolves spontaneously (Willard et al 1991). Generally, meeting the animal's caloric needs by feeding a low-fat diet or a homemade diet is adequate. The author no longer advises supplementing the diet with low- or medium-chain triglycerides (MCT) because cats find these supplements to be unpalatable and their efficacy is questionable.
Surgical options
Surgical intervention is warranted in cats that do not have underlying disease and in whom medical management becomes impractical, such as in cats that require thoracentesis more frequently than once a week, or those in which repeat thoracentesis fails to relieve the dyspnoea. Surgical options in cases uncomplicated by severe fibrosing pleuritis include mesenteric lymphangiography and thoracic duct (TD) ligation (Fossum 1993, Kerpsack et al 1994), subtotal pericardiectomy (Fossum 2001), omentalisation (Williams & Niles 1999), passive pleuroperitoneal shunting (Peterson et al 1989), active pleuroperitoneal or pleurovenous shunting (Smeak et al 1987, Donner 1989), and pleurodesis (Laing & Norris 1986, Birchard et al 1989). Of these, only the first two (TD ligation and pericardiectomy) are recommended as first-line therapies by the author.
Thoracic duct ligation with mesenteric lymphangiography
Thoracic duct ligation is performed in cats from a left lateral intercostal thororocotomy or transdiaphragmatically. The mechanism by which TD ligation is purported to work is that following TD ligation, abdominal lymphaticovenous anastomoses form for the transport of chyle to the venous system. Therefore, chyle bypasses the thoracic duct and the effusion resolves. Unfortunately, TD ligation results in complete resolution of pleural effusion in only 50% of dogs operated on (Birchard et al 1988); in cats, the success rate is even less (≤ than 40%) (Harpster 1986, Fossum et al 1991). Advantages of TD ligation are that if it is successful, it results in complete resolution of pleural fluid (compared with palliative procedures described below), and may prevent fibrosing pleuritis from developing. The disadvantages include that operative time is long, which is problematic in debilitated cats, there is a high incidence of continued or recurrent chylous or non-chylous (from pulmonary lymphatics) effusion, and mesenteric lymphangiography is often difficult to perform in cats. Without mesenteric lymphangiography, complete ligation of the TD cannot be assured; however, an experimental paper assessing lymphangiography in cats suggested that this technique might not be uniformly successful in verifying complete ligation of the TD (Martin et al 1988). Additionally, some animals may form collateral lymphatics past the site of the ligature, and thus re-establish TD flow. If chyle flow is directed into the diaphragmatic lymphatics, chylothorax may continue or recur.
For lymphangiography, food is withheld for 12 h prior to surgery. Either the left side of the thorax and left abdomen, or just the abdomen if a midline celiotomy is being performed, is prepared for aseptic surgery. If a thoracic approach to the TD is being used, a left paracostal incision is made in order to exteriorise the cecum. Once the cecum has been exteriorised, a lymph node adjacent to the cecum is located. A small volume (0.5–1 ml) of methylene blue (USP 1%, American Quinine, Shirley, NY, USA) may be injected into the lymph node to increase visualisation of lymphatics. Repeated doses of methylene blue should be avoided due to the risk or inducing a Heinz body anaemia or renal failure (Osuna et al 1990). Careful dissection of the mesentery near this node allows large lymphatic vessels to be visualised and cannulated with a 22-gauge over-the-needle catheter (Kagan & Breznock 1979). Cannulation of this lymphatic is more difficult in the cat than in the dog because cats have more fat in their mesentery, and their lymphatics are significantly smaller. Two sutures (3–0 silk) are placed in the mesentery and used to secure the catheter and an attached piece of extension tubing in place (the ends of the suture can be looped over the hub of the extension tubing). An additional suture may be placed around the extension tubing and through a segment of intestine to prevent dislodgement of the catheter. A three-way stopcock is attached to the end of the extension tubing, and a water-soluble contrast agent is injected at a dosage of 1 ml/kg diluted with 0.5 ml/kg of saline. A lateral thoracic radiograph is taken while the last millimetre is being injected. This lymphangiogram can be used to help identify the number and location of branches of the TD, which need to be ligated, and it can be repeated following ligation to help determine the extent of lymphangiectasia present in the cranial thorax (Fig 3).
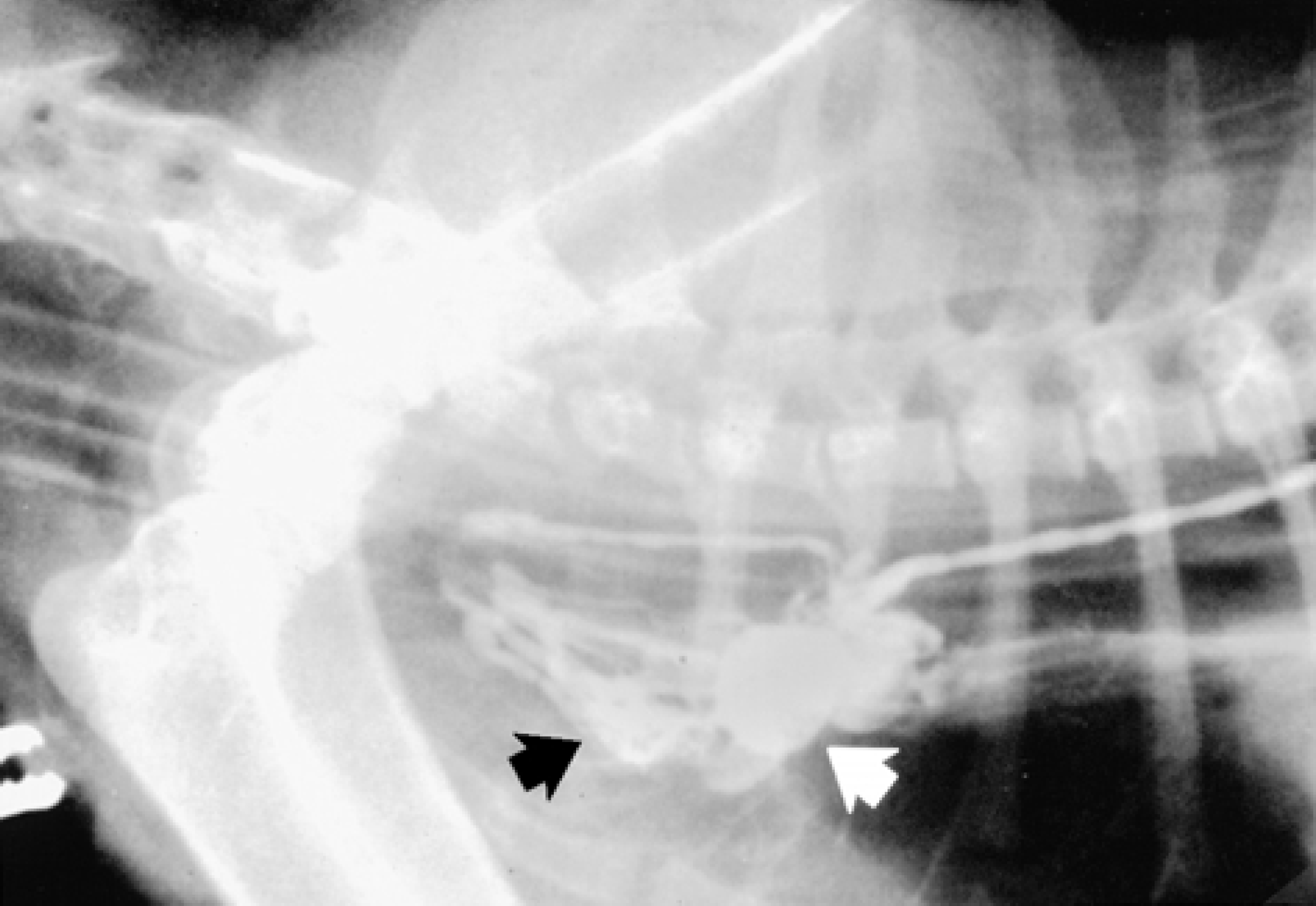
Lymphangiogram performed in a cat with chylothorax and thoracic lymphangiectasia. Note the multiple, dilated lymphatics near the entrance of the thoracic duct (arrow) into the venous system.
The thoracic duct is approached in the cat through a left caudal intercostal thoracotomy (eighth, nineth, or tenth intercostal space) or via an incision in the left diaphragm. Once the duct has been located, haemostatic clips can be used to ligate it. The advantage of using haemoclips (Edward Weck and Co. Inc., Research Triangle Park, NC, USA) is that they can be used as a reference point on subsequent radiographs if further ligation is necessary. However, the author prefers to also place a non-absorbable suture, such as silk, on the duct. Visualisation of the thoracic duct can be aided by injecting methylene blue into the lymphatic catheter (Fig 4). If a catheter is not placed, the dye can be injected into a mesenteric lymph node; however, this results in less than optimal TD filling.

Identification of the thoracic duct can be aided by injecting methylene blue into the lymphatic catheter, or directly into a mesenteric lymph node.
Pericardiectomy
Thickening of the pleura and pericardium occurs in animals with chylothorax. Although the role of the thickened pericardium in this disease is not well understood, it has been the observation of the author that some cats benefit from pericardiectomy. If the thickened pericardium causes even a slight elevation in venous pressures, the TD may see this as an obstruction with resultant formation of numerous lymphatics in the cranial thorax. These lymphatics may likely leak transmurrally (Haider et al 1987). Although experimental at this point, the author has had some success with pericardiectomy as the sole treatment of cats with chylothorax. Further studies and additional experience are necessary to recommend this technique in affected animals; however, it looks promising as sole therapy or as adjunctive to TD ligation in animals with chylothorax.
Omentalisation
Omentalisation has been reported as a technique to treat animals with chylothorax when other surgical treatments are not successful, or deemed impossible (Williams & Niles 1999). A fifth or sixth space intercostal thoracotomy is made to provide access to the cranial thorax. A paracostal incision is then made so that a dorsal omental pedicle flap can be raised. The omental flap is brought through an incision in the pars costalis of the diaphragm. Care should be taken to avoid rotation of or excessive tension on the omental pedicle. The omentum is spread out within the thorax to provide a large surface area. An omentopexy is performed by using synthetic absorbable suture to anchor the omentum to the mediastinum in the region of the lymphaticovenous anastomoses, between the thoracic duct and the cranial vena cava. Sutures should be placed so that they do not interfere with the blood supply of the omentum. The success of this technique is unproven at this point.
Other treatments
Passive pleuroperitoneal shunting has been recommended as treatment of chylothorax in cats, but this technique is no longer recommended by the author. The goal of placing a fenestrated silastic sheet in the diaphragm was to allow drainage of the chylous fluid into the abdomen where the fluid could be reabsorbed by visceral and peritoneal lymphatics, thereby alleviating the respiratory distress and need for subsequent thoracentesis (Peterson et al 1989). The author has not found this technique to be effective, and chronic irritation of the sheeting may be associated with neoplastic transformation of tissues.
Active pleuroperitoneal or pleurovenous shunting (Denver double valve peritoneous shunt, Denver Biomaterials Inc., Evergreen, CO, USA) has been recommended for the treatment of chylothorax in dogs and cats, and may be a reasonable consideration in animals in which all other therapies have failed. Commercially made shunt catheters are available and can be used to pump fluid from the thorax to the abdomen (Smeak et al 1987, Willauer & Breznock 1987, Donner 1989). The catheter is placed under general anaesthesia. A vertical incision is made over the middle of the fifth, sixth and seventh ribs. A purse-string suture is placed in the skin at this site and following the placement of fenestrations in the venous end of the shunt catheter; the catheter is bluntly inserted into the pleural space. A tunnel is created by blunt dissection under the external abdominal oblique muscle, and the pump chamber is pulled through the tunnel. The efferent end of the catheter is then placed into the abdominal cavity through a preplaced purse-string suture and incision located just caudal to the costal arch. The shunt must be placed with the pump chamber directly overlying a rib so that the chamber can be compressed effectively (Fig 5). Advantages of pleurovenous or pleuroperitoneal shunting of chyle are that it may allow more complete drainage of the thorax than passive peritoneal absorption, which may occur with pleuroperitoneal shunting. Disadvantages are that the shunts are expensive, they may easily occlude with fibrin, some animals will not tolerate compression of the pump chamber, and they require a high degree of owner compliance and dedication. Additionally, thrombosis, venous occlusion, sepsis and electrolyte abnormalities have been reported in humans (Smith et al 1984, Vacek et al 1987, Fildes et al 1988, Holm et al 1989, Moskovitz 1990).

Diagram depicting placement of a pleuroperitoneal shunt. The pump chamber should be positioned over a rib so that it can be manually compressed. (Reproduced from Fossum (ed.) Small Animal Surgery, Mosby, Publishing Co. 1997.)
Pleurodesis is the formation of generalised adhesions between the visceral and parietal pleura. Adhesions may occur spontaneously in association with pleural effusion, or in some species they can be induced following instillation of an irritating substance into the pleural cavity (Birchard et al 1989, Jerram et al 1999). This technique has been recommended for the treatment of chylothorax in dogs and cats, but is not recommended by the author. In order for pleurodesis to occur, the lungs must be able to contact the body wall; however, many animals with chronic chylothorax have some thickening of their visceral pleura, which prohibits normal lung expansion (see fibrosing pleuritis above). Neither mechanical (surgical) pleurodesis or talc administration resulted in pleurodesis in experimental dogs; however, thickening of the pleura did occur in some animals (Jerram et al 1999). Chemical or surgical pleurodesis is unlikely to be successful in cats with chylothorax.
