Abstract
Feline herpesvirus-1 (FHV-1) infection is ubiquitous in the domestic cat population worldwide. The most common clinical ocular manifestations of infection with FHV-1 are conjunctivitis and keratitis. This paper reviews the pathogenesis of feline herpesvirus-1 and discusses the various clinical ocular manifestations, diagnostic techniques and treatment of FHV-1-induced diseases. Ocular manifestations include: conjunctivitis, keratitis, stromal keratitis, keratoconjunctivitis sicca, ophthalmia neonatorium, symblepharon, corneal sequestrum, eosinophilic keratitis and anterior uveitis. Diagnostic techniques discussed include: virus isolation, fluorescent antibody testing, serum neutralising titers, ELISA and polymerase chain reaction. Various therapies are also discussed.
Feline herpesvirus-1 is the most frequent cause of conjunctivitis and keratitis in domestic cats. The virus is ubiquitous in domestic cat populations. Feline herpesvirus-1 (FHV-1) is also the most studied infectious cause of ocular surface disease in cats. Diagnosis and treatment of ocular manifestations of feline herpesvirus can be difficult, frustrating and expensive, and recurrences are common in infected animals. Cats infected with FHV-1 can present with conjunctival, corneal or a combination of conjunctival and corneal signs. They may also present with or without systemic illness and upper respiratory signs. FHV-1-associated ocular diseases include chronic conjunctivitis, symblepharon, keratoconjunctivitis sicca, eosinophilic keratitis, stromal keratitis and corneal sequestrum. This paper will review the pathogenesis of FHV-1 and discuss the various clinical ocular manifestations, diagnostic techniques, and treatment of FHV-1-induced diseases.
Ocular anatomy and physiology
The feline cornea is comprised of five layers: outermost stratified squamous epithelium, epithelial basement membrane, stroma, Descemet's membrane and innermost endothelium. Feline conjunctival and corneal epithelium are embryologically derived from surface ectoderm, and the corneal stroma is derived from neural crest cells (Cook 1999). The corneal epithelium in the normal cat is five to six cell layers thick, which measures approximately 25–40 μm, and is comprised of stratified squamous epithelium, wing cells and basal cells (Samuelson 1999). An epithelial basement membrane anchors the corneal epithelium to the underlying stroma. The corneal stroma is comprised of parallel lamellar collagen bundles, and constitutes the majority of the corneal thickness (Samuelson 1999). Corneal sensory nerves, which are branches of the ophthalmic division of the trigeminal nerve, are located in the superficial corneal stromal layer. Descemet's membrane is an acellular layer of collagen fibrils between the stroma and the inner corneal endothelium (Samuelson 1999). The endothelium is a single cell layer that functions to keep the cornea from imbibing water from the aqueous humor bathing the tissues inside the eye. The mean central corneal thickness has been measured as 578 ± 64 μm by ultrasonic pachymetry in the cat (Gilger et al 1993). The presence of a corneal ulceration (absence of epithelium and varying amounts of stroma) also causes anterior uveitis due to a proposed axon reflex (Gum et al 1999). Following epithelial removal, activation of the corneal sensory nerves normally located in the superficial corneal stroma results in stimulation of the ciliary body musculature and secondary anterior uveitis and pain. It is important to remember that whenever a corneal ulcer is present, there is also secondary anterior uveitis and pain. Corneal sensitivity has been measured in cats with unilateral dendritic corneal ulcers, and 41/61 (67%) corneas were found to be less sensitive than the fellow normal eye (Boydell 1997). The decreased corneal sensitivity was not correlated with the presence or absence of secondary anterior uveitis and pain in this study, and there was no speculation about the relationship between hypoaesthaesia and corneal ulceration. Decreased corneal sensitivity or hypoaesthaesia is reported to occur in humans with Herpes simplex keratitis (Martin 1988).
Pathogenesis
FHV-1 is a DNA α-herpesvirus that induces damage to mucosal epithelial cells during replication and is a cause of feline viral rhinotracheitis (Gaskell & Dawson 1998). The disease is prevalent in domestic cat populations, and there are no other known reservoirs (Gaskell & Dawson 1998). Nasal, oral and conjunctival routes of infection have been demonstrated, and the virus is primarily shed in secretions from these body systems for 1–3 weeks following infection (Gaskell & Dawson 1998). Certain animals also shed virus transiently in faeces and urine (Povey 1990), but there is no documented ‘in utero’ transmission (Gaskell & Dawson 1998). One to 2 days following exposure of naive animals to FHV-1, virus replication and epithelial cell necrosis occur in the nasal turbinates, nasopharynx and conjunctival mucosa (Gaskell & Dawson 1998). In the ocular tissues, FHV-1 has been shown to preferentially infect and cause necrosis of the conjunctival epithelium, as well as replicate in the corneal epithelium (Nasisse et al 1989a). Concurrent secondary bacterial infection can exacerbate clinical signs of FHV-1-induced upper respiratory tract disease. Mortality rates as high as 70% of infected kittens have been reported (Povey 1990), but the mortality rate is low when all susceptible animals are considered (Gaskell & Dawson 1998). Maternally derived antibody protection persists between 2 and 10 weeks, but may not protect cats from subclinical infection (Gaskell & Dawson 1998). Many FHV-1 infections are considered subclinical or mild, and often are not presented for veterinary attention.
Following infection, approximately 80% of susceptible animals become FHV-1 carriers, and 45% of those spontaneously reactivate and either asymptomatically shed or develop clinical disease manifestations (Gaskell & Povey 1977). With the advent of molecular diagnostic techniques, the percentage of latently infected carriers reported has actually increased (Reubel et al 1993, Weigler et al 1997). FHV-1, an α-herpesvirus, is able to establish latency in neuronal tissue. FHV-1 has been detected in the trigeminal ganglion of latently infected cats (Gaskell et al 1985, Nasisse et al 1992, Ohmura et al 1993), as well as acutely infected cats by day 4 post-infection (Nasisse et al 1992). Latency is assumed to be lifelong. However, periods of shedding may or may not occur depending on the presence of stress events in the cat's life.
Clinical manifestations of FHV-1
Conjunctivitis
Primary FHV-1 infection and viral replication with secondary bacterial infection results in conjunctivitis. Following a 2–6 day incubation period in neonatal and adolescent cats, serous ocular and nasal discharge, sneezing, inappetence and fever are common clinical signs (Gaskell & Dawson 1998). By day 4 post-infection, there is diffuse necrosis of the conjunctival epithelium and often large numbers of intranuclear inclusions in conjunctival epithelial cells (Nasisse 1990). The conjunctivitis is usually bilateral, and is manifested as hyperaemia or redness with serous discharge that progresses over several days to mucopurulent ocular discharge (Fig 1). Chemosis, which is swelling or oedema of the conjunctiva, may occur but to a lesser extent than is observed with bacterial conjunctivitis. Conjunctival infection always occurs with primary FHV-1 infection, and there may also be limited corneal replication of the virus (Nasisse 1990). Most cats recover in 10–20 days with no ocular sequelae. Severe infection or immunosuppression may result in chronic or recurrent conjunctivitis (Gaskell & Dawson 1998). Some adult cats may exhibit recrudescence as conjunctivitis in one or both eyes. The course of clinical disease varies from weeks to months and can recur (Nasisse 1990).

The left eye of an adolescent cat presented with signs of upper respiratory tract disease and conjunctivitis attributed to feline herpesvirus-1 infection. Conjunctival hyperaemia and mild chemosis are present.
Keratitis
FHV-1 is the only documented viral cause of feline keratitis. FHV-1-induced corneal ulceration is primarily a disease of adult cats, and is likely a reactivation of latent virus. Reactivation of latent virus has been associated with systemic immunosuppression (feline leukaemia virus and/or feline immunodeficiency virus infection), environmental stress (boarding, surgery, moving to a new environment, addition of a new pet) or systemic corticosteroid administration.
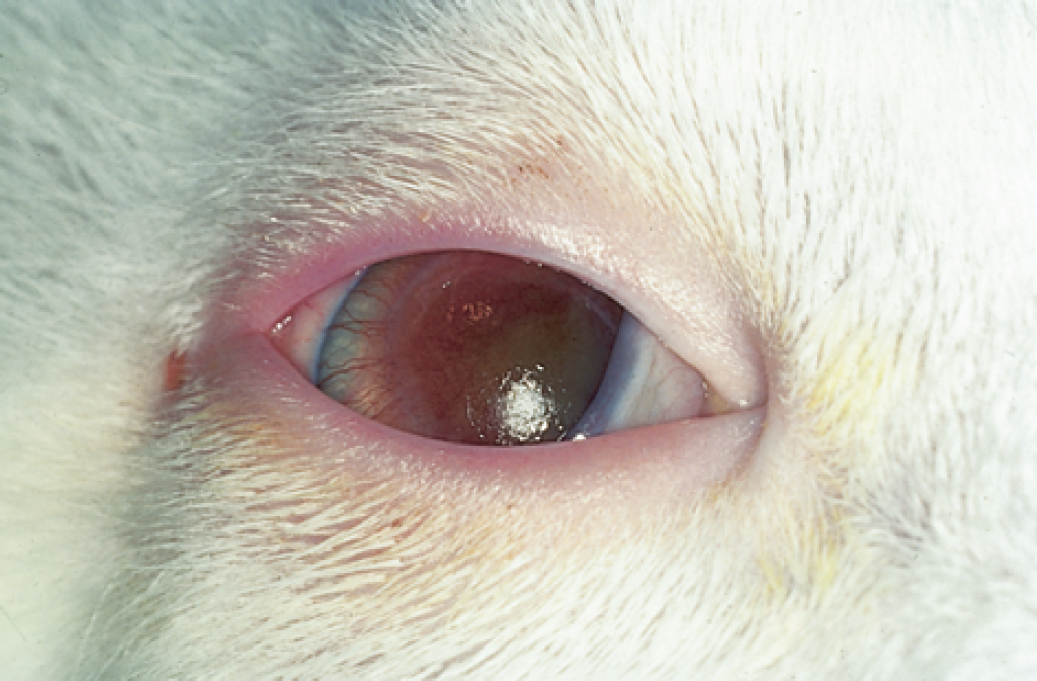
Dendritic corneal ulcers are almost pathognomonic for FHV-1 infection (Fig 2) (Roberts et al 1972, Nasisse & Weigler 1997), and result from direct cytopathic effect of the virus in the basal cell layer of the corneal epithelium (Roberts et al 1972). Theoretically, local immune response suppression allows FHV-1 to reach the corneal stroma, and subsequent keratitis is mediated by an immune response to parts of the viral antigen causing stromal damage that is unrelated to virus replication (Nasisse et al 1995). Dendrites that coalesce to a map-like appearance are called geographic ulcers (Nasisse 1990).

A unilateral dendritic corneal ulcer in a feline herpesvirus-1 positive cat stained with fluorescein.
The ocular clinical signs depend on the depth of corneal infection and the chronicity. Acutely, mild to moderate conjunctivitis, blepharospasm and ocular discharge are seen. The keratitis may be unilateral or bilateral, and respiratory disease is absent. Chronically, stromal oedema and vascularisation may occur. Stromal ulceration can occur, but it seems unlikely that FHV-1 is able to induce corneal stromal melting (Nasisse 1990).
Stromal keratitis
Stromal keratitis refers to infection and inflammation of the deeper corneal tissue. Stromal keratitis is a less common manifestation of FHV-1 infection, but is considered to be a very significant form of disease due to the potential for vision-threatening stromal opacification and scarring (Fig 3) (Nasisse 1990). The mechanism of stromal disease manifestation is not known, but chronic recurrent episodes of FHV-1 keratitis result in stromal collagen damage and opacification (Nasisse 1990). Prolonged absence of corneal epithelium precedes stromal keratitis, and stromal keratitis is not a manifestation of primary FHV-1 infection (Nasisse et al 1989a).

Stromal keratitis caused by feline herpesvirus-1 infection (courtesy of Dr Michelle Willis, Columbus, OH, USA). The inflammation extends into the deeper stroma of the left eye of this cat as evidenced by the opacity and vascularisation of the cornea.
Keratoconjunctivitis sicca
Keratoconjunctivitis sicca (KCS) occurs in cats with either chronic or recurrent blepharoconjunctivitis, sometimes caused by FHV-1 infection (Glaze & Gelatt 1999). It is unknown whether the damage is to secretory glandular tissue that provides the aqueous portion of the tear film or to the ductules that carry the aqueous tears to the cornea (Glaze & Gelatt 1999). Clinical signs can include conjunctival hyperaemia, dry appearance of the cornea, corneal epithelial hyperplasia and corneal ulceration (Fig 4). Diagnosis of feline KCS is based on clinical signs and Schirmer tear test measurement. Normal Schirmer tear test value for the cat is 17 ± 5.7 mm wetting/60 s (Veith et al 1970). KCS is defined as tear production of less than 5 mm wetting/60 s (Glaze & Gelatt 1999). Treatment of feline KCS may include administration of preservative-free artificial tear ointment or solution as often as possible. Application of 0.15% Hylashield (I-med Pharma), a viscoelastic substance containing a hyaluronan derivative manufactured to have an increased contact time, requires less frequent administration. Oral 0.25% pilocarpine can be administered with food, but caution must be taken to avoid systemic effects of parasympathomimetic drug toxicity. Topical cyclosporine A (Optimmune 0.1%, Schering-Plough) is the treatment of choice for canine KCS, but its efficacy and safety have not been reported in the cat (Glaze & Gelatt 1999). There is also concern about topical cyclosporine use in a cat with FHV-1, as it may cause local immunosuppression and recurrence of latent infection.
Keratoconjunctivitis sicca in a 2-year-old cat with chronic feline herpesvirus conjunctivitis. Note the dullness and dry appearance of the cornea.
Ophthalmia neonatorium and symblepharon
Normally, the eyelids of kittens remain closed for the first 10–14 days of life to allow complete development of ocular tissues. Fetal infection with FHV-1 before the eyelids are open can result in mucopurulent conjunctivitis causing distension of the eyelids or, ophthalmia neonatorium. The eyelids are fused with varying amounts of material between the lids and the cornea.
Symblepharon is the adhesion of the conjunctiva to itself or the cornea and is not uncommon in young animals with a history compatible with FHV-1 infection. Upper respiratory tract disease and FHV-1-induced conjunctival epithelial necrosis could theoretically precede symblepharon (Nasisse 1990), although this has not been proven. The epithelial necrosis needed to produce symblepharon is profound, and intuitively it would appear that FHV-1 could produce this necrosis (Glaze & Gelatt 1999). Generally, there is no treatment for this condition as removal of adhered tissue induces more damage and can cause further adhesion. Referral to a specialist is indicated if the owner desires to pursue treatment.
Corneal sequestrum
Corneal sequestrum has also been called corneal mummification or corneal nigrum, and is a disease that is unique to the cat. It is reportedly more common in brachycephalic cat breeds (Persian, Himalayan, Burmese) (Glaze & Gelatt 1999), and can occur following corneal ulceration. The exact cause and pathogenesis of the disease are not known, but stromal collagen degeneration and tan to brown pigment deposition are characteristic (Fig 5) (Glaze & Gelatt 1999). While not a specific cause of corneal sequestrum, FHV-1 has been associated with corneal sequestrum in the cat (Glaze & Gelatt 1999). Chronic corneal FHV-1 infection causing stromal damage may result in sequestrum formation (Nasisse 1990). Polymerase chain reaction (PCR) tests performed on corneal sequestra have been positive for FHV-1 in 18% (5/28) (Stiles et al 1997a) to 55.5% (86/156) (Nasisse et al 1998) of cases depending on the sensitivity of the test. However, in the brachycephalic breeds of cats, FHV-1 detection in corneal sequestrum samples was less prevalent than expected (Nasisse et al 1998). Multiple treatment options exist, including observation, keratectomy, conjunctival pedicle flap and corneoconjunctival transposition.

A corneal sequestrum in a 6-year-old domestic shorthair cat that had suffered from recurrent corneal ulcerations caused by feline herpesvirus-1. The axial, darkly pigmented plaque is in the superficial one-third of the corneal stroma.
Eosinophilic keratitis
Eosinophilic keratitis or proliferative keratoconjunctivitis is an infiltrative, progressive form of corneal disease that occurs in cats. On presentation, a pink to white, irregular, vascularised mass in the peripheral (lateral or nasal) limbal area that may invade cornea and conjunctiva affects one or both eyes (Fig 6) (Glaze & Gelatt 1999). Cytologic examination of a tissue scraping reveals eosinophils, plasma cells and lymphocytes, and is considered diagnostic (Glaze & Gelatt 1999). FHV-1 has been isolated from eosinophilic keratitis specimens isolated by indirect immunofluorescence assay in 33.3% (9/27) of samples (Morgan et al 1996) from one laboratory, and in 76.3% (45/59) of cases by PCR from another laboratory (Nasisse et al 1998). The role of FHV-1 in eosinophilic keratitis has yet to be elucidated. As treatment of eosinophilic keratitis requires topical or systemic corticosteroid administration, active or latent FHV-1 infection can be worsened.

Eosinophilic keratitis in a 3-year-old domestic shorthair cat with chronic feline herpesvirus-1 infection. Both eyes are similarly affected. There is a temporal corneal mass that is pink in colour. The cornea has been stained with topical fluorescein dye.
Anterior uveitis
A recent report found FHV-1 DNA in the aqueous humor of 12/86 cats (14%) with clinical signs of anterior uveitis that tested negative for other known causes of uveitis in cats (toxoplasmosis, feline immunodeficiency virus, feline leukaemia virus-induced lymphosarcoma and feline infectious peritonitis) (Maggs et al 1999). It is not known if the presence of intra-ocular FHV-1 was a cause or the result of uveitis, but the results suggest that FHV-1 can induce anterior uveitis in some cats (Maggs et al 1999). Further research is needed to determine the prevalence of FHV-1 as a causative factor for so-called idiopathic uveitis of cats.
Diagnosis
It is important to perform thorough physical and ophthalmic examinations on all cats suspected of having FHV-1 infection. Acute FHV-1 infection is usually diagnosed based on ocular and respiratory clinical signs, but sometimes laboratory testing is necessary for diagnosis. Conjunctival cytology samples may be examined in acute, primary FHV-1 infections particularly for the presence of intranuclear inclusion bodies; however, the inclusions are not identifiable with Wright-Giemsa stain and therefore may be overlooked (Nasisse & Weigler 1997). In chronic cases, conjunctival cytology may be supportive but is rarely diagnostic because the predominant cell type observed is the neutrophil (Nasisse & Weigler 1997).
Characteristic clinical signs help lead to a diagnosis of FHV-1 keratitis. Staining with rose Bengal will help identify early dendritic lesions prior to the disruption of the corneal epithelium. Rose Bengal is a vital dye that stains dead and degenerating cells, and maybe normal cells at high stain concentrations (Strubbe & Gelatt 1999). Application of topical sodium fluorescein stain will identify corneal epithelial disruption and resultant corneal ulceration. If fluorescent antibody testing is to be considered, the sample must be obtained prior to fluorescein staining for ulceration (da Silva Curiel et al 1991). Laboratory confirmation is often difficult with chronic keratitis and, in these cases, if clinical suspicion and response to therapy are not convincing for FHV-1, PCR sample submission should be considered.
Virus isolation (VI) is considered the ‘gold standard’ for diagnosis of herpesvirus infections (Nasisse & Weigler 1997). Virus isolation detects cytopathic effects of FHV-1 replication in feline cell lines (Nasisse & Weigler 1997). Samples that cannot be run immediately should be stored in the refrigerator (4°C) to prevent loss of virus titre with freezing and thawing (Nasisse & Weigler 1997). The test is sensitive in acute infections, but less sensitive for chronic infections (Nasisse & Weigler 1997), unless during a reactivation episode.
Serology (ELISA) or serum neutralising (SN) titres are diagnostic tests performed by many laboratories. The tests are not considered to be clinically useful because of the high prevalence of vaccination against feline viral rhinotracheitis (FHV-1) with resultant systemic antibody titres (Gaskell & Dawson 1998). In testing chronic infected cats, paired serum sample titres are not likely to be of benefit because the titres tend to plateau with chronic infections (Nasisse & Weigler 1997).
Fluorescent antibody (FA) testing can be performed on corneal or conjunctival smears. Both direct and indirect assays are available, but most laboratories run an indirect testing method because antibody conjugation is not required (Nasisse & Weigler 1997). Fluorescent antibody testing for FHV-1 lacks sensitivity because of the required subjective judgement of laboratory personnel. Also, particularly in chronic infections, virus may already be bound to host antibody (Nasisse & Weigler 1997), and result in a false negative test. A recent report found that FA testing was more often positive with acute respiratory tract disease than in normal cats or cats with chronic ocular disease (Maggs et al 1999). If samples are to be collected for FA testing, it is recommended to do so prior to staining the eye with topical sodium fluorescein, as the dye may create false positive results during FA testing (da Silva Curiel et al 1991).
PCR tests allow amplification and identification of extremely small amounts of material of interest. The test is highly sensitive and specific, and has revolutionised diagnostic testing for many infectious organisms including FHV-1. False positive results can occur with PCR. Nested PCR, which utilises a second primer pair internal to the initial primer, is particularly susceptible to contamination (Stiles et al 1997b). PCR has been shown to be useful in diagnosing cats with FHV-1 conjunctivitis, which has been a difficult subset of cats to diagnose (Nasisse et al 1998). Currently in the USA, FHV-1 PCR testing is available through Antech Diagnostics (Irvine, CA) and the Colorado State University Veterinary Diagnostic Laboratory (Ft. Collins, CO). A positive PCR demonstrates that the animal was infected at some time point but does not prove that an active viral infection is occurring at the time of sample collection (Lutz et al 1999). FHV-1 DNA has been detected in numerous ocular tissues using PCR technology. The exact relationship between the DNA and presence or absence of ocular disease is still open to debate. Similarly, the role of ocular disease in the reactivation scheme is poorly understood.
Recent reports have compared the usefulness of various available diagnostic tests for FHV-1. One report compared VI, FA, SN and ELISA in normal cats, cats with upper respiratory tract signs, and cats with chronic ocular signs (Maggs et al 1999). A high seroprevalence was found with ELISA in all populations. FA and VI commonly detected FHV-1 in normal cats, as well as in cats with clinical signs of FHV-1 infection, and the authors concluded that if both tests were run in conjunction and found to be negative, FHV-1 could be excluded as a cause of disease (Maggs et al 1999). Nested PCR testing has identified FHV-1 infection in 54% of conjunctivitis cases, 12% of normal conjunctival samples, 18% of corneal sequestra and 46% of normal corneas (Stiles et al 1997a). Nasisse et al (1998) reported positive single PCR test results of FHV-1 detection in 76.3% of eosinophilic keratitis samples, 55.1% of corneal sequestra and 5.9% of normal corneal tissue. Nested PCR tests were reported to be more sensitive than VI or FA testing in cats with conjunctivitis or with upper respiratory tract disease and conjunctivitis (Stiles et al 1997b).
Therapy
Primary FHV-1 infections are typically confined to the conjunctiva and are generally self-limiting. Supportive care of the upper respiratory signs, including management of anorexia and dehydration, is important. Non-specific treatment of FHV-1-induced conjunctivitis is directed towards prevention of secondary bacterial conjunctival infection. The most common bacterial isolates are Chlamydia and Mycoplasma. Topical oxytetracycline or chloramphenicol is used four times daily for its effectiveness against Chlamydia and Mycoplasma. Antiviral medications are usually reserved for FHV-1 keratitis.
Response to treatment is variable at best. Epithelial keratitis has a much better prognosis than chronic stromal keratitis, in which the response to antiviral drugs is often poor (Glaze & Gelatt 1999). The mechanism of action of topical antiviral medications is static not cidal; therefore, frequent administration is recommended. This is often difficult for cat owners to accomplish as many of the medications are topically irritating to cats. The in vitro efficacy of topical anti-virals against FHV-1 is trifluridine > idoxuridine > vidarabine > bromovinyldeoxyuridine > acyclovir (Nasisse et al 1989b). Trifluridine or trifluorothymidine (Viroptic, GlaxoWellcome) is typically administered four to nine times daily for 2 days, and then the frequency is gradually reduced over the next 14–21 days. It may cause mild transient irritation of the conjunctiva and cornea. Cats that are irritated by trifluridine administration may benefit from use of a different topical antiviral medication. Resistance to trifluridine may also occur and substitution with idoxuridine or vidarabine (adenine arabinoside) is recommended in those cases.
Systemic administration of antiviral drugs has been investigated in the cat. Acyclovir is an effective drug for human herpesvirus, but cannot reach effective plasma concentration in cats, and is therefore not recommended (Nasisse 1989b, Weiss 1989). Valacyclovir is another purine nucleoside that is considered more bioavailable and is converted to acyclovir after oral dosing but was found to be extremely toxic to cats and is not recommended (Nasisse et al 1997b).
Oral supplementation with the amino acid L-lysine has been shown to decrease the viral shedding rate in FHV-1 positive cats (Maggs & Nasisse 1997). L-lysine may be a competitive inhibitor of arginine as well as an arginase inducer, and FHV-1 growth in vitro has been shown to be markedly inhibited with L-lysine supplementation (Collins et al 1995). Current recommendation is to administer 250 mg of L-lysine orally once daily. Low dose oral interferon α-2a (Roferon-A, Roche Pharmaceuticals) was shown to have a dose-dependent beneficial effect on severity of clinical signs with acute FHV-1 infection in experimental cats, especially if administered before infection occurred (Nasisse et al 1996). However, pre-exposure administration is unlikely to happen in clinical practice.
Due to the local immune suppression and retardation of corneal epithelialisation that occur with topical corticosteroid use, corticosteroid administration should not be considered with most forms of FHV-1 infections (Nasisse 1990). Corticosteroid use may also encourage reactivation of latent infection and hasten ocular disease sequelae. Rarely, topical steroids are used with chronic stromal keratitis in an attempt to decrease the FHV-1 antigenic immune response and conceivably decrease corneal scar size (Nasisse 1990). This must be done very selectively and with great caution.
Conclusions
Infection with FHV-1 is common in domestic cats despite vaccination. There are many acute and chronic ocular manifestations of FHV-1 infection. Conjunctivitis and keratitis are the two ocular disease forms most commonly encountered in feline practice. Conjunctivitis is usually self-limiting, although chronic infections can occur and are challenging to diagnose and treat. Keratitis can be vision threatening, and aggressive management to limit sequelae is indicated. Virus isolation, fluorescent antibody testing, serum neutralising titers, ELISA and PCR each has a place in the diagnosis of FHV-1 infection.
