Abstract
Microsporum canis infection was induced in 21 healthy SPF-derived cats. Once infection was established (4 weeks after inoculation) the cats were divided into three equal groups housed in separate rooms and monitored for 16 weeks. During this time, group A cats received oral griseofulvin at approximately 50 mg/kg daily and were shampooed twice weekly with a product containing chlorhexidine and miconazole. Group B cats were treated with griseofulvin alone, and group C cats served as untreated controls. The cats were examined on a weekly basis and the severity of lesions was scored semi-quantitatively. In addition, hair samples were collected from each cat on a weekly basis by the MacKenzie brush technique and by the sticky-tape method. A semi-quantitative scoring system was also used for the assessment of fungal (M canis) growth. Generally, significant differences in clinical scores were not seen between the groups although at weeks 3, 4 and 11 there was a significant difference (P≤0.015) with cats in group A having significantly lower median scores than those in group C. Median times to clinical resolution (return of clinical scores to zero) in groups A, B and C were at treatment weeks 2, 9 and 12, respectively (P>0.05). Median times for mycological resolution (persistently negative culture results) for groups A, B and C were at treatment weeks 2, 9 and 12, respectively, for the MacKenzie brush technique and at weeks 4, 8 and 12 for the sticky-tape technique. For both these results, the groups differed significantly (P≤0.001) and in both instances group A had significantly more rapid resolution than groups B or C. Median culture scores were significantly different between the three groups using one or both of the sampling techniques at week 2 through to week 12 of treatment with median scores for either group A alone, or groups A and B being significantly lower than group C (P≤0.026). These results showed a benefit from the addition of twice-weekly chlorhexidine-miconazole shampooing to systemic griseofulvin therapy alone in the treatment of M canis infected cats.
Systemic therapy is generally regarded as the treatment of choice for feline dermatophytosis (Moriello & DeBoer 1995) although topical therapy with creams, ointments, shampoos or dips has been widely recommended as an adjunct therapy for generalised dermatophytosis, and even as the sole therapeutic agent for localised lesions (Foil 1986, Medlau & Moriello 1992, Medlau & White-Weithers 1992, Carney & Moriello 1993, Foil 1993). Nevertheless, despite the common recommendation to use topical therapy, there is little information available on the efficacy of this treatment modality in cats. The in vitro efficacy of certain antifungal agents against M. canis-infected dog and cat hairs was assessed by White-Weithers and Medleau (1995), but although these authors demonstrated an effect of exposure to 2% chlorhexidine, in a subsequent in vivo study, the use of topical chlorhexidine was not found to alter the course of experimentally-induced feline dermatophytosis (DeBoer & Moriello 1995).
In a more recent study (Paterson 1999), a shampoo containing 2% chlorhexidine and 2% miconazole was assessed as an adjunctive to systemic therapy with griseofulvin in a group of Persian cats with naturally-occurring M canis infection. The use of this shampoo appeared beneficial in reducing environmental contamination with viable dermatophyte particles, and furthermore the cats that were shampooed showed significantly more rapid resolution of clinical signs, with a trend towards more rapid mycological cures in comparison to a group treated with griseofulvin alone.
The current study was designed to evaluate the efficacy of a topical shampoo containing a combination of chlorhexidine and miconazole (Malaseb shampoo; Leo Animal Health) as an adjunctive therapy in experimental feline M canis infection under controlled conditions. To assess this therapy critically, one group of cats was treated with a combination of the shampoo and systemic griseofulvin, and results from this group were compared to results from two other groups—one receiving griseofulvin therapy alone, and one receiving no therapy at all.
Materials and methods
Cats used and infection protocol
For the study, 21 healthy specific-pathogen-derived, barrier-maintained domestic shorthair cats were used. They comprised 20 females (all entire) and one neutered male. The cats were all between 18 and 24 months of age at the start of the study.
Infection was induced in the cats using a previously described protocol (Sparkes et al 1995). Briefly, a fresh clinical M canis isolate of feline origin which caused fluorescence of infected hairs under Wood's lamp illumination was identified from samples submitted to the Feline Diagnostic Service (School of Veterinary Science, University of Bristol). Hairs bearing spores were inoculated on to six 10 cm diameter plates of Sabouraud's dextrose agar, which were incubated at 30°C for 10 days. At the end of this period, the plates were flooded with 20 ml sterile distilled water, and a mycelial suspension was prepared by gently scraping the surface of the cultures with a sterile bacteriology loop. The mycelial suspension was stored at 4°C with the first inoculation of the cats occurring within 24 h of preparation of the suspension.
The cats were lightly sedated by intramuscular administration of 2 mg/kg−1 medetomidine (Domitor; Pfizer), and a 5 cm×5 cm area of skin on the left caudo-lateral thorax was clipped of hair. The surface of the skin was gently scarified with a 23-gauge hypodermic needle and approximately 2 ml of the mycelial suspension was applied to the scarified skin and was rubbed into the entire clipped area. Further 2 ml aliquots of the mycelial suspension were applied to the same area daily for the next 3 days.
Monitoring the clinical course
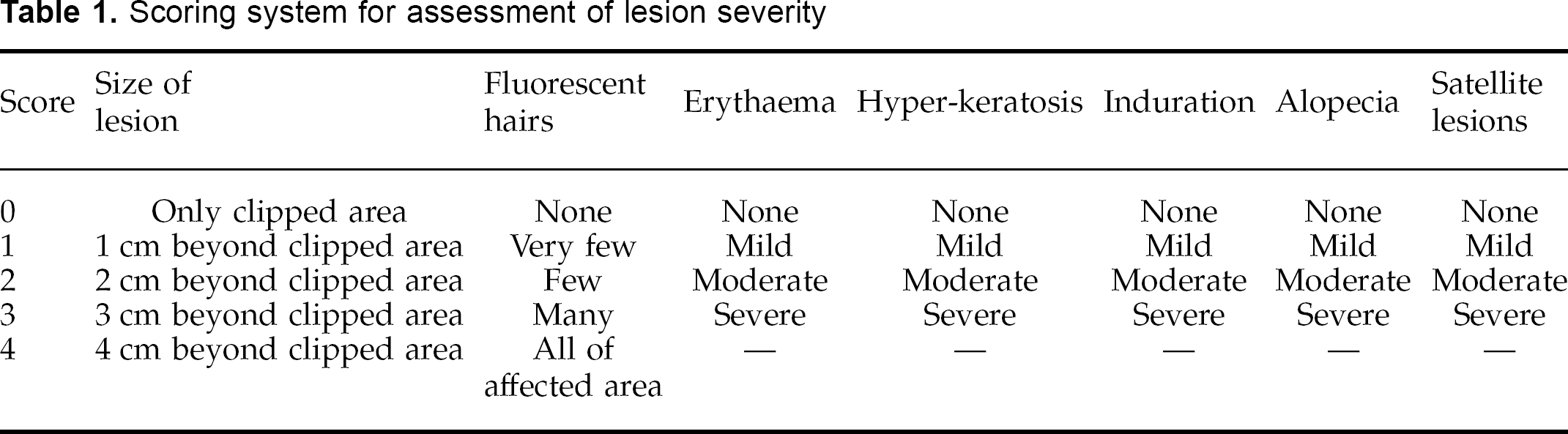
The course of the infection was monitored by regular clinical examination of the cats, and examination of the haircoats with a Wood's lamp. All cats were examined 3 weeks after inoculation (week 0) to determine their infection status, and then randomly assigned in to one of three different treatment groups. Thereafter, each cat was examined on a weekly basis. At each examination, the severity of the lesions was assessed subjectively using a scoring system detailed in Table 1.
Scoring system for assessment of lesion severity
Treatment of the cats
Treatment was commenced one week after the cats had been assigned to their treatment groups (week 1). Each of the three treatment groups contained seven cats. Group A received oral griseofulvin once daily at approximately 50 mg/kg given as a single dose before feeding. In addition, the cats in this group were shampooed twice weekly (every 3–4 days) with a commercial shampoo containing 2% w/v chlorhexidine and 2% w/v miconazole (Malaseb shampoo; Leo Animal Health). This was carried out using a defined protocol wherein the entire haircoat of the cat (excluding the head) was thoroughly wetted with warm water. Ten millilitres of shampoo was then applied to the wetted hair and massaged to develop a good lather and to distribute the shampoo throughout the hair. The shampoo was left on the haircoat for 10 min before being rinsed with clean, warm water and the cats were then dried with clean cotton towels. Cats in group B received only oral griseofulvin once daily at approximately 50 mg/kg given as a single dose prior to feeding. Cats in group C served as untreated controls, and were given no medication throughout the study period.
For the duration of the study (16 weeks of treatment), the three groups of cats were kept in separate rooms. Once weekly the concrete floors in the rooms were disinfected with a halogenated tertiary amine solution (TriGene; MediChem International, Sevenoaks, UK) at the recommended dilution (1:100) and then rinsed with water. The cats were fed a commercial tinned cat food once daily to meet their calorific requirements, and water was available ad libitum.
Mycological monitoring of the cats
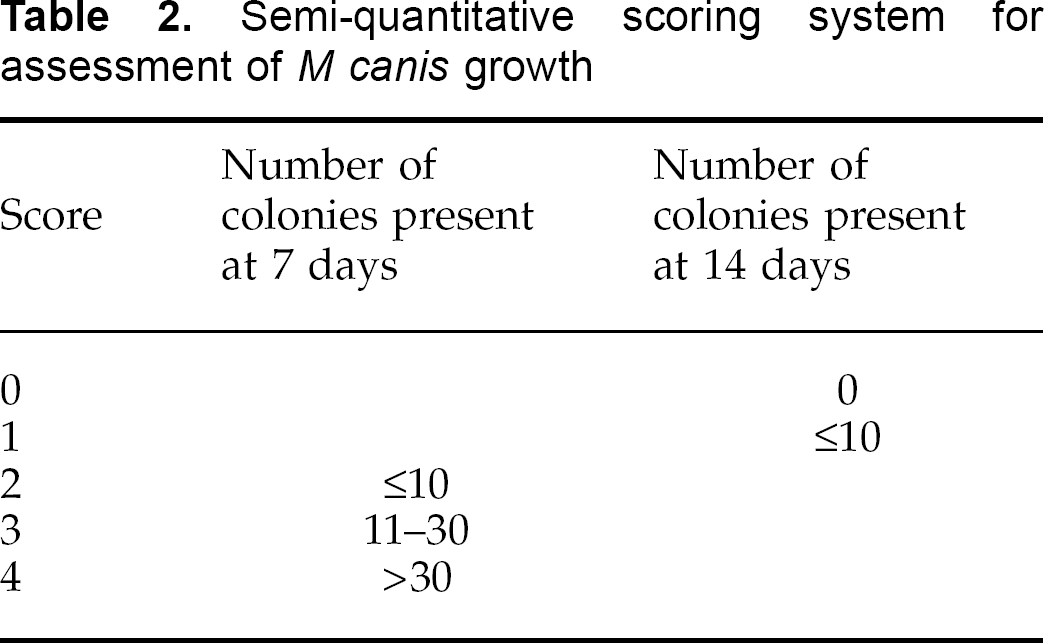
Following routine clinical scoring as described, samples of hair were collected from each cat on a weekly basis for mycological assessment in two ways. Firstly, samples were collected employing a modification of the MacKenzie brush technique (MacKenzie 1961, Ive 1966) using an 8 cm diameter plastic scalp massage brush with 125 bristles (Denman D6 Brush; Denroy International, Bangor, N Ireland). Each cat was brushed vigorously to ensure hairs were trapped in the bristles, and the entire coat was brushed at least twice. Fungal culture was performed by pressing the bristles of the brushes onto plates of Sabouraud's dextrose agar incorporating cylcloheximide (0.04%) and chloramphenicol (0.005%), ensuring transfer of collected hairs to the surface of the culture medium. Secondly, a 4 cm length of sticky tape (Sellotape; 3M products) was gently pressed over the entire area originally used to inoculate the cats with the mycelial suspension. Each strip of sticky tape was pressed several times over the entire surface of a plate of Sabouraud's agar. Inoculated plates were incubated at 30°C and examined for fungal growth after 7 and 14 days. Growth of M canis was assessed on a semi-quantitative basis using the scoring system outlined in Table 2. For the cats in group A, samples for fungal culture were collected using the two described methods prior to shampooing and, in addition, a second set of samples was collected each week from these cats 24 h after they had been shampooed.
Semi-quantitative scoring system for assessment of M canis growth
Statistical analysis
Differences in median results between treatment groups were compared using the Kruskal–Wallis test, and a P value of <0.05 was considered significant. Post-test analysis was carried out using Dunn's test where appropriate to identify which groups were varying significantly.
Differences in proportions between treatment groups were assessed using the chi-squared or Fisher's exact test, and a P value of <0.05 was considered significant.
Results
Clinical disease
Following inoculation with M canis mycelium, 3 weeks later (week 0) all 21 cats exhibited signs of infection with the appearance of fluorescent hairs under Wood's lamp illumination. Treatment was started 1 week later (week 1), and the course of clinical disease in the three groups is shown in Table 3. At week 0, when the cats were randomised to the three groups, there was no significant difference in median clinical scores between them. At week 1, however, when treatment was commenced, there was a significant difference (P=0.015), with group C having a significantly higher median score than group B. This difference was maintained at week 2, but by week 3, group A had a significantly lower median score than group C (P=0.002), and this difference was also evident at weeks 4 and 11 (Table 3).
Clinical scores in the three groups of cats during the course of the study
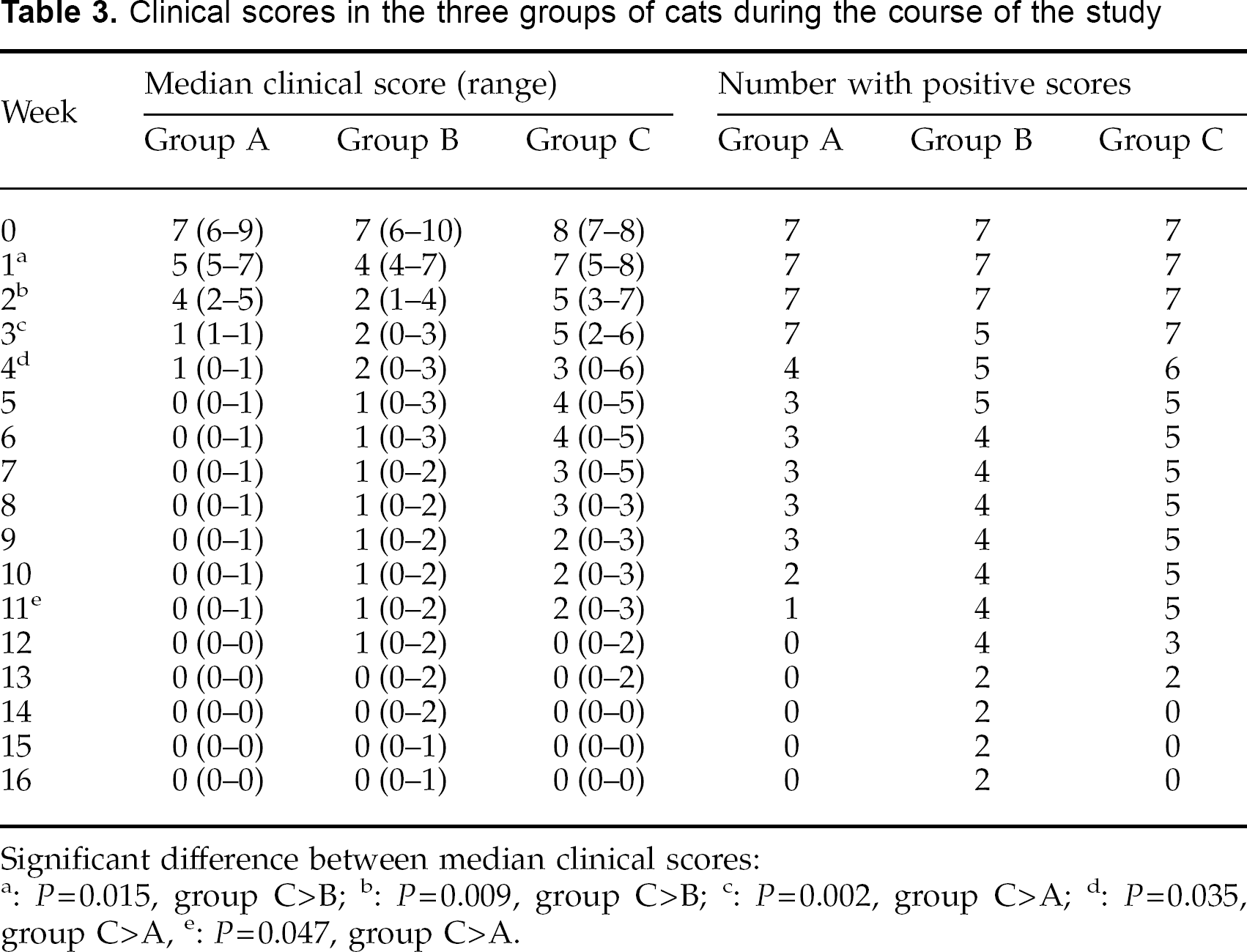
Significant difference between median clinical scores:
P=0.015, group C>B;
P=0.009, group C>B;
P=0.002, group C>A;
P=0.035, group C>A;
P=0.047, group C>A.
Median times of clinical resolution (return of clinical scores to 0) in groups A, B and C occurred at weeks 4 (range 3–11), 12 (range 2–15) and 11 (range 3–13) respectively. However, these differences were not significant.
None of the cats developed satellite lesions at any time point, and only cats in group C developed lesions beyond the original clipped area (evident during weeks 2 to 7 inclusive). In both groups A and B, clinical scores for induration, scaling and erythaema were all zero from week 4, whereas in group C these scores had not all returned to zero until week 8. The total scores for fluorescence reached a peak in groups A and B at week 1, and in group C at week 3. In groups A and B, from week 2 onwards the fluorescence observed in individual hairs no longer extended to the base of the hair, ie, the new hair growth emerging from the hair follicle in these affected hairs was not fluorescing. Such a feature was not observed in cats from group C.
During the final 3 weeks of the study (weeks 14–16) there were only two cats (both from group B) that still had fluorescent hairs, and in these two cats it was fluorescence of just a few hairs at their tips. On completion of the study, these fluorescent hairs were plucked and cultured on Sabouraud's agar, and the hairs from one of the cats grew typical M canis colonies.
Mycological monitoring of the cats
Results of weekly Denman brush and stickytape cultures are summarised in Tables 4 and 5. Median culture scores from Denman brushes were significantly different between the three groups of cats from weeks 2 to 12 inclusive (P≤0.036). At weeks 2, 3, 4 and 10 median scores for group A were significantly lower than for group C; and at weeks 6, 7, 8, 9, 11 and 12 median scores for groups A and B were both significantly lower than those for group C. Median culture scores from sticky tapes were significantly different between the three groups of cats from weeks 3 to 11 inclusive (P≤0.026). At weeks 3, 4, 5, 6, 10 and 11 median scores for group A were significantly lower than for group C, and at weeks 7, 8 and 9 median scores for groups A and B were both significantly lower than those for group C.
Denman brush culture results in the three groups of cats during the course of the study
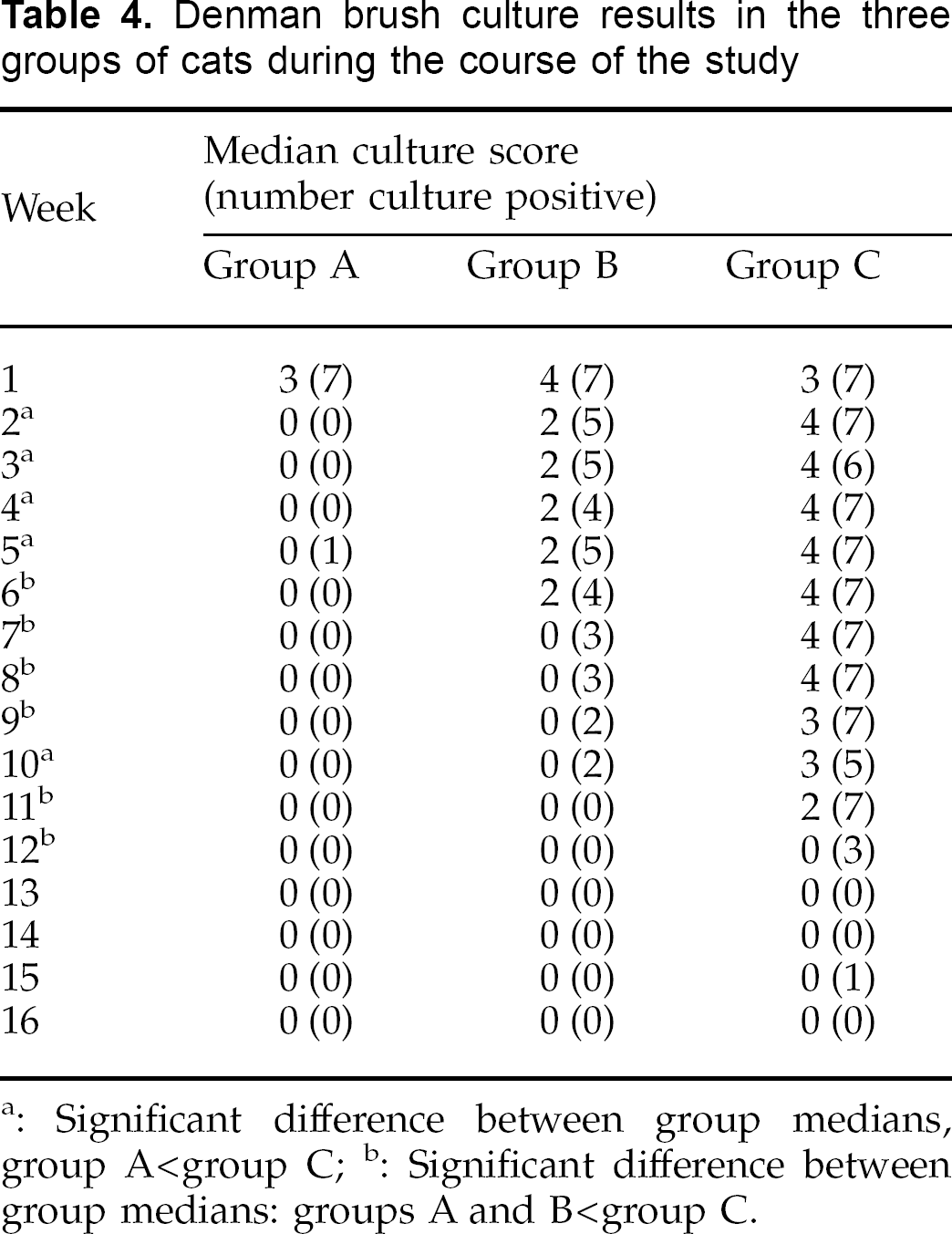
Significant difference between group medians, group A<group C;
Significant difference between group medians: groups A and B<group C.
Sticky-tape culture results in the three groups of cats during the course of the study
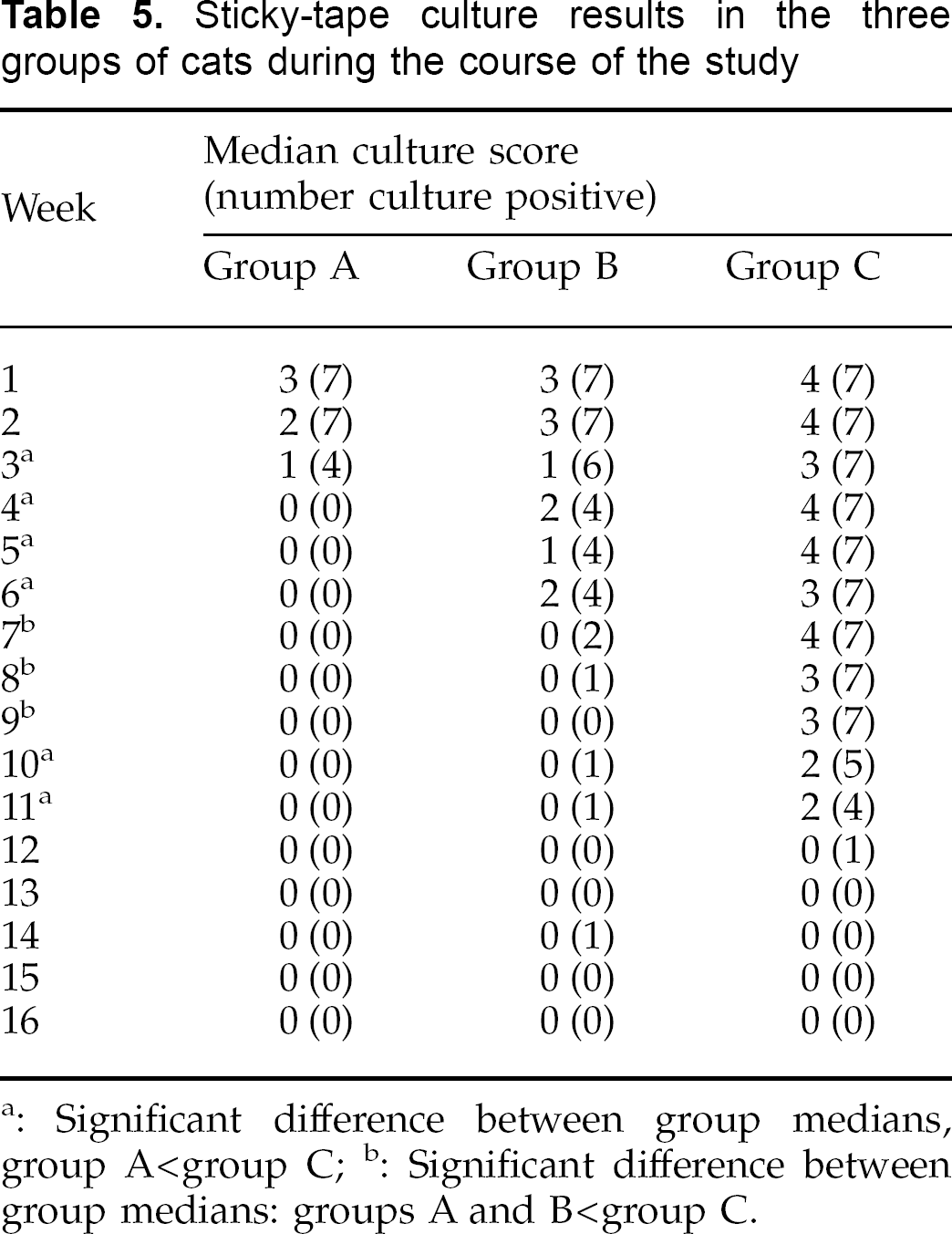
Significant difference between group medians, group A<group C;
Significant difference between group medians: groups A and B<group C.
Taking mycological resolution as the point from which culture results for any individual remained persistently negative, using the Denman brush technique the median (and range) of mycological resolution for groups A, B and C were 2 weeks (2–6), 9 weeks (2–11), and 12 weeks (12–16) respectively. These results were significantly different (P=0.0002) with Group A having significantly more rapid resolution than either groups B or C. Using the sticky-tape method, the median (and range) of mycological resolution for groups A, B and C were 4 weeks (3–4), 8 weeks (4–15), and 12 weeks (10–13) respectively. These results were again significantly different (P=0.001) with group A having significantly more rapid resolution than either groups B or C.
Comparison of brush and sticky-tape cultures from group A, before and after shampooing
The results of brush and sticky-tape cultures from the cats in group A before and 24 h after shampooing are summarised in Table 6. Comparing the pre- and post-shampooing results, median Denman brush culture scores were significantly lower on weeks 2 (P=0.003) and 3 (P=0.004) pre-shampooing; and median stickytape culture scores were significantly lower (P=0.016) post-shampooing on week 1. On other occasions, there were no significant differences between pre- and post-shampooing culture scores.
Denman brush and sticky-tape culture results in group A cats, before and after shampooing
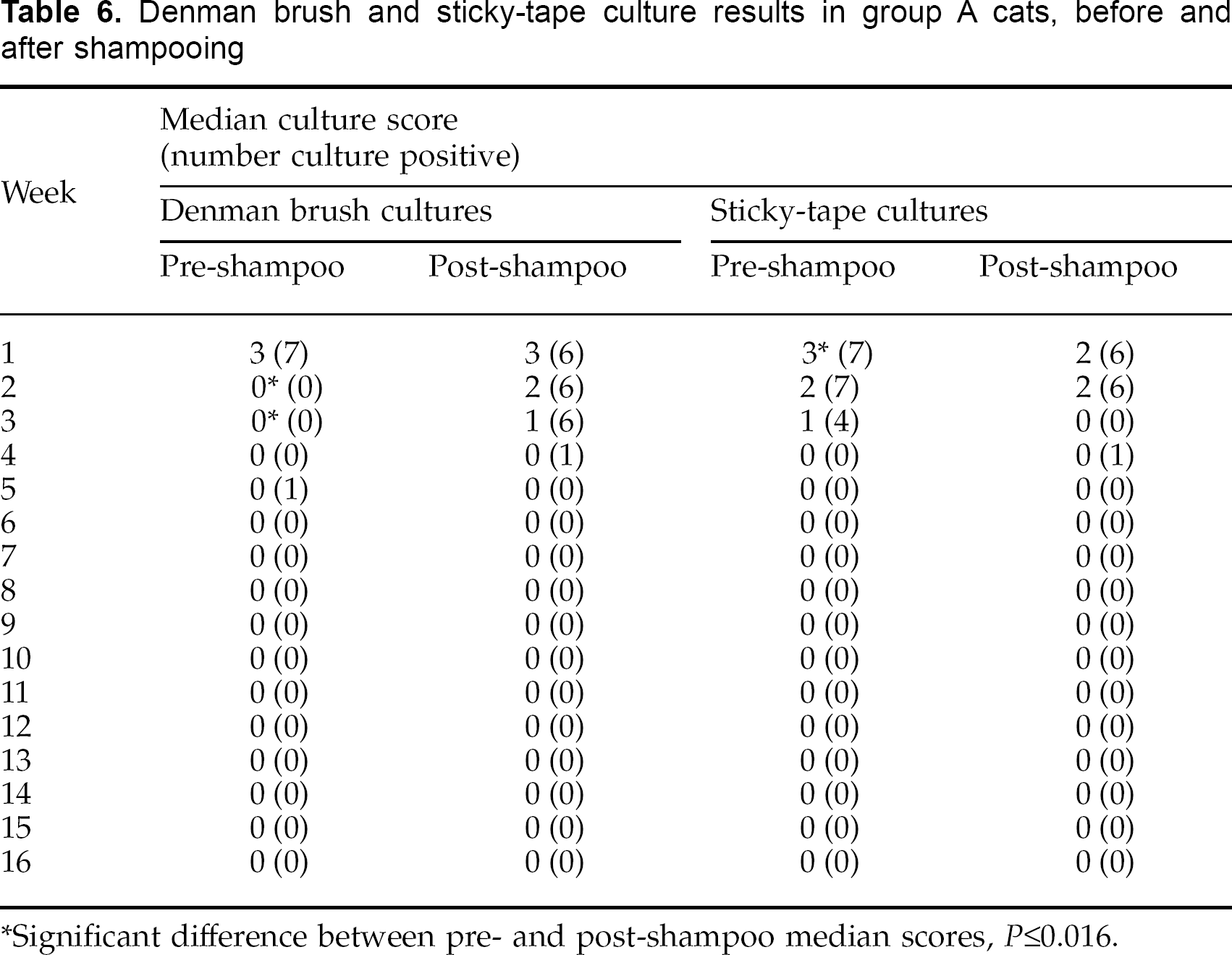
Significant difference between pre- and post-shampoo median scores, P≤0.016.
Discussion
In humans with tinea capitis, the use of a fungicidal shampoo (selenium sulphide) has been shown to be of benefit as an adjunct to systemic treatment (Allen et al 1982), with enhanced mycological cures being evident both in comparison to patients treated with griseofulvin alone, and those treated with griseofulvin and a bland shampoo. However, topical therapy for feline dermatophytosis has been a controversial area. Although certain antifungal agents have been shown to have an in vitro effect on M canis-infected dog and cat hairs (White-Weithers and Medlau 1995), it has been suggested (DeBoer & Moriello 1995, Moriello & DeBoer 1995) that the major value of topical therapy is limited to reducing environmental contamination by killing spores on the haircoat of infected cats. There is little doubt that topical therapy alone does not adequately penetrate the hair follicle and that optimal treatment of dermatophytosis caused by species that invade the hair follicle and hair shaft (such as M canis) requires systemic therapy for effective penetration to this site (Borgers et al 1993). However, the suggestion that topical therapy in feline M canis infection is only of real benefit in reducing environmental contamination appears to come mainly from the observation that certain topical therapies alone were unable to alter the course of experimentally-induced infection in cats (DeBoer & Moriello 1995). In that study, neither weekly shampooing with 0.5% chlorhexidine shampoo followed by dipping in 0.06% chlorhexidine solution, nor weekly shampooing with 2% glyceryl monolaurate in a detergent base appeared to affect lesion scores or mycological cultures in comparison with untreated control cats. In contrast to these results though, Paterson (1999) reported that the use of a 2% chlorhexidine/2% miconazole shampoo in a group of naturally M canis-infected Persian cats produced a significant clinical benefit. In her study, Paterson used the shampoo twice weekly for 2 weeks followed by once weekly, in addition to oral griseofulvin in one group and compared this to griseofulvin therapy alone in another. The study showed significantly decreased lesion scores and a trend (though not significant) to more rapid mycological cure in the shampoo/griseofulvin group in comparison to the use of griseofulvin alone.

Clinical scores during course of the study.
Paterson's results (1999) therefore suggested a beneficial effect on clinical recovery in cats treated with a topical shampoo, although in contrast to the study by DeBoer and Moriello (1995), Paterson used a shampoo that contained a specific and potent antifungal agent—miconazole (VanCutsem & Thienpont 1972)—in addition to chlorhexidine, and the shampoo was used as an adjunct to systemic therapy rather than as a sole therapy.
In the controlled study reported here, we used the same shampoo preparation used by Paterson (1999), although under somewhat different conditions. As with the Paterson study, we allowed a 10 min contact time with the shampoo before rinsing it from the coat, but we continued twice-weekly shampooing for the duration of our study rather than reducing to once weekly after the first 2 weeks of therapy. As with the Paterson study, the shampoo was used as an adjunct to systemic therapy with griseofulvin at 50 mg/kg daily, but we also included an untreated control group for additional comparison. The results of this study demonstrated a clear and positive effect for adjunctive shampoo therapy on the course of experimental M canis infection in cats.
All 21 cats used for this study developed clinically and mycologically confirmed infection with M canis. Treatment was not commenced until 4 weeks after infection (week 1 of the study) to allow the lesions to become established and thus reflect more closely the situation that would arise with treatment of naturally occurring feline M canis infections. Monitoring the clinical course in group C (untreated cats) revealed a pattern of disease similar to that described previously in experimental M canis infection (Sparkes et al 1995), with clinical resolution occurring between weeks 3 and 13 (ie 7–17 weeks post-infection).
In comparison to the cats in group C, cats in group A had significantly lower median clinical scores at weeks 3, 4 and 11, and in addition had an earlier median time of clinical resolution (4 weeks as compared to 12 and 11 weeks for groups B and C). However, the latter difference did not reach statistical significance.
Although a relatively small difference was evident in clinical scores between the three groups of cats, mycological monitoring revealed much more substantial differences. Results of Denman brush and sticky-tape cultures (Tables 4, 5) were comparable to each other, although overall, the sticky-tape technique appeared to be a little more sensitive. By week 2 of treatment, median Denman brush culture scores for group A were significantly lower than group C, and the same was true of sticky-tape cultures by week 3. With the exception of one cat with a positive Denman brush culture at week 5, all cats in group A were persistently negative by Denman brush culture from week 2, and by sticky-tape culture from week 4. In comparison, positive cultures were still obtained by either the Denman brush or sticky-tape technique from group B cats at weeks 10 and 14, and from group C cats at weeks 15 and 12, respectively. The more rapid mycological cure in group A cats was evidenced by the significantly shorter median mycological resolution time in this group, compared to both groups B and C.
Comparing culture results in group A before and after shampooing showed positive post-shampooing Denman brush cultures at weeks 2, 3 and 4 whereas the pre-shampooing cultures at these times had been negative (Table 6). Two obvious factors might explain this finding: firstly, the act of shampooing may fracture infected hair-shafts and liberate M canis arthrospores, which then get dispersed through the haircoat and result in positive cultures; secondly, the shampooing and resultant wetting of the hair-coat may have resulted in enhanced contamination of the haircoat with arthrospores from the environment in which the cats were kept. Culture scores from individual cats at these times were invariably low (1 or 2) reflecting far fewer viable M canis particles than at week 1.
Griseofulvin is known to be a fungistatic rather than a fungicidal agent (Hay 1990), and this is illustrated by the viability of arthrospores in the fluorescent tips of hairs remaining after 16 weeks of therapy in one of the cats in group B. This also emphasises the potential value of clipping the haircoat of infected cats to remove infective particles. Based on the appearance of visibly healthy (non-fluorescent) hair growing out of the hair follicle after 2 weeks of therapy, maximum benefit from this procedure may be anticipated if it were performed 2–4 weeks after commencement of therapy. Delaying clipping until healthy (non-fluorescent) hair growth is clearly visible, such that clipping would remove all the fluorescent portion of the hair, may help to reduce the temporary exacerbation of clinical signs that has sometimes been observed with this procedure (Moriello & DeBoer 1995).
In conclusion, this study has demonstrated a beneficial effect from the addition of twice weekly topical chlorhexidine-miconazole shampoo to systemic griseofulvin in the treatment of feline M canis infection. Although a small benefit could be observed from the clinical scoring of the cats in group A, the major difference was seen in results of mycological monitoring of the cats. This beneficial effect may be due to a combination of the anti-mycotic effects of the shampoo and the physical act of shampooing helping to remove infected hairs. In view of the lack of effect of topical shampoo therapy reported in a previous study when used as a sole agent for the treatment of feline dermatophytosis (DeBoer & Moriello 1995), however, it may be that chlorhexidine-miconazole shampoo possesses good anti-myctoic effects in vivo. Regardless of the precise mechanisms involved, the results of this study lend strong support to the clinical use of chlorhexidine-miconazole shampoo as an aid to treating clinical M canis infection in cats. In addition, by producing a more rapid mycological resolution, the use of this shampoo can be recommended as an aid to controlling the spread of infection to other animals and man.
Footnotes
Acknowledgements
The authors gratefully acknowledge the support of Leo Animal Health in conducting this study. S Caney, C Bludell and M Cobb are thanked for their help with the study. A H Sparkes was supported by the Feline Advisory Bureau.
