Abstract
A 2-year-old entire female British Shorthair cat was referred to the University of Bristol for investigation of lethargy, weakness, constipation and hypothermia. Clinical examination revealed a profoundly weak, hypovolaemic and hypothermic cat. Serum biochemistry revealed hyponatraemia, hyperkalaemia and hyperphosphataemia and the urine was isosthenuric. Lack of response to exogenous adrenocorticotrophic hormone confirmed a diagnosis of hypoadrenocorticism. Treatment consisted initially of intravenous fluid therapy and subsequently a combination of fludrocortisone and prednisolone per os. At follow-up, 20 months after the initial diagnosis the cat remained stable and free of clinical signs.
Primary hypoadrenocorticism, or Addison's disease, results from deficient adrenal production of mineralocorticoids and glucocorticoids due to adrenal cortex destruction (Feldman & Nelson 1996a). This disease is rare in cats (Feldman & Nelson 1996b) with only 11 naturally occurring cases having been reported (Peterson et al 1989, Berger & Reed 1993) and none previously reported from outside the United States.
A 2-year-old entire female British Shorthair cat was referred to the Feline Centre at the University of Bristol for investigation of suspected neurological disease. Five weeks prior to referral she suffered an episode of extreme lethargy, weakness, apparent ataxia, constipation, and hypothermia. The primary care veterinarian administered an enema and initiated treatment with intravenous fluid therapy and enrofloxacin (Baytril; Bayer). After an initial clinical improvement the cat's condition relapsed, resulting in referral. The cat's appetite was described as being poor prior to referral.
On clinical examination, the cat was found to be extremely weak and unable to move more than a few paces, during which she appeared unsteady. She was estimated to be approximately 5% dehydrated and was hypothermic (rectal temperature 36.7°C). Heart rate and rhythm were both normal and there was no pulse deficit. A neurological examination was attempted but could not be carried out due to the extreme weakness present.
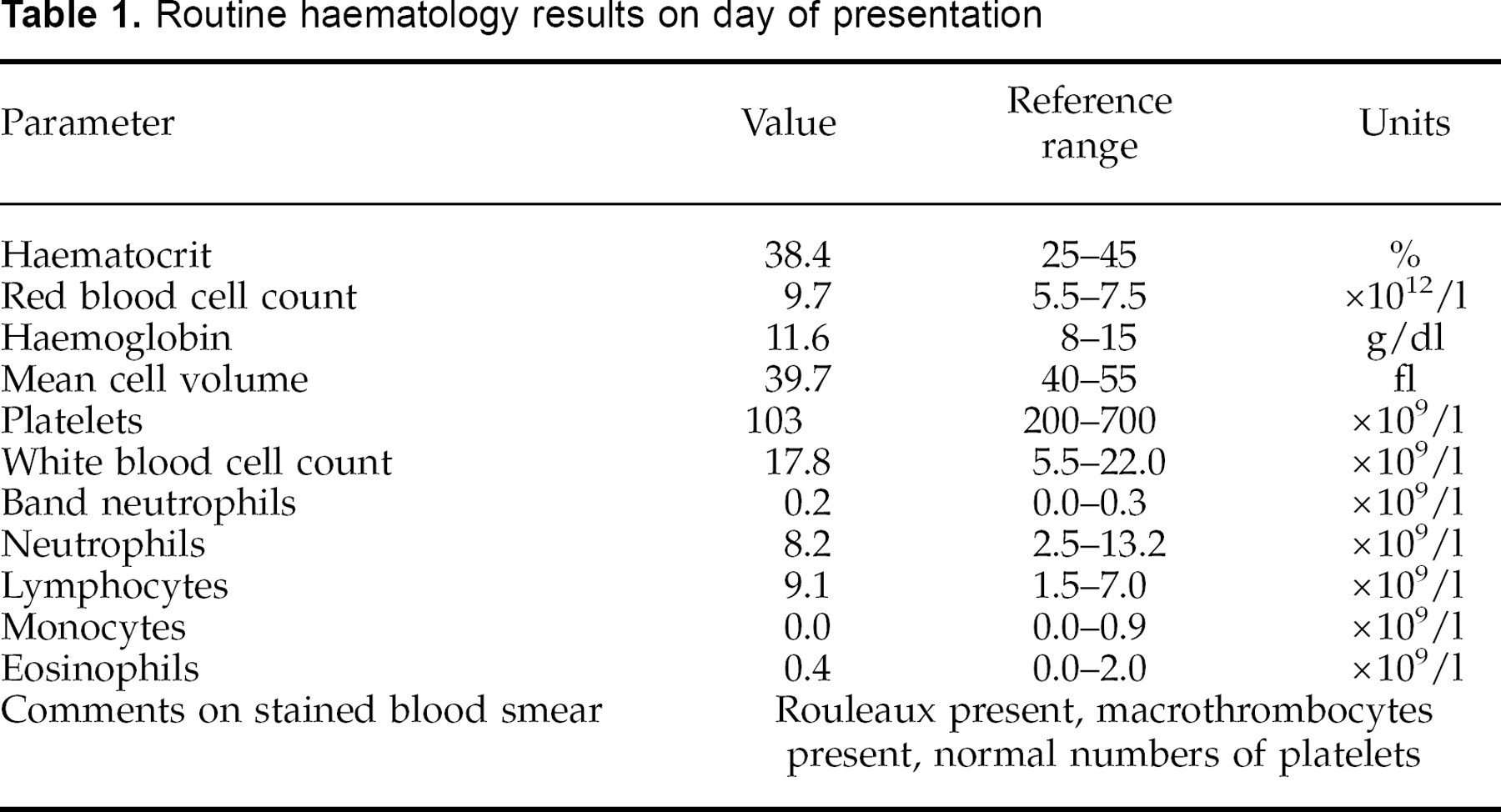
Routine haematology (Table 1) revealed an absolute lymphocytosis. Serum biochemistry (Table 2) showed hyponatraemia, hyperkalaemia, and hyperphosphataemia. Urinalysis revealed minimally concentrated urine (specific gravity of 1.017) but was otherwise unremarkable. Feline leukaemia virus and feline immunodeficiency virus tests were both negative. Lateral thoracic radiography revealed microcardia and pulmonary hypoperfusion (Figure 1), but abdominal radiography was normal.

Lateral thoracic radiograph illustrating microcardia and pulmonary hypoperfusion.
Routine haematology results on day of presentation
Serum biochemistry results on day of presentation and following treatment
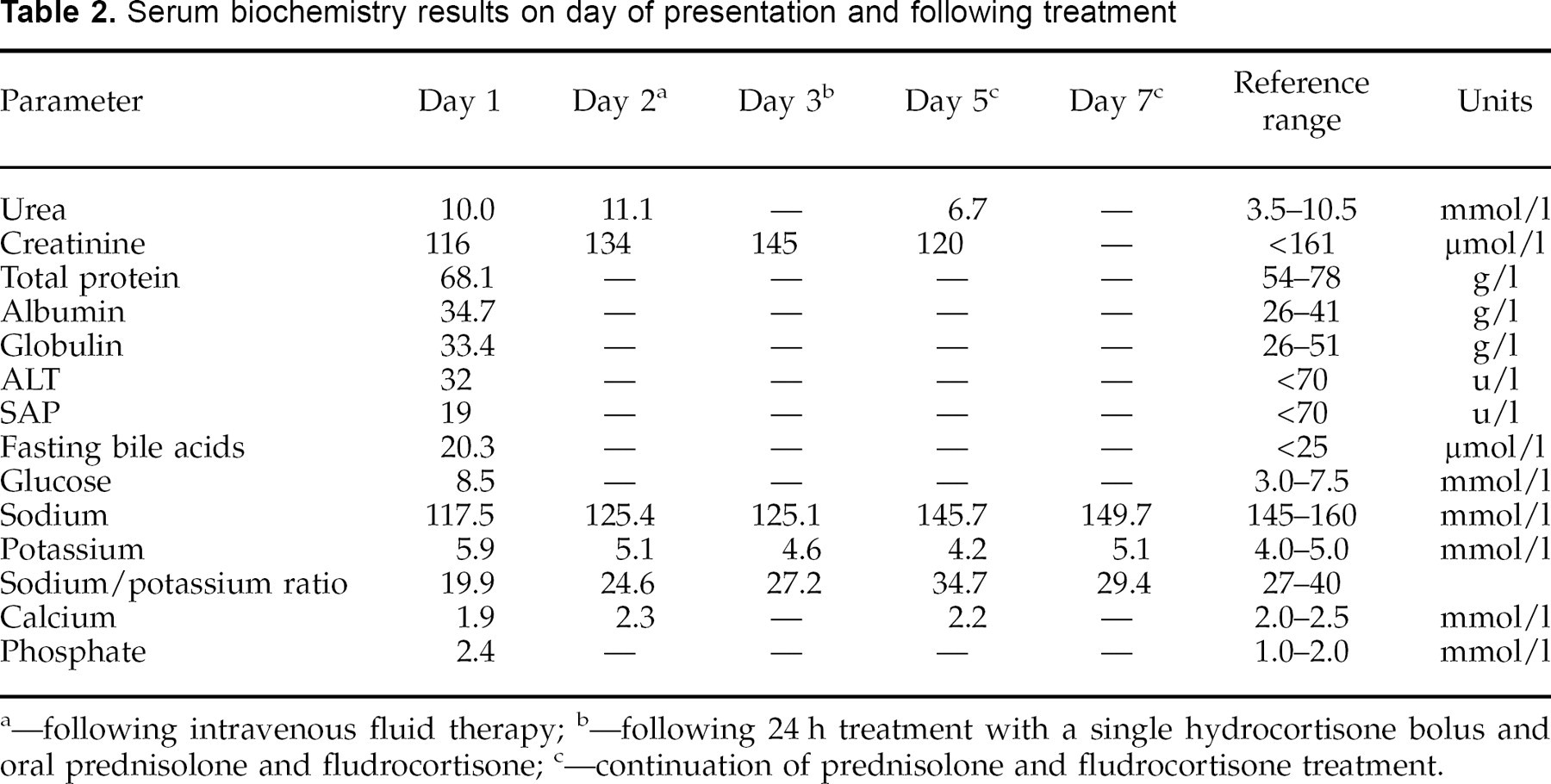
—following intravenous fluid therapy
—following 24 h treatment with a single hydrocortisone bolus and oral prednisolone and fludrocortisone
—continuation of prednisolone and fludrocortisone treatment.
The cat was given intravenous fluid therapy consisting of 0.9% NaCl at a rate of twice maintenance (10 ml/h) and was placed on a heat pad. Serum biochemistry was repeated the following day (Table 2) which revealed a mild increase in urea along with persistent hyponatraemia and hyperkalaemia. An ACTH stimulation test was subsequently performed by injecting 0.125 mg synthetic ACTH (tetracosactrin, Synacthen; Ciba Laboratories) intravenously after obtaining a basal blood sample, and repeating blood samples 60 and 180 min later for cortisol measurement (Sparkes et al 1990, Peterson & Kemppainen 1992).
As the clinical signs and laboratory findings were suggestive of hypoadrenocorticism, treatment for this disease was started on day 2 while awaiting the results of the ACTH stimulation test. A single bolus of 0.3 mg hydrocortisone was given intravenously followed by oral therapy with prednisolone (Prednicare; Animalcare Limited), 0.2 mg/kg daily and fludrocortisone acetate (Florinef; Squibb and Sons Limited), 0.1 mg once daily. In addition, a pinch of plain table salt was added to the cat's food. The ACTH stimulation test revealed a basal cortisol level of <20 nmol/l (normal range 20–270 nmol/l) which failed to increase at either 60 or 180 min post-ACTH, confirming a diagnosis of hypoadrenocorticism.
In the 4-day period following initiation of treatment, a clinical improvement was observed, with an increase in the cat's appetite and activity and no evidence of ataxia. Serum electrolyte levels normalised (Table 2), but mild hypothermia persisted despite attempts to warm the cat. A thyrotropin stimulation (TSH) test was performed to rule out concurrent hypothyroidism, but the results were normal, with serum thyroxine concentrations rising from a basal 33 nmol/l to 48 nmol/l 6 h after the intravenous injection of 1U bovine TSH (Sparkes et al 1991). In the following 2 days the hypothermia slowly resolved.
The cat was discharged following 6 days of treatment. Three weeks later, the cat was examined by the referring veterinarian, at which time no further clinical abnormalities were apparent. Repeat serum biochemistry including electrolytes was unremarkable. The cat is currently stable and asymptomatic on continuing treatment (prednisolone and fludrocortisone) 20 months after diagnosis.
The cat in this report presented with lethargy and weakness, signs commonly seen in cats with hypoadrenocorticism. In one report of 10 cases of feline hypoadrenocorticism, all presented with lethargy and nine cats showed weakness (Peterson et al 1989). Severe weakness can also create the illusion of ataxia as in this case. The cat in this report was 2 years old, which appears to be relatively young for the development of hypoadrenocorticism. In the 10 cases reported by Peterson et al, the ages ranged between 1.5 and 14 years with a median of 4.0 and a mean of 5.8. Other clinical findings commonly reported by Peterson included anorexia, weight loss, dehydration, and hypothermia; this cat exhibited all of these signs. Clinical signs reported less frequently by Peterson et al included vomiting, polyuria/polydipsia, and bradycardia, none of which were observed in this case.
Many of the laboratory abnormalities that are characteristic of hypoadrenocorticism were present in this case. Hyponatraemia and hyperkalaemia are the hallmark biochemical findings of this disease, with sodium-to-potassium ratios of less than 27:1 considered to be highly suggestive of hypoadrenocorticism in cats (Peterson et al 1989). This cat was both hyponatraemic and hyperkalaemic, and had a sodium-to-potassium ratio of less than 20:1 on the day of presentation. Azotaemia and hyperphosphataemia are also typical findings, reported in all of Peterson's 10 cases of feline hypoadrenocorticism. In this case, while hyperphosphataemia was present on day 1 (presumably due to reduced renal excretion), only a very mild elevation in urea was found on a single occasion (day 2). A marginally low serum calcium concentration was present initially, which subsequently normalised. Ionised calcium concentrations were not measured. In contrast, one of Peterson's 10 cases demonstrated hypercalcaemia, and approximately 30% of dogs with hypoadrenocorticism are reported to be hypercalcaemic (Feldman & Nelson 1996a), although the elevation in total calcium is usually mild and ionised calcium levels are often normal. On haematology, the only remarkable finding in this case was lymphocytosis, an abnormality also present in two of Peterson's 10 cases. Finally, poor renal concentrating ability in the face of any degree of azotaemia and dehydration, as documented in this case, is a recognised problem in cats (Peterson et al 1994) and other species with hypoadrenocorticism. The hyponatraemia impairs renal concentrating ability by decreasing the medullary solute gradient, and possibly also interferes with vasopressin release (Hardy 1995).
On radiography, pulmonary hypoperfusion and/or microcardia can be apparent as a result of the hypovolaemia associated with hypoadrenocorticism. These abnormalities were present in this case and in five of the 10 cats reported by Peterson.
An ACTH stimulation test is required to confirm a diagnosis of hypoadrenocorticism (Feldman & Nelson 1996b). The presence of a low basal cortisol level with little or no response to exogenous ACTH administration, as found in this case, is diagnostic for this condition. The timing of peak cortisol responses to intravenous administration of 0.125 mg ACTH has varied in different studies of healthy cats, resulting in several protocols for ACTH stimulation tests being derived. In one study, peak cortisol levels were seen 60–90 min after ACTH administration (Peterson & Kemppainen 1992), but in another study 60% of cats did not achieve peak levels until 180 min after ACTH administration (Sparkes et al 1990). Therefore in this case cortisol levels were determined before and both 60 and 180 min following intravenous ACTH administration in order to maximise the detection of any peak in cortisol production.
Initial treatment of hypoadrenocorticism in the cat requires aggressive intravenous fluid therapy to replace circulating blood volume and correct electrolyte imbalances in addition to glucocorticoid and mineralocorticoid supplementation. The cat in this report showed good clinical response to treatment over a 4-day period, similar to the response time seen in other cases (Peterson et al 1994). The basis of maintenance therapy is daily steroid replacement, usually with fludrocortisone acetate (mineralocorticoid) and physiological doses of prednisolone (glucocorticoid). The previously reported cats with hypoadrenocorticism were treated long-term with both mineralocorticoids and glucocorticoids (Peterson et al 1989), as was this cat.
The 20-month follow-up period for this case suggests that the prognosis for cats with hypoadrenocorticism receiving appropriate hormone replacement is very favourable.
