Abstract
Two cases of paroxysmal hemicrania (PH) associated with trigeminal neuralgia are reviewed. The paroxysmal hemicrania component in one patient was episodic, while it was chronic in the other. Each headache type responded completely to separate treatment, highlighting the importance of recognizing this association. We review the six other cases of chronic paroxysmal hemicrania-tic (CPH-tic) reported, and suggest that the term paroxysmal hemicrania-tic syndrome (PH-tic) be used to describe this association.
Keywords
Introduction
The association of chronic paroxysmal hemicrania with trigeminal neuralgia has been called the CPH-tic syndrome (1). The name was chosen because of the analogy to cluster-tic syndrome (1). We could identify six cases in the literature (2–4). After the second case was reported in 1998, Hannerz (5) commented that the patient probably had episodic paroxysmal hemicrania-tic (EPH-tic), not CPH-tic, because the patient was able to stop indomethacin without a recurrence of the paroxysmal hemicrania. Authors of a recent case report described a patient with remitting CPH-tic and debated whether their patient should be diagnosed with EPH-tic or CPH-tic (4). We review two cases of paroxysmal hemicrania, one episodic and the other chronic, associated with trigeminal neuralgia, and suggest that the term paroxysmal hemicrania-tic syndrome be used to describe this association (6).
Case 1
A 67-year-old, left-handed woman presented with a 15-year history of two main types of head pain.
The first type of pain had a usual duration of 20–30 min with a usual frequency of eight to 10 attacks per day. The attacks occurred during the night or the day, and displayed clustering. The bouts of pain would last 1–3 months with subsequent remission periods lasting 6–9 months. The pain was strictly unilateral, being localized to the right orbit, forehead, temple, nose, cheek, upper and lower teeth, jaw, and the lower lip. It was described as severe, sharp, and constant with superimposed throbbing and shooting pain. There was associated ipsilateral ptosis, eyelid swelling, lacrimation, and rhinorrhoea. She was agitated during an attack and preferred to move about. She felt nauseated and vomited during the attack, but denied photophobia, phonophobia, or osmophobia. She denied aura symptoms. Triggering factors included touching the lower lip, talking, chewing, cleaning her teeth, and wind blowing against her face. Alcohol had no triggering effect.
The second type of pain had a usual duration of 1–2 s. The usual frequency was four to five times per day, but it could occur once every 30 min. This pain type did not display attack clustering, and was present daily or near-daily from the onset. However, the frequency of the attacks seemed to increase during a bout of the first type of pain. The pain was again strictly unilateral involving the right lower lip and teeth radiating to the cheek, ear, and jaw. It was described as a sudden, sharp, severe, stabbing pain. The triggering factors were identical to those that led to her first type of head pain.
The attacks of the two types of pain could happen simultaneously, but they usually occurred independently. Interictally during a bout of the first type of pain, the patient also complained of a mild, featureless, continuous, dull ache on the right side of her face.
Medications at the time of consultation included tryptophan, hyoscine butylbromide, and ibuprofen 800 mg tds. Ibuprofen had no effect on her head pains. Other ineffective medications included verapamil at 80 mg bd, which was not tolerated, sodium valproate, and amitriptyline. Sumatriptan nasal spray and dihydrocodeine gave no relief. She could not tolerate high-flow oxygen because any pressure on her face would worsen the pain. On one occasion subcutaneous sumatriptan six mg was felt to shorten the duration of her first type of head pain from 30 to 20 min.
Her past medical history was significant for hypercholesterolaemia and an inguinal hernia repair. Her father had multiple sclerosis, and her mother had migraine. She drank 21 units of alcohol per week, and had quit smoking 3 months prior to neurological consultation, having previously smoked five cigarettes per day for 40 years. General and neurological examinations were normal. Magnetic resonance imaging (MRI) was normal, but magnetic resonance angiography (MRA) was not performed.
Her first type of headache responded completely to indomethacin at a dose of 50 mg tds. For 1 month after starting this medication, both types of pain were absent. She subsequently noted that the first type of pain would occur approximately 6 h after taking indomethacin, near the end of the dosing interval. Once the indomethacin-responsive pain recurred, the neuralgiform pain reappeared at a frequency higher than usual for the patient. Indomethacin was increased to 50 mg qds, which abolished the first type of pain but had no effect on the neuralgiform pain. The neuralgiform type of head pain subsequently responded completely to carbamazepine at a dose of 200 mg qds.
Case 2
A 60-year-old right-handed man presented with a 2-year history of two types of intermittent, daily head pains.
The first type of pain had a usual duration of 15–35 min, and a usual frequency of 10 attacks per day. The maximal attack frequency was 20 times per day. The pain was strictly unilateral, centred on the left forehead, temple, and orbit. It was described as excruciating. There was associated lacrimation, conjunctival injection, and nasal blockage. He denied nausea, vomiting, or photophobia. There were no triggers.
The second type of pain had a usual duration of 1 s, with a usual frequency of three to five attacks per day. The pain was again strictly unilateral, involving the left jaw, upper and lower teeth, forehead, and temple. It was described as a sudden, sharp, severe, stabbing or shooting pain. Unlike the first type of pain, the second type could be triggered by cleaning his teeth, chewing, cold drinks, and touching his face.
Most of the time the two types of pain occurred independently, but rarely they happened simultaneously. Interictally, the patient complained of a mild, featureless, continuous, dull ache over his left temple.
At the time of consultation, the patient was taking verapamil 80 mg bd without effect. Other ineffective medications included phenytoin, pizotifen, dothiepin, tramadol, and dihydrocodeine.
His past medical history was significant for hypertension. General and neurological examinations were normal. MRI of the brain was normal. MRA revealed a prominent left vertebral artery, and an ectatic vertebrobasilar junction/basilar artery in close relation to the left trigeminal root entry zone (Fig. 1).
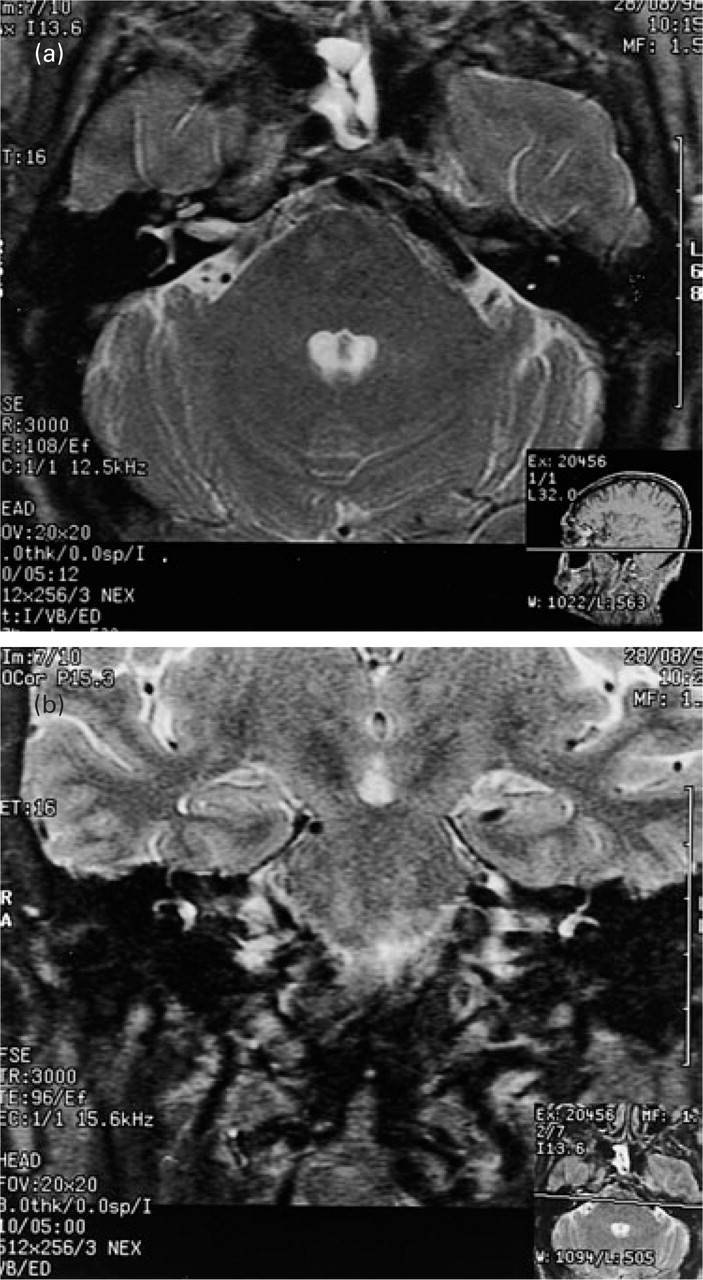
MRI/A axial (a) and coronal (b) sections showing an ectatic vertebrobasilar junction/basilar artery in close proximity to the left trigeminal nerve root entry zone.
His first type of headache responded completely to indomethacin at a dose of 200 mg per day. Verapamil was discontinued. Unfortunately he developed gastritis and duodenitis with gastrointestinal bleeding, so the indomethacin had to be stopped. He was therefore restarted on verapamil and had an incomplete response on 480 mg per day. While on verapamil, the neuralgiform type of head pain responded completely to carbamazepine at 300 mg bd, but he developed a significant rash and carbamazepine had to be stopped. Verapamil alone had no effect on the neuralgiform pain, so topiramate was added. Topiramate at a dose of 25 mg per day slightly decreased the severity of the indomethacin-responsive type of head pain but not the frequency. Topiramate at this dose was more effective in treating the tic pain, although the effect was incomplete. The patient was tried on rofecoxib 25 mg per day, but this had no effect on his indomethacin-responsive head pain, so it was stopped. At the time of publication, the patient was on verapamil 480 mg per day and topiramate 300 mg per day. On this combination he noted decreased severity of both types of head pain, but the response was incomplete.
Discussion
Both patients reviewed had two types of head pain. One type was approximately 20–30 min in duration, occurred frequently, was associated with autonomic features, and responded completely to indomethacin. Because of the clustering of attacks with subsequent pain-free months seen in the first patient, one could use the term EPH-tic to describe the phenotype. The second patient would then be given the diagnosis of CPH-tic as his attacks did not occur in discrete bouts. Both of these patients would satisfy the International Headache Society (IHS) criteria for CPH (7). In one patient the indomethacin-responsive pain was triggerable, while in the other it was not. The patients also had a brief, triggerable, neuralgiform type of pain that responded to carbamazepine and would fit the IHS criteria for trigeminal neuralgia (7). In the first patient there was a 1-month period where indomethacin seemed to stop both types of pain, but this was not sustained. In general, each type of pain required separate, specific treatment. The locations of the two types of pain overlapped, and the attacks of the two types of pain could occur concurrently or non-concurrently in both patients. Both patients complained of an interictal, ipsilateral dull ache while experiencing the indomethacin-responsive type of pain.
Table 1 reviews the clinical data from the eight PH-tic cases reported to date. There are still too few cases to comment categorically on the typical phenotype, but some generalizations can be made. Attacks can occur concurrently, non-concurrently, or both, as exemplified by our two patients. The paroxysmal hemicrania component of the syndrome is often episodic. In six of eight patients, the PH was episodic at some time during the illness, and in four of eight it was episodic at the time of the case report. Thus the paroxysmal hemicrania attacks may start and remain episodic throughout the illness, making the term CPH-tic confusing. The tic component is always triggerable to some degree, and in some cases (five of eight) the PH component is also triggerable.
Clinical features of the paroxysmal hemicrania-tic syndrome

∗For purposes of this table, temple pain=V3 pain.
†Concurrent=attacks of the two types of pain overlap temporally once both types have started to occur.
‡Non-concurrent=attacks of the two types of pain do not overlap temporally once both types have started to occur.
In six of the eight patients, each type of pain required a separate treatment, analogous to the usual circumstance in cluster-tic (8–11). Our second patient had an ectatic basilar artery close to the trigeminal root entry zone, ipsilateral to the side of his head pains. No patients with PH-tic have had vascular decompression procedures yet, but the four cluster-tic patients described by Solomon had relief of their tic but not their cluster headaches after this procedure (8). Because of the distinct treatment responses, it is important to question closely for both types of pain during the headache history.
In two case reports, indomethacin has relieved both types of pain (2, 3). In this regard, it is interesting that our first patient had 1 month of complete pain relief while on indomethacin alone, but subsequently redeveloped tic at an increased frequency when the indomethacin dosage was no longer adequate to control her PH pain. This patient also noted that her daily tic pains were more frequent during bouts of concomitant PH pain. This implies that one type of pain has an influence on the other in the PH-tic syndrome.
The relationship between PH and trigeminal neuralgia is not completely clear. The two conditions might occur coincidentally, or alternatively could be comorbid (6). It may be that the brain abnormality that causes PH allows a peripheral stimulus in trigeminal neuralgia to be more readily expressed (6). Given the hypothalamic activation seen in cluster headache (12, 13) and SUNCT attacks (14), and the phenotypic similarity between PH, cluster, and SUNCT, it seems likely that a CNS mechanism underlies the aetiology of PH. Fromm has stated that trigeminal neuralgia has a peripheral cause and a central pathogenesis (15). Perhaps in PH-tic, the CNS abnormality includes impaired inhibitory mechanisms that normally control afferent activity in the trigeminal nucleus (16), as well as hypothalamic dysfunction.
Finally, we recommend that the term paroxysmal hemicrania-tic syndrome be used rather than CPH-tic or EPH-tic. In this regard it is useful to remember that Hannerz first suggested the term CPH-tic because of the analogy to cluster-tic. The cluster component of cluster-tic can be episodic or chronic, but this is not spelled out in the title. In this light we propose that PH-tic be noted in the revised IHS system (17) and the patients be coded with two phenotypes for research purposes.
Footnotes
Acknowledgements
M.S.M. is a Migraine Trust Research Fellow. P.J.G. is a Wellcome Trust Senior Research Fellow.
