Abstract
This paper reports an analysis of two randomized controlled trials of rizatriptan, in which the 24-h Migraine Quality of Life Questionnaire© was used to assess migraine-specific quality of life in patients receiving acute treatment. The objective of the analysis was to determine which clinical effects of a migraine medication, as measured by traditional clinical trial endpoints, contribute to a better short-term health-related quality of life. The results demonstrate that patients who experience complete pain relief and are able to function at their normal ability within 2 h and experience no headache recurrence have the highest migraine-specific quality of life scores. Patients who were satisfied with medication at 2 h had higher migraine-specific quality of life scores than those who were not satisfied. In conclusion, migraine therapy that provides rapid, complete, and sustained pain relief, with restoration of functional ability, has the most beneficial impact on short-term health-related quality of life for migraineurs.
Introduction
Migraine imposes a considerable burden on sufferers, both in terms of impairment of daily activities during an attack and limitations in lifestyle in anticipation of attacks (1). For example, migraine is a significant cause of absence from work or school and reduced productivity at work, and is thus a possible cause of reduced educational and occupational achievement (2–5). Home life is also affected, as many sufferers report that they are unable to carry out household tasks or care for their families during a migraine attack (6–8). In addition, migraineurs may limit their social and work activities in order to avoid triggering factors or due to fear of having a migraine attack (9). The burden of migraine does not only fall on the individual sufferer and their families, but also on society. For example, it has been estimated that in the USA, migraine costs employers $13 billion each year due to missed work days and reduced productivity at work (10).
Effective treatment of migraine attacks has been shown to improve patients' health-related quality of life. Some studies have investigated long-term effects of treatment; for example, patients treated with sumatriptan experienced health-related quality of life (HRQoL) improvements as measured by generic and disease-specific quality of life instruments (11–13). However, as these studies were not controlled, it is difficult to determine whether the observed improvements were directly caused by the treatment. Another approach is to assess health-related quality of life over the time of an individual migraine attack with or without treatment. The 24-h Migraine Quality of Life Questionnaire© (24-h MQoLQ©) is a validated questionnaire that assesses short-term migraine-specific quality of life (14, 15). The 24-h MQoLQ was developed to examine the impact of the migraine attack and its treatment on the patient's life over a clinically relevant, focused period of time. The results of the validation study suggested that short-term migraine-specific quality of life was associated with better clinical outcomes of pain relief and associated symptoms (15). Use of this questionnaire demonstrated that patients receiving rizatriptan 10 mg experienced significantly (P=0.004) better migraine-specific quality of life compared with those on placebo (16).
Given that effective migraine treatment can improve patients' health-related quality of life, what are the specific attributes of effective therapy that are associated with this benefit? The objective of this analysis was to determine which treatment outcomes are associated with short-term migraine-specific quality of life after treatment of acute migraine episodes. Since an important goal of treatment is to improve HRQoL, the endpoints which best predict improved HRQoL may merit special consideration in the evaluation of migraine therapies.
Methods
Study data used in the analysis
Data from two multicentre, randomized, double-blind, parallel group clinical trials were used in this analysis (17, 18). Two studies were used in order to examine the consistency of the results.
Study 1 (17) was a crossover trial comparing rizatriptan 10 mg with placebo in the treatment of four discrete migraine attacks. A total of 473 patients from 23 sites in the USA were randomized to one of five treatment sequence groups. Patients in four of the groups received rizatriptan 10 mg for three out of four attacks and placebo for the remaining attack (placebo was given for the first, second, third, or forth attack in each of the respective sequence groups). Patients in the fifth sequence group received rizatriptan 10 mg for all four attacks.
Study 2 (18) involved 1268 patients recruited at 47 sites in 21 countries (including Canada, Latin America, Europe, the Middle East, South Africa, and Australia). Patients were randomized to receive rizatriptan 5 mg, rizatriptan 10 mg, sumatriptan 100 mg, or placebo for the treatment of a single migraine attack.
In both studies, patients were instructed to take study medication when they developed a moderate or severe migraine headache that did not start to resolve spontaneously. Patients who still had a moderate or severe headache at 2 h after taking study medication could take additional rescue analgesia (opiates, paracetamol, or non-steroidal anti-inflammatory drugs).
Outcome measures
In both studies, each patient recorded outcome measures in a diary at the following time points: pre-dose, and at 0.5, 1, 1.5, 2, 3, and 4 h after dosing. At each time point, patients rated headache pain intensity on a four-point scale (0=no headache; 1=mild; 2=moderate; or 3=severe pain) and functional ability on a four-point scale (0=normal; 1=daily activities mildly impaired; 2=severely impaired; or 3=unable to do activities, requires bed rest). Associated symptoms of photophobia, phonophobia, nausea, and vomiting were rated pre-dose and at each subsequent time point on a dichotomous scale (0=no and 1=yes).
At 2 h after taking study medication, patients completed a seven-point satisfaction with migraine medication question (1=completely satisfied, couldn't be better; 2=very satisfied; 3=somewhat satisfied; 4=neither satisfied nor dissatisfied; 5=somewhat dissatisfied; 6=very dissatisfied; or 7=completely dissatisfied, couldn't be worse).
Patients recorded in their diaries the incidence of headache recurrence. A headache recurrence was defined as the return of a moderate or severe headache within 24 h in patients who had initially responded (i.e. had no headache or mild pain) to study treatment within 2 h.
Twenty-four hours after the onset of each treated migraine attack, patients completed the 24-h MQoLQ. The questionnaire consists of 15 questions covering five domains that were found important to patients during an acute migraine attack: work functioning, social functioning, energy and vitality, migraine symptoms, and feelings or concerns. Each question was scored on a seven-point scale where 1 indicated maximum impairment and 7 indicated no impairment; each domain could therefore have a maximum score of 21 (best HRQoL) and a minimum score of 3 (worst HRQoL).
Statistical analysis
The analysis used data on pain intensity, functional ability, and associated symptoms at 2 h after taking study medication in order to avoid confounding from the use of rescue medication after 2 h. Pain status at 2 h was defined as: no relief= moderate or severe pain (score= 2 or 3); headache relief= mild or no pain (score= 1 or 0); pain free= no pain (score= 0); sustained response= headache relief and no recurrence; and sustained pain free= pain free and no recurrence. Functional ability was categorized as normal (score= 0), slight impairment (score= 1), or severe impairment (score= 2 or 3). Satisfaction with migraine therapy was categorized as satisfied (scores 1–3) or not satisfied (scores 4–7). The relationship between the 24-h MQoLQ domain scores and pain status, satisfaction, and functional ability was explored using mixed model analyses for the multiple attack study (study 1) and simple linear regression for the single attack (study 2). For study 1, all attacks were included in a mixed model assuming equal correlation between headache characteristics and MQoLQ within patients across multiple attacks. Since the variables examined in these analyses from these two clinical trials were found to be highly correlated with one another, a model examining the independent contribution of each predictor (i.e. pain relief, functional status) was not possible. The purpose of this analysis was to examine the relationships between the migraine-related quality of life measure and other patient-reported outcomes regardless of treatment group, and therefore treatment assignment was not included in the models.
Results
A total of 407 patients in study 1 and 1099 patients in study 2 took medication and so were included in the analysis. Patient characteristics were similar in the two study populations; the mean ages of the patients were 40.6 years and 38.2 years, and 84.0% and 82.0% of patients were women in study 1 and study 2, respectively.
The relationship between pain status at 2 h, recurrence of headache between 2 h and 24 h, and 24-h MQoL score in the two studies is shown in Fig. 1. Across the five domains, patients with no headache relief had the lowest quality of life scores, while those who were pain free with no recurrence (sustained pain free) had the highest migraine-specific quality of life scores, typically 5–6 points higher than those with no relief. Notably, patients who were pain free and experienced no recurrence had scores that were 1–2 points higher than those who were pain free but did have headache recurrence. Similarly, among patients who had headache relief, those without headache recurrence had migraine-specific quality of life scores that were 2–3 points higher than those who did experience recurrence of their migraine attack.

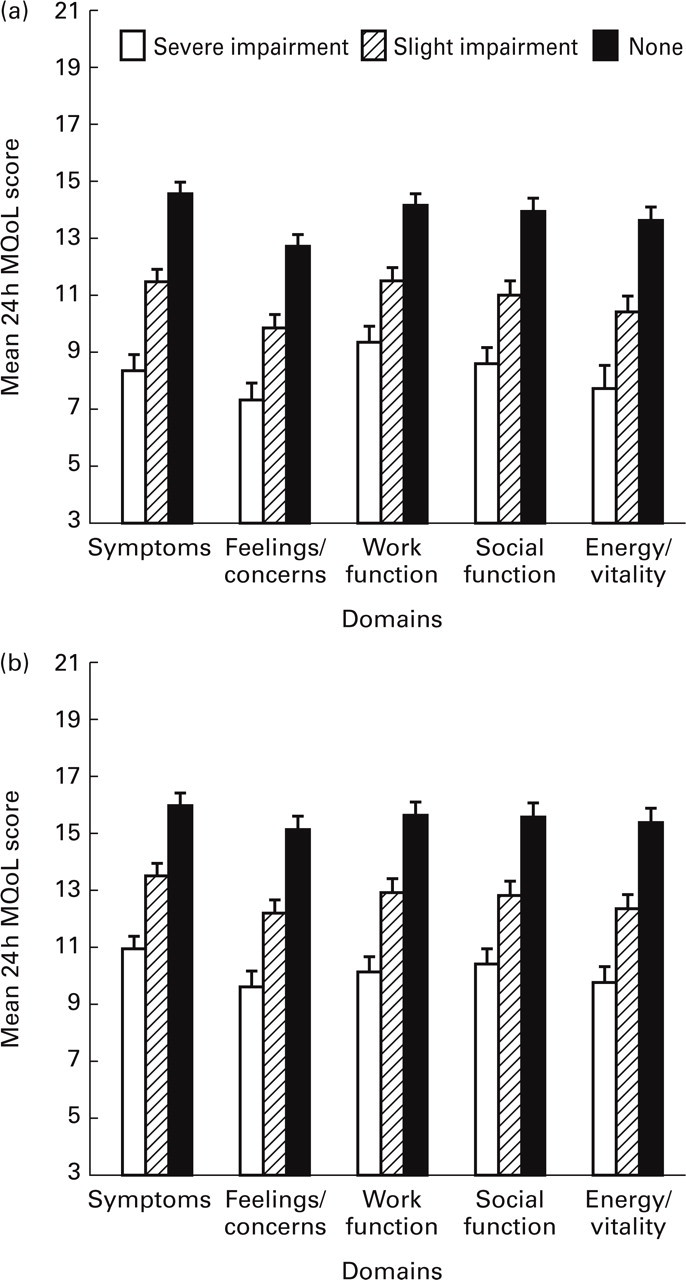
There was a clear relationship between functional ability and quality of life in both studies (Fig. 2). In all five domains, patients who rated themselves as having no impairment in their functional abilities at 2 h had higher migraine-specific quality of life scores than those who had slight or severe impairment. A similar relationship was seen between satisfaction with migraine treatment and migraine-specific quality of life (Fig. 3). In all five migraine quality of life domains, patients who were at least somewhat satisfied with the effects of their treatment at 2 h scored higher on the 24-h MQoL questionnaire than those who were dissatisfied with their treatment.

Discussion
This analysis of two large randomized studies of rizatriptan in the treatment of migraine has demonstrated that patients who are free of pain at 2 h and who have no recurrence over 24 h have the highest scores on a validated measure of migraine-specific quality of life (MQoL), the 24-h Migraine Quality of Life Questionnaire. The group with headache relief did far better than the group with no relief. Headache recurrence was associated with reduced migraine-specific quality of life both in patients with headache relief and in those with pain-free status. Restoration of function at 2 h also predicted MQoL over the subsequent 24 h. These findings were consistent across two independent clinical trials, one conducted in the USA and the other conducted on six continents. The relationships we describe therefore seem broadly generalizable and not tied to a particular culture or geographical area.
Results from survey research support the findings of this study. In a telephone survey of 688 patients with migraine, conducted to determine their preferences and needs regarding the characteristics of migraine treatments, complete relief of pain was the characteristic most commonly identified as very important or important, with lack of recurrence of pain and rapid onset of pain relief ranked next in importance (19). In the present study, similar characteristics were identified as determinants of better HRQoL: those who were completely free of migraine pain within 2 h experienced better quality of life than those who had only partial headache relief. Pain-free response is now generally accepted as a stringent and desirable endpoint in migraine clinical trials. These results support the importance of this endpoint, as it is a better predictor of MQoL than headache response.
Medication that relieves pain and allows individuals to return to their daily activities as soon as possible is clearly desirable to patients. Rapid onset of pain relief was one of the characteristics of migraine treatment identified as desirable by patients (19), and a recent study has demonstrated a close association between time to onset of pain relief and patient satisfaction with their migraine treatment (20). This analysis did find a strong relationship between satisfaction with migraine treatment at 2 h and MQoL at 24 h.
This report demonstrates that patients who had normal functional ability at 2 h had higher migraine-specific quality of life scores than those with slight or severe functional impairment. This analysis also demonstrates that lack of headache recurrence was an important determinant in migraine-specific quality of life both in patients with relief and in patients free of pain at 2 h.
The 24-h MQoLQ has demonstrated that effective migraine therapy, such as treatment with rizatriptan, is superior to placebo on measures of migraine-specific quality of life (21). Over four separate migraine attacks assessed, MQoL scores in the rizatriptan group were approximately 2–4 points higher than those in the placebo group, similar to the differences seen in the current analysis between patients who did not and patients who did experience headache recurrence, and between those who experienced headache relief and those who were pain free.
For the analyses presented in this paper, we categorized patients into groups based on the endpoints they achieved (i.e. ‘no relief’, ‘headache relief’, or ‘pain-free’ at 2 h, with or without subsequent recurrence) independent of the treatment they received. This artificial grouping is useful in determining the relationships among clinical trial endpoints and migraine-specific quality of life. In reality, however, not all patients in a treatment group will achieve either headache relief or freedom from pain with a migraine therapy within 2 h, and of those who achieve relief, a certain percentage will go on to experience a recurrence. In making treatment decisions, the objective is to optimize the proportion of patients achieving these desirable states, while recognizing that not all patients will respond at this level for every migraine attack. None the less, we believe that this analytic approach is useful for identifying the endpoints of treatment which predict better MQoL during an attack. These analyses have shown that patients treated with migraine therapy who experience complete and sustained pain relief, and those who experience normal functional ability at 2 h, have better short-term health-related quality of life, as measured by the validated 24-h MQoL questionnaire.
