Abstract
This study investigates the CNS effects of sumatriptan and rizatriptan, with temazepam as an active comparator, in healthy female volunteers. Sixteen volunteers completed a randomized, double-blind, crossover study and on four separate occasions received either 100 mg sumatriptan, 20 mg rizatriptan or 20 mg temazepam. The main parameters were eye movements, EEG, body sway, visual analogue scales and a cognitive test battery. Rizatriptan and sumatriptan decreased saccadic peak velocity by 18.3 (95% CI: 5.7, 30.8) and 15.0 (2.2, 27.9)°/sec, respectively, about half the decrease induced by temazepam (35.0 (22.1, 47.8)°/sec). Body sway increased (30% for rizatriptan (16%, 45%) and 14% for sumatriptan (1%, 27%), respectively). Temazepam caused larger, similar effects. In contrast to temazepam, sumatriptan and rizatriptan decreased reaction times of recognition tasks and increased EEG alpha power (significant for sumatriptan, 0.477 (0.02, 0.935). Therapeutic doses of sumatriptan and rizatriptan caused CNS effects indicative of mild sedation. For EEG and recognition reaction times the effects were opposite to temazepam, indicating central stimulation.
Introduction
5HT1B/D agonists are becoming increasingly popular in the treatment of migraine attacks. This type of drug is able to activate the 5HT1-like receptors on the main cranial vessels, resulting in vasoconstriction and thus providing relief of symptoms (1). The popularity of the 5HT agonists in the acute treatment of migraine is mainly due to the high efficacy and the relatively low rate of side-effects compared with other treatments.
The first registered 5HT1B/D agonist was sumatriptan, at a recommended oral dose of 50 mg followed by another 50 mg 2 h later in case of recurrence of symptoms. Following oral administration sumatriptan has a bioavailability of 14% (2), which is controlled by dosing to effective concentrations. Sumatriptan is metabolized by monoamineoxidase (MAO) to an inactive N-desmethyl metabolite; the elimination half-life is around 2 h (3). Other triptans quickly followed sumatriptan. Although these triptans demonstrate increased bioavailability over sumatriptan they appear to offer few clinical advantages.
Rizatriptan has been demonstrated to be absorbed more rapidly compared with sumatriptan (4) and has a bioavailability of 40–45% after oral administration (5), with an elimination half-life of around 2 h (3). It is metabolized by MAO into an active N-desmethyl metabolite and may have a longer effect.
It has been claimed that rizatriptan has superior efficacy compared with sumatriptan, whereas at therapeutically relevant doses there are indications that rizatriptan may cause more adverse events, such as dizziness or drowsiness, compared with sumatriptan (6–8). The mechanisms involved in these adverse events, and the best methods to quantify them, are unknown. It has long been assumed that triptans were not able to penetrate the central nervous system, which might have led to a relatively low number of studies and assessments of these types of adverse effects.
The current study investigated the effects of clinical doses of sumatriptan and rizatriptan on the central nervous system, compared with placebo and temazepam as an active comparator for dizziness and drowsiness. An array of neuropsychometric parameters was used that has been shown to be sensitive to sedative agents (9–11). Additionally, the relationship between the plasma concentrations and pharmacodynamics of sumatriptan and rizatriptan was evaluated. The study was performed in female subjects only, because of the higher prevalence of migraine in females.
Methods
Subjects
After approval of the protocol by the Ethical Review Board of the Leiden University Medical Centre, 16 healthy female volunteers were recruited, plus one replacement subject. Written informed consent was obtained before entering the study. After this the subjects received a full medical examination. Drugs were administered to the participants only after a negative pregnancy test (Pacific Biotech Inc., San Diego, USA) and urinary drug-screen (ONTRAK Rapid Assays for Drug abuse; Roche (diagnostic systems), Mijdrecht, The Netherlands) had been obtained on the morning of each study day. Volunteers refrained from nicotine, alcohol and caffeine from 48 h prior to each study day until 24 h after dosing. Strenuous exercise was to be avoided for 48 h before each study safety screen and 12 before and after each dose. Concomitant medication was not permitted during each study period.
Design and procedure
The study was performed as a double-blind, randomized, placebo-controlled four period cross-over comparative trial with a minimum washout period of 3 days and a maximum washout period of 1 week. The study medication contained sumatriptan 50 mg tablets, rizatriptan 10 mg encapsulated tablets, temazepam 20 mg capsules and placebo tablets and capsules. Rizatriptan and temazepam were encapsulated into identical hard gelatine capsules. On each study day subjects received either: a sumatriptan 50 mg tablet and a placebo capsule followed by the same treatment after 2 h; rizatriptan 10 mg capsule and a placebo tablet followed by the same treatment after 2 h; a placebo tablet and capsule followed by temazepam 20 mg capsule and a placebo tablet after 2 h; or a placebo tablet and capsule, followed by the same treatment after 2 h. The triptans were administered according to clinical practice for repeated dosing. Temazepam was administered at 2 h to have a comparable tmax for temazepam and the triptans (3–5 h after initial dose).
All tests (except the cognitive test battery) and blood sampling were performed before the intake of medication and at the following times after the first intake: 20, 40 min, 1, 1.5, 2, 2.5, 3, 4, 6, 8, 10 and 12 h. The cognitive test battery was executed in the 3–5-h period after the first administration of the study drug. Adverse events were checked regularly with open-ended questions (‘How do you feel?’). Before the intake of medication (around 09.15) a light standard breakfast was served. Standard lunch and dinner were served 5 and 8 h post-dose.
Pharmacodynamics
Eye movement analysis
Recording and analysis of saccadic and smooth-pursuit eye movements was performed with a microcomputer-based system, as described previously (12). The equipment used for stimulus display, signal collection and amplification was from Nihon Kohden (Nihon Kohden Corporation, Tokyo, Japan). Saccadic eye movements were recorded for stimulus amplitudes of 15 degrees to either side. Interstimulus intervals varied randomly between 3 and 6 s, 15 saccades were recorded. The average values of saccadic peak velocity, latency (reaction time) and inaccuracy were used as parameters. For smooth-pursuit eye movements the target moved sinusoidally at frequencies ranging from 0.3 to 1.1 Hz, increasing by steps of 0.1 Hz. The amplitude of target displacement corresponds to 20 degrees eyeball rotations to both sides. Four cycles were recorded for each stimulus frequency.
Electroencephalograms
Electroencephalograms were recorded and analysed using CED software (Cambridge Electronics Design, Cambridge, UK), as described previously (13). EEG recordings were made using silver-silver chloride electrodes, fixed with collodion at Fz, Cz, Pz and Oz, with the same common ground electrode as for the eye movement registration (international 10/20 system). The electrode resistances were kept below 5 kOhm. All recordings were done with the subjects' eyes closed. EEG signals were obtained from leads Fz-Cz and Pz-Oz. The signals were amplified by use of a Nihon Kohden AB-621G bioelectric amplifier (Nihon Kohden Corporation) with a time constant of 0.3 s and a low pass filter at 100 Hz. Eight consecutive blocks of 8 s were recorded per session over a 2-min period. The sampling rate was 1024 Hz. Datablocks containing artefacts were identified by visual inspection and these were excluded from analysis. Fast Fourier transform analysis was performed to obtain the sum of amplitudes in the delta (0.5–3.5 Hz), theta (3.5–7.5 Hz), alpha (7.5–11.5 Hz) and beta (11.5–30 Hz) frequency ranges. The total recording bandwidth was 0–50 Hz.
Body sway
Body sway was measured using an apparatus similar to the Wright ataxiameter (11), which adds up the amplitude of body movement transferred through a string that is attached to the waist (apparatus: TNO/NIPG, Leiden, The Netherlands). Measurements of sagittal body sway (forwards/backwards) were made for 2 min with the eyes closed while standing on an unstable surface consisting of a 10-cm foam pad covered with a rigid plateau. The subjects were standing comfortably with their feet slightly apart and arms along their body.
Visual analogue scales
Visual analogue scales as originally described by Norris (14) were used to quantify subjective effects. Three factors corresponding to alertness, mood and calmness were derived from these measurements (15).
Cognitive test battery
Cognitive performance was tested using the ‘FePsy’ software package (Instituut voor Epilepsiebestrijding, Heemstede, The Netherlands) (16). This system contains a battery of computerized tests of different complexity measuring diverse cognitive skills. The battery has been extensively validated in epilepsy patients (17, 18). The following tests were performed:
Simple auditory and visual reaction time, measuring attention.
The binary choice task, a more complex reaction time task measuring information processing and response inhibition.
The visual searching task, which evaluates the visual (complex) information processing and perceptual–mental strategies. The main parameter of the visual searching task was the number of errors, the secondary endpoint was the mean searching time of correct response (19, 20).
The card sorting task, which assesses abstraction ability, conceptualization, failure to maintain set and perseverance (21).
Serial recognition of words and figures and simultaneous recognition of words and figures, which are recognition and short-term memory tasks.
The Corsi Block Tapping test (22), a direct recall memory test.
Pharmacokinetics
Blood samples were drawn from an intravenous cannula inserted into a forearm vein. The blood samples were immediately centrifuged after collection at 4°C at 1000
Sample times were related to the time of (first) dose (for study drugs administered twice). Pharmacokinetics for all drugs were assessed using non-compartmental methodology as implemented in WinNonlin V2.1 (Pharsight Corporation, Mountainview, California, USA). For each subject and each treatment the following pharmacokinetic parameters were determined: maximum plasma concentration (Cmax), the time at which the maximum concentration was reached, relative to dosing (tmax), the terminal halflife (t1/2) and the area under the plasma concentration-time curve up to the last quantifiable concentration (AUClast) and extrapolated to infinity(AUCinf). Descriptive statistics were reported for each pharmacokinetic parameter. The results for sumatriptan and rizatriptan were compared with the literature and with data on file to verify that the bioavailability was not altered by the reformulation.
Statistics
All repeatedly measured dynamic variables were characterized using changes from pre-treatment value expressed as areas under the effect curve (AUECs). These AUECs were calculated using the linear trapezoidal rule and were divided by the corresponding time-span, resulting in a time-weighted average outcome. AUECs were calculated over the entire sampling period (12 h) and over the first 6 h because most of the response was expected to occur during this period. Results are only reported relative to baseline over the 0–6-h period.
The cognitive test results were compared among treatments using standard analysis of variance methods. The primary contrasts were sumatriptan vs. rizatriptan, sumatriptan vs. placebo and rizatriptan vs. placebo, calculated within the
The relationship between concentrations and effects was assessed by plotting the effect vs. the interpolated concentration. The interpolated concentration was used because of a time difference between blood samples and dynamic parameters. Placebo correction was implemented by subtracting the response during the placebo occasion from the response during active treatment at corresponding nominal times on an individual basis. The relationships between interpolated concentrations and placebo corrected SPV and log body sway were assessed by calculating linear concentration-effect relationships using non-linear mixed effect modelling (NONMEM version V; NONMEM Project Group, UCSF, California, USA). This procedure analyses all curves collectively while preserving differences between individuals. Different slope and intercept estimates for the different drugs were implemented. Additive intra- and interindividual error models were used and parameters were estimated using first order conditional estimation (FOCE) with the ‘interaction’ option. The significance of slope parameters was assessed by calculating 95% confidence intervals using the reported standard errors.
Results
Sixteen subjects were initially recruited. One subject repeated a dosing period, due to a non-treatment-related adverse event that led to discontinuation of the third study day. One subject was replaced because of a non-serious adverse event during the third study day. The mean age of the 17 participants was 21.4 years (range 18–28). Body mass index (weight (kg)/length (m)2) was below 30 for all participants. The pharmacodynamic results are based upon 16 subjects who completed all treatment periods and one subject who completed two dosing periods. The pharmacokinetic results are based upon plasma concentrations of 17 subjects for rizatriptan and 16 subjects for sumatriptan and temazepam. The safety analysis was performed on all 17 subjects that received one or more treatments.
Adverse events
In all treatment groups sedation (40%), headache (19%) and dizziness (9%) were recorded most. Drug-related adverse events, as judged by the investigator, were also mainly sedation (75%), headache (13%) and dizziness (11%). No large differences were seen between the different treatments, but temazepam appeared to induce slightly more sedation of moderate intensity than the other treatments (Table 1). Events were generally mild and did not require treatment. Two subjects experienced moderate adverse events (flu-like symptoms) that led to the discontinuation of a trial day. During those events paracetamol was used for symptomatic treatment.
Summary of main adverse events (number reported)

Pharmacodynamics
Eye movements
Both rizatriptan and sumatriptan caused a small but statistically significant reduction in saccadic peak velocity of −18.3 (− 30.8, −5.7) °/s (mean difference, 95% confidence interval) and −15.0 (− 27.9, −2.2) °/s, respectively, compared with placebo. This was about half the effect of temazepam, which caused a reduction of −35.0 (− 47.8, −22.1) °/s (Fig. 1). The difference of 3.3 (− 9.6, 16.1) °/s between the two triptans was not statistically significant. The triptans did not have significant effects on saccadic latency or inaccuracy. Temazepam showed a clear but non-significant effect on the smooth-pursuit eye movements. Both triptans did not alter smooth-pursuit eye movements compared with placebo.
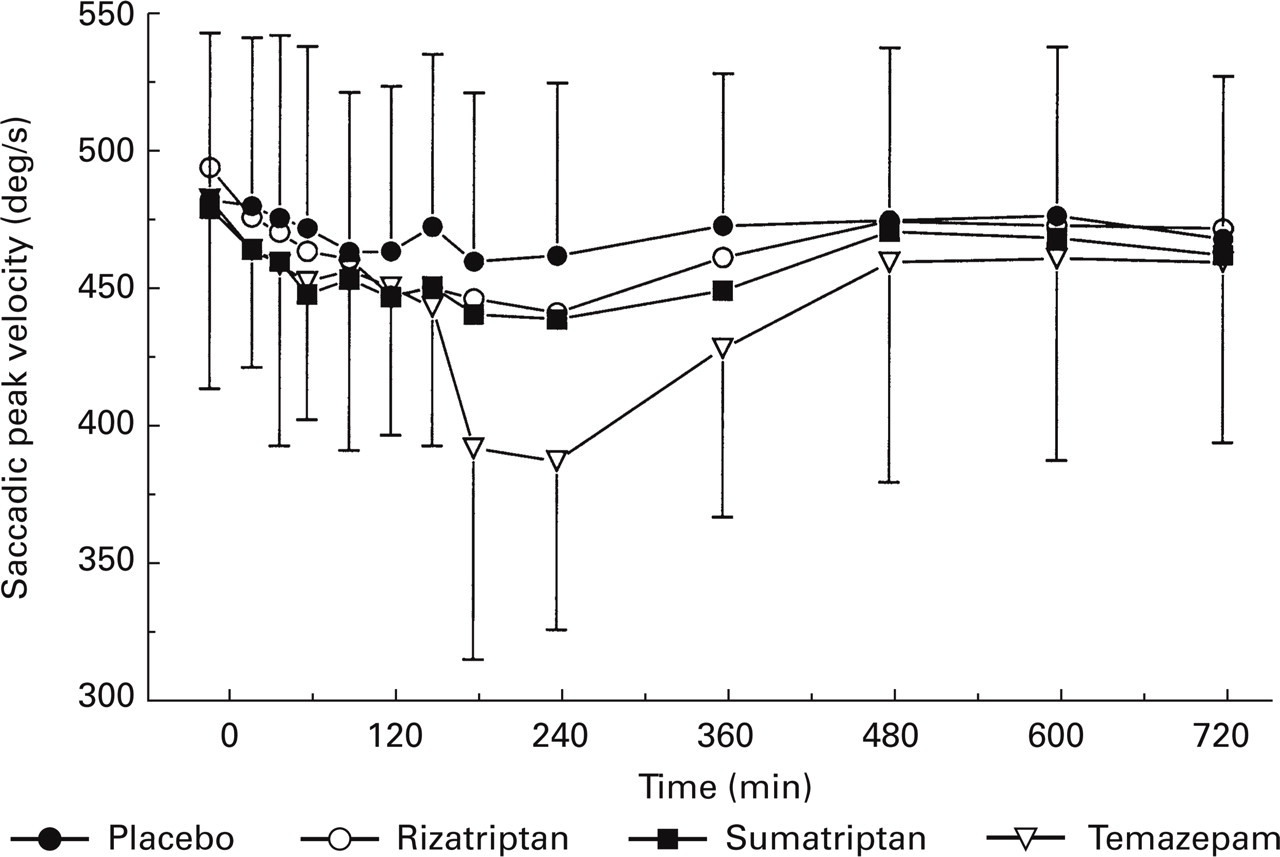
Average time-effect curve of the used drugs and saccadic eye movements peak velocity.
Electroencephalograms
Sumatriptan caused a statistically significant increase in alpha power compared with placebo for the frontal leads (0.477 (0.020, 0.935) µV/min). Rizatriptan showed a non-significant increase in alpha power for the frontal leads (0.366 (− 0.081, 0.812) µV/min). The average time-effect profiles of temazepam suggested a decrease of frontal and occipital alpha power, but this did not reach statistical significance relative to placebo (− 0.399 (− 0.856, 0.58), −0.439 (− 1.171, 0.292) µV/min, frontal and occipital leads, respectively) (Fig. 2a). The mean difference and confidence intervals for frontal leads alpha power are displayed graphically in Fig. 2(b). Temazepam showed a non-significant elevation of frontal beta power (0.142 (− 0.025, 0.309) µV/min). No effect of temazepam or the triptans was seen on the occipital beta amplitude. There were no significant changes in delta power for any of the treatments compared with placebo. A non-significant decrease of frontal (− 0.319 (− 0.654, 0.017) µV/min) and occipital (− 0.271 (− 0.577, 0.035) µV/min) theta power was observed for temazepam compared with placebo.

(a) Average time-effect curve for EEG alpha power (FC and PO leads). (b) Mean difference from placebo and 95% confidence intervals of EEG alpha power.
Body sway
The blinded review of body sway data indicated that body sway needed to be analysed on a log-scale. Both triptans caused small statistically significant increases in body sway compared with placebo (Fig. 3). Rizatriptan caused an increase of 30 (16, 45)% and sumatriptan had a smaller effect of 14 (1, 27)%. By comparison, temazepam caused an increase in body sway of 31 (17, 47)%. There was a statistically significant difference between the two triptans of 15 (2, 29)% (P=0.022).

Average time-effect curve for log-transformed body sway.
Visual analogue scales
Temazepam gave a clear decrease of the VAS alertness score of 4.35 (0.03, 8.68) cm compared with placebo. The triptans did not have any effect on VAS for alertness. No differences were seen in mood and calmness comparing the different treatments with placebo and the triptans with each other.
Cognitive test battery
Simple auditory and visual reaction time
The reaction time tests were performed with the dominant and the non-dominant hand. Temazepam caused an increase in mean auditive and visual reaction times for the dominant (51.8 (31.4, 72.1) and 35.9 (15.5, 56.4) ms) and the non-dominant hand (47.8 (32.3, 63.3) and 69.7 (24.3, 115.1) ms, respectively) compared with placebo. Rizatriptan and sumatriptan both caused an increase of mean auditive and visual reaction times. This was not significant for the dominant hand, but rizatriptan caused a significant increase in the mean auditive reaction time for the non-dominant hand (20.9 (5.7, 36.1) ms) compared with placebo. A significantly longer reaction time for rizatriptan compared with sumatriptan was found for the mean auditive reaction times of the non-dominant hand (18.2 (3.0, 33.4) ms).
Binary choice task
The mean reaction times for the binary choice task were significantly affected following temazepam. There was an increase compared with placebo of 99.5 (46.2, 152.8) ms. Rizatriptan and sumatriptan caused much smaller, non-significant increases in the mean reaction times.
Computerized visual searching task
Temazepam caused a significant increase in the number of errors (0.85 (0.17, 1.53) items), the main parameter of the visual searching task, and prolonged the mean searching time for the correct responses (by 1.44 (0.37, 2.51) s). Sumatriptan and rizatriptan had no significant effects compared with placebo or each other.
Simultaneous and serial recognition of words and figures
For simultaneous recognition of words, a significant increase in reaction times of correct responses of 0.34 (0.08, 0.60) s during temazepam treatment was observed. Also, for this task a significant reduction in the number of correct responses was seen for temazepam compared with placebo (2.02 (0.55, 3.49) items). Compared with temazepam, the average number of correct items during sumatriptan and rizatriptan treatments decreased slightly but not significantly. However, for simultaneous recognition of words the reaction times for the correct responses decreased significantly for both sumatriptan (0.31 (0.04, 0.58) (s) and rizatriptan (0.26 (0.00, 0.52) (s) compared to placebo.
The reaction times for correct responses during temazepam treatment increased significantly compared with placebo for both simultaneous recognition of figures (0.67 (0.23, 1.11) (s) and the serial recognition of words (0.45 (0.23, 0.66) (s). No effect of temazepam was found on the number of correct responses of both tests. Both triptans showed no effect on the reaction times or numbers of correct responses. No significant effect of temazepam, rizatriptan or sumatriptan was observed on the serial recognition of figures.
Corsi block tapping task and card sorting test
No significant effect of temazepam, rizatriptan or sumatriptan was observed on the corsi block tapping task or card sorting test.
Pharmacokinetics
Pharmacokinetic parameters are presented in Table 2. A recent pharmacokinetic study reported Cmax after rizatriptan 10 mg orally as 28.6±13.5 ng/ml (23). Thus, Cmax was found to be approximately 14% lower after the first dose in the current study, with encapsulated rizatriptan. In the same study tmax was reported as 42±12 min (23), which is 56 min earlier than in the current study. Thus, encapsulation of rizatriptan caused a delay in absorption compared with the tablet formulation as presented in recent literature. The pharmacokinetic properties of sumatriptan and temazepam were consistent with those presented in the literature (24–26). Dynamic measurements were performed after the second dose and around Cmax for the second dose of all treatments.
Pharmacokinetic parameters

Pharmacokinetic–pharmacodynamic modelling
Pharmacokinetic–pharmacodynamic relationships were investigated for all repeated measurements that showed a significant difference between both triptans and placebo, i.e. saccadic peak velocity (SPV) and log body sway.
For saccadic peak velocity corrected for placebo, the average slopes ((°/s)/(ng/ml)) and their 95% confidence intervals (CI) were as follows: sumatriptan −0.168 (− 0.37, 0.04), rizatriptan −0.494 (− 0.79, −0.20) and temazepam −0.164 (− 0.21, −0.12). Thus, no significant concentration-effect relationship was shown for sumatriptan, in contrast to rizatriptan and temazepam.
For log body sway corrected for placebo, the average slopes ((log mm/2 min)/(ng/ml)) and their 95% confidence intervals (CI) were as follows: sumatriptan 0.76 (− 0.39, 1.91), rizatriptan 2.98 (1.40, 4.56) and temazepam 0.495 (0.34, 0.65). As for saccadic peak velocity, a significant concentration-effect relationship was shown only for rizatriptan and temazepam.
Discussion
Until recently, it has been assumed that triptans do not penetrate the central nervous system. However, the current study showed that sumatriptan and rizatriptan had effects on several central nervous system measurements. Both triptans reduced saccadic peak velocity, which is indicative of a mild sedative drug effect. The increase in body sway and the increases in simple auditive and visual reaction times during sumatriptan and rizatriptan treatment support this finding. The effects of these triptans were generally smaller and less consistent than those of temazepam. For instance, the decrease in saccadic peak velocity caused by sumatriptan and rizatriptan was only half that caused by a moderately sedative dose of temazepam. Sedation caused by a 5HT1B/D agonist is rarely reported in human studies and could be of clinical importance. Sedative drug effects of sumatriptan have been demonstrated during safety studies in cynomolgus monkeys after administration of very high doses of 20 and 40 mg/kg (27). In a recent meta-analysis of oral triptans, the incidence of CNS effects (consisting among others of agitation, confusion, dizziness and somnolence) was reported around 4% after sumatriptan 50 mg and around 9% for rizatriptan 10 mg (28).
The apparent difference in effect size between sumatriptan and rizatriptan was small. Rizatriptan appeared to have a greater effect on the measurements than sumatriptan. This difference between these triptans only became significant for body sway and some of the cognitive tests. Rizatriptan CNS responses in this study may have been have been underestimated as rizatriptan encapsulation appears to have caused a decrease in expected Cmax of around 14%, and a delay in tmax of about 56 min, compared with pharmacokinetic data available for rizatriptan (23).
Cognitive testing showed clear neurocognitive effects of temazepam. The drug primarily caused an increase in reaction times for the different tests, an effect that is well known for benzodiazepines and other sedative agents (29). There are indications that benzodiazepines interfere primarily with procural and/or storage of information (29). This is supported by our findings. Temazepam decreased the number of correct responses on the simultaneous recognition of words and increased the number of errors on the visual searching task. Triptan effects on cognition and EEG were partly different from those of temazepam, suggesting slightly enhanced impulsivity. First, sumatriptan (and rizatriptan non-significantly) both caused an elevation of frontal-central alpha power. Such an increase in alpha power is compatible with a state of relaxed wakefulness (30–33). As expected, temazepam caused a decrease in alpha power, which was non-significant in this case but has been described repeatedly (34–36). A similar observation, an increase with the triptans and decrease with temazepam, was made in occipital-central theta power. This may be explained by subharmonic alpha waves that present as theta waves, a phenomenon which is often seen in young people's EEGs. However, it must be noted that the relationship between states of alertness and frontal alpha power is complex.
Secondly, both sumatriptan and rizatriptan caused a small but significant decrease in reaction times for the simultaneous recognition of words, while the number of correct responses declined (though not significantly). These effects are opposite to temazepam, which caused a significant increase of reaction times, consistent with moderate sedation. These findings may be caused by a potentially class-specific effect of sumatriptan and rizatriptan on information or decision processes. A change in strategy under influence of these triptans may be responsible for the decrease in reaction times: instead of choosing for accuracy subjects may have favoured speed. The same changes in decision processes were not observed for other reaction time tasks (e.g. the visual searching task and the card sorting task). In contrast to the word recognition task, these tasks give feedback on the accuracy (but not the velocity) of the responses, which may have stimulated accuracy.
The enhanced speed and reduced accuracy suggests that sumatriptan and rizatriptan stimulate impulsivity, although the effects were very small. This may explain why they were seen in only one test of the cognitive test battery: simultaneous recognition of words is regarded as one of the most difficult memory tests in the battery, and could therefore be more sensitive to a slight impairment. On the other hand, this isolated finding may be spurious.
Previous reports of other 5HT1 agonists, like buspirone and umespirone, have shown mixtures of sedative and stimulant effects (37–41). Buspirone showed a subjective sedative effect on analogue rating scales, impaired memory and increased reaction times for cognitive tests. Umespirone also increased reaction times. Stimulant effects such as an increase in subjective alertness and a decrease in reaction times were observed for buspirone (39) and improvements in word recognition and mood were observed for umespirone (37). Our observations on the 5HT1B/D agonist-triptans are partly compatible with these findings.
Recently, there is increasing evidence that triptan penetration into the central nervous system is not only responsible for some of the adverse effects, but also contributes to the relief of migraine symptoms. The mechanisms of action could be the inhibition of activity of the trigeminal nucleus caudalis activity (42) or the decrease of cortical spreading depression as occurs in the early onset of a migraine attack (43). Sumatriptan was also shown to normalize EEG changes associated with migraine, such as an increase in the theta and delta range and a decrease in the alpha and beta range (44).
In summary, these results show that sumatriptan and rizatriptan cause small but clear effects on the central nervous system. These effects may differ from those caused by the benzodiazepine temazepam. They may be due to subtle changes in information and decision processing, compatible with enhanced impulsivity. The effects of rizatriptan appear to be slightly larger than for sumatriptan, although in most cases this did not reach statistical significance. The difference may have been decreased by an alteration of rizatriptan pharmacokinetics as a result of encapsulation. In any case, the CNS-effects of both sumatriptan and rizatriptan were small in comparison to those of 20 mg temazepam, and do not seem to compromise clinical tolerability to any appreciable extent. Although this study was performed in a small number of subjects with many parameters and does not univocally establish CNS effects of sumatriptan and rizatriptan, these results could be regarded as a guide for further studies.
