Abstract
This study was a multinational, multicentre, double-blind, active controlled phase III trial designed to investigate efficacy and safety of 300 mg acetylsalicyclic acid (ASA) (n = 135) vs. 200 mg metoprolol (n = 135) in the prophylaxis of migraine. In total 270 (51 male and 219 female) patients, aged 18–65 years, suffering between two and six migraine attacks per month were recruited. The main objective was to show equivalence with respect to efficacy, defined as a 50% reduction in the rate of migraine attacks. A run-in phase was carried out with placebo for 4 weeks, followed by a 16-week drug phase. In both treatment groups the median frequency of migraine attacks improved during the study period, from three to two in the ASA group and from three to one in the metoprolol group; 45.2% of all metoprolol patients were responders compared with 29.6% with ASA. Medication-related adverse events were less frequent in the ASA group (37) than in the metoprolol group (73). The findings from this trial show that metoprolol is superior to ASA for migraine prophylaxis but has more side-effects. Acetylsalicylic acid is better tolerated than metoprolol. Using a strict responder criterion ASA showed a responder rate comparable with the placebo rate in the literature.
Introduction
Migraine is characterized by recurrent attacks with individually different frequency and intensity, and a duration of between a few hours and days. If the number of attacks is rare it is recommended that patients use symptomatic treatment. If patients suffer from three or more severe attacks within 1 month, prophylactic treatment may be of benefit for the patient. Therefore a useful prophylactic therapy should reduce the frequency of attacks and eventually the duration or severity of migraine, combined with a good tolerance and safety profile.
Although the mode of action of most compounds recommended for migraine prophylaxis is not known and no animal model is established to study pharmacodynamic properties (1), a variety of drugs has proven to be effective. One study with acetylsalicylic acid (ASA) demonstrated a reduction of attacks in migraine patients using a daily dose of 1300 mg (2). The Physicians Health Study, a primary prevention trial with myocardial infarction or stroke as endpoints, found a decrease in migraine frequency in physicians taking ASA in comparison with placebo (3). One potential benefit of low-dose migraine prophylaxis with ASA (300 mg per day) compared with other compounds, especially betablockers, could be a good safety profile. This report describes the results of a clinical trial comparing the efficacy and safety of ASA and metoprolol in a randomized and double-blind design.
The betablocker metoprolol (200 mg per day) is widely used in Europe and recommended by the Germany Headache Society as the treatment of choice for migraine prophylaxis (4). Its efficacy has been shown in several controlled trials (5–16). Therefore metoprolol was selected to be the comparative drug. The mechanism of action of betablockers is still not understood. There is no correlation between the efficacy of these drugs and their entry into the central nervous system, membrane stabilizing properties, or ability to block 5HT receptors (17). However, those blockers that are effective in reducing migraine attacks increase peripheral vascular resistance. They are contraindicated in patients with asthma, congestive heart failure, cardiac dysrhythmia or a history of depression. Therefore, there is a need to investigate alternatives for migraine prophylaxis with drugs not affecting vascular systems.
Study design
In accordance with the diagnostic criteria of the International Headache Society (18), male or female migraine outpatients, aged 18–65 years and suffering from migraine for at least 1 year, were enrolled into the clinical trial. Prior to enrolement there was a 4-week preliminary phase (if patients were already keeping a diary with all items required, this diary could be considered as preliminary phase) where the patient recorded frequency, characteristics and intensity of headache, as well as all drugs taken for treatment of migraine attacks. During the preliminary period at least two but not more than six migraine attacks had to occur. Patients with non-migraine types of headaches on more than 6 days were excluded. After this period treatment was started with placebo in both treatment groups for 4 weeks. This was followed by a 16-week drug phase, with group 1 receiving 300 mg acetylsalicylic acid per day and group 2 receiving 100 mg metoprolol per day during the first week and 200 mg per day thereafter. During the treatment period the patient had to keep a headache diary in which the above-mentioned parameters had to be recorded. Follow-up visits were scheduled at weeks 4, 8, 12, 16 and 20.
The trial was carried out in accordance with the provisions of the written protocol, the European Guidelines for Good Clinical Practice, §§ 40 and 41 of the German drug law (AMG), and with the ethical principles that have their origins in the Declaration of Helsinki, revised version of Hong Kong. The protocol and supporting documents (information sheet, consent form, standard product information, protocol) were submitted to local ethics committees and approved. Before being enrolled into the study, all patients were informed about all aspects of the study, the risks and possible side-effects and about their right to withdraw at any time from the study without specification of reasons. Written informed consent was obtained before any trial-related procedures were conducted. The study was monitored by an independent company hired by the sponsor.
After collecting the CRFs a second in-house check for plausibility and completeness took place. All variables were entered twice by two different persons into the database. All inconsistencies were documented and clarified with the investigator. According to the protocol and agreement with the sponsor, measures of quality assurance were performed (pre-trial-, on-site- and report-audit).
Assessment of compliance was carried out by comparing the number of delivered medication packages with the returned containers and the medication unused. The investigator had to document the number of unused capsules in the CRF.
Patients, inclusion and exclusion criteria
Nineteen different centres out of 31 initiated centres took part in the randomization for the study. The centres were located in Austria, Belgium, Germany and Switzerland and recruited from one to 61 patients during the study period. Two hundred and seventy male or female outpatients, aged 18–65 years, suffering from migraine for at least 1 year and having between two and six migraine attacks per month were included in the study. The headache-free interval had to be at least 24 h between attacks. The migraine criteria according to the classification of the International Headache Society applied.
General exclusion criteria were: participation in a clinical study within the last 30 days or concurrent participation in another clinical study, inability to meet the requirements of the study protocol, women with childbearing potential not using adequate contraceptive precautions, pregnancy or lactation, psychiatric illness which in the opinion of the investigator rendered the patient unsuitable for entry into the study, or epilepsy. Study-specific exclusion criteria were: known or probable allergic reaction to one of the used study-medications or other antirheumatic agents, history of peptic ulcer or other types of gastrointestinal bleeding, history of drug abuse (including analgesics, alcohol > 315 g/week, benzodiazepins, sleeping drugs and stimulants), any current or past uncontrollable diseases of the kidney, liver, lung and heart; neurological or cerebral disorders, severe chronic diseases, myocardial infarction within the last 3 months, cerebral infarction within the last 3 months, stroke or TIA, chronic diseases influencing absorption, metabolism and excretion of the study drugs. Intake of prophylactic migraine treatment with metoprolol, propranolol, flunarizin, pizotifen, methysergide or dihydroergotamine within the previous 4 weeks was also an exclusion criteria.
Additional exclusion criteria were: prophylactic treatment with antiplatelet drugs, treatment with antidepressants within the previous 3 months, treatment with analgesics on more than 12 days per month, contraindications against acetylsalicylic acid (gastric or intestinal ulcers, bronchial asthma, allergy) or other antirheumatics, contraindications against metoprolol (heart failure: stage III or IV according to the NYHA classification, AV-block II and III°, bradycardia, late stage peripheral arterial disease, hypotonia with systolic blood pressure under 90 mmHg, asthma, diabetes mellitus, endogenous depression), platelet count < 100.000/mm3 (to be established at the time of inclusion), or non-migraine headache on a maximum of 6 days per month, if the patient was able to distinguish those pain sensations from the migraine attacks.
Study objectives and safety evaluations
The primary response criterion was the rate of patients showing a reduction in the number of their migraine attacks greater than 50% during the last 4 weeks of the treatment phase, compared with the 4-week run-in period. The frequency and character of migraine attacks during treatment was taken from the patient diaries. Secondary response criteria were:
Pain intensity and duration of migraine attacks.
Concomitant symptoms (presence, severity).
Side-effects in both treatment groups regarding type, intensity, frequency.
Number of days with migraine within 28 days (excluding days with interval headache).
Number of migraine attacks with severe headaches.
Duration of migraine attacks (hours).
Symptomatic treatment during migraine attacks: the number of doses per month were calculated. Single doses of sumatriptan, ergotamine tartrate or other ergotamine-containing medication were multiplied by a factor 3. Medication containing dihydroergotamine or codeine was multiplied by 2. All other analgesics, pain relief and migraine medications were multiplied by 1. Other triptans than sumatriptan were not available at the time of the trial.
Frequency of vomiting.
Number of drop-outs per treatment arm.
Adverse events were collected in a standard manner and classified as either adverse events or serious adverse events. Safety parameters and adverse events were documented in the CRF and in the original patient documentation. Safety parameters included blood pressure and heart rate in sitting position, body weight and respiration.
Statistics
The analysis of efficacy in this report according to protocol was based upon the comparison of the response rate as the primary objective variable in both treatment groups. The binary random variable response was defined as a reduction of the observed frequency of migraine attacks between the pre-study phase and the study endpoint greater than 50%. The last notation of response was consequently the basis of the confirmatory data analysis in this project. The study design can be characterized as a typically longitudinal investigation (repeated measurement factor: time) of parallel (independent) treatment groups.
In total seven visits (one admission visit and six further visits) each of 28 days ± 3 days were planned. Some objective variables (the number and duration of migraine attacks) observed in the study period Pi and reported at visit ti obviously depended upon the varying length Li of Pi. In order to avoid uncontrollable bias in the comparison of all these variables between the treatment groups the respective parameters were adjusted to the length Li of Pi. The observed frequency of migraine attacks Ai in the time interval Li was computed directly proportional to the time span L. Under this assumption the frequency of migraine attacks had to be corrected. The binary random variable response Ro was defined for each patient with the corrected values for the migraine attacks Acorr as follows:
if Acorr during last interval < 0.5 × Acorr during first interval then Ro = 1 (Yes).
if Acorr during last interval3 0.5 × Acorr during first interval then Ro = 0 (No).
The response Ro represents a typical pointwise pre–post comparison.
The sample size was determined as follows: based on a reduction of migraine attacks by 50% in 50% of patients prophylactically treated with metoprolol, an equivalence range of 30–70% for patients with a reduction of the attack rate of 50% and with alpha=0.05 (two-sided) and beta=0.2 the number of statistically evaluable patients required was n = 2 × 111 = 222. As a drop-out rate of 15% had to be expected, 270 patients had to be enrolled into the study.
Results
Patients
Three hundred and three patients gave informed consent to participate in the trial and were checked for entry criteria. Thirty-three patients were not eligible. The remaining 270 patients were randomized and received placebo on day 0. Forty out of 270 patients never received or took study medication (ASA or metoprolol) or had no data entry regarding attack frequency. The remaining 230 patients had at least one single intake of ASA or metoprolol and a minimum of one entry of attack frequency and were considered as intention-to-treat-collective (ITT) for the equivalence testing. Thirty-five additional protocol violators were identified throughout the study course. Therefore, 195 patients were treated according to protocol and were analysed as per protocol collective (PP). Seventy patients did not finish the complete study duration of 20 weeks and were withdrawn at different time-points. All patients with premature study termination were excluded from the PP-analysis as well as five additional patients, two patients not fulfilling entry criteria for migraine and three patients with incomplete recording of migraine attacks. No group difference regarding this issue could be observed.
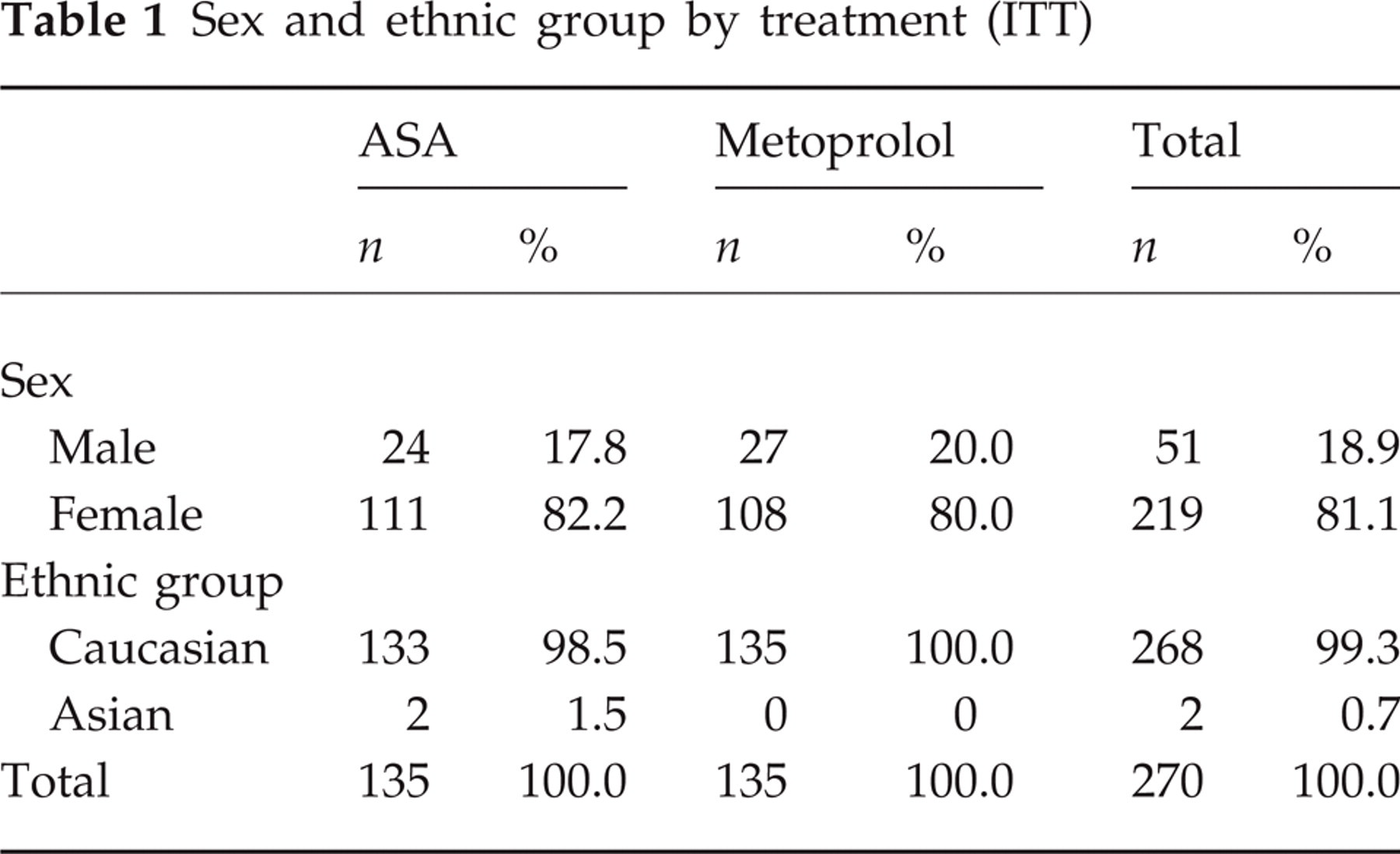
The demographic data and baseline characteristics are described for all patients receiving study medication (n = 270, 135 in each group) in Table 1. For equivalence testing the ITT (n = 230) and the PP-collective (n = 195) were used; 219 (111 ASA and 108 metoprolol) women and 51 (24 ASA and 27 metoprolol) men were included. The mean age of the female study patients was 38.9 years and for males it was 41.8 years. The two treatment groups did not show any relevant differences regarding weight and height. Female patients had a mean height of 166.5 cm and a mean body weight of 64.7 kg(166.5 cm and 64.8 kg in the ASA group and 166.6 cm and 64.5 kg in the metoprolol group). Male patients had a mean height of 178.5 cm and a mean body weight of 80.2 kg (179.1 cm and 79.6 kg in the ASA group and 178.0 cm and 80.8 kg in the metoprolol group). Possible confounding factors such as smokers (51) or women using hormonal contraceptives (89) were equally distributed. In both treatment groups most patients suffered from migraine without aura compared with migraine with aura (n = 50). The mean duration of the attacks was 20.5 h (20.9 ASA and 20.0 metoprolol). Patients suffered from migraine for 13.8 years on average.
Sex and ethnic group by treatment (ITT)
The frequency of concomitant symptoms such as nausea, vomiting, photo- and phonophobia, was comparable in both treatment groups. Regarding the description of pain there was a non-significant difference (about 10%) in moderate and severe pain between treatment groups. More patients of the metoprolol group had moderate pain, whereas the ASA group patients suffered more from unbearable pain (Table 2). Concerning all other mentioned concomitant symptoms both groups were comparable.
Description of pain and concomitant symptoms (ITT all)

Only 53 patients (ASA 25, metoprolol 28) had used medication for migraine prophylaxis before the start of the study, mostly with poor results regardless of the class of medication administered.
Efficacy
Equivalence of ASA treatment to metoprolol treatment could not be demonstrated in the intention-to-treat collective (ITT); 45.2% in the metoprolol group were responders according to the definition of the protocol, whereas 29.6% were responders in the ASA group (Fig. 1). These findings were confirmed in the per protocol analysis (Fig. 2). For patients who had not fulfilled the complete study duration the data were recorded as reported up to the last data entry. For both efficacy and safety parameters an ITT evaluation was performed (in terms of ‘last value carried forward’). The statistical examination of the data for centre and treatment by centre interactions showed no evidence of centre and treatment by centre interactions in the main objective. The computation of the 90% confidence interval of the response rate difference in both treatment groups within the study centres showed a complete covering of the estimated confidence interval by the planned equivalence range (− 20, + 20%).

Responder rates (improvement of headache frequency by ≥50% in the last 4 weeks compared with the 4-week pre-study phase. Data are shown for the intention-to-treat and per protocol analysis. □ ASA; ▪ metoprolol.
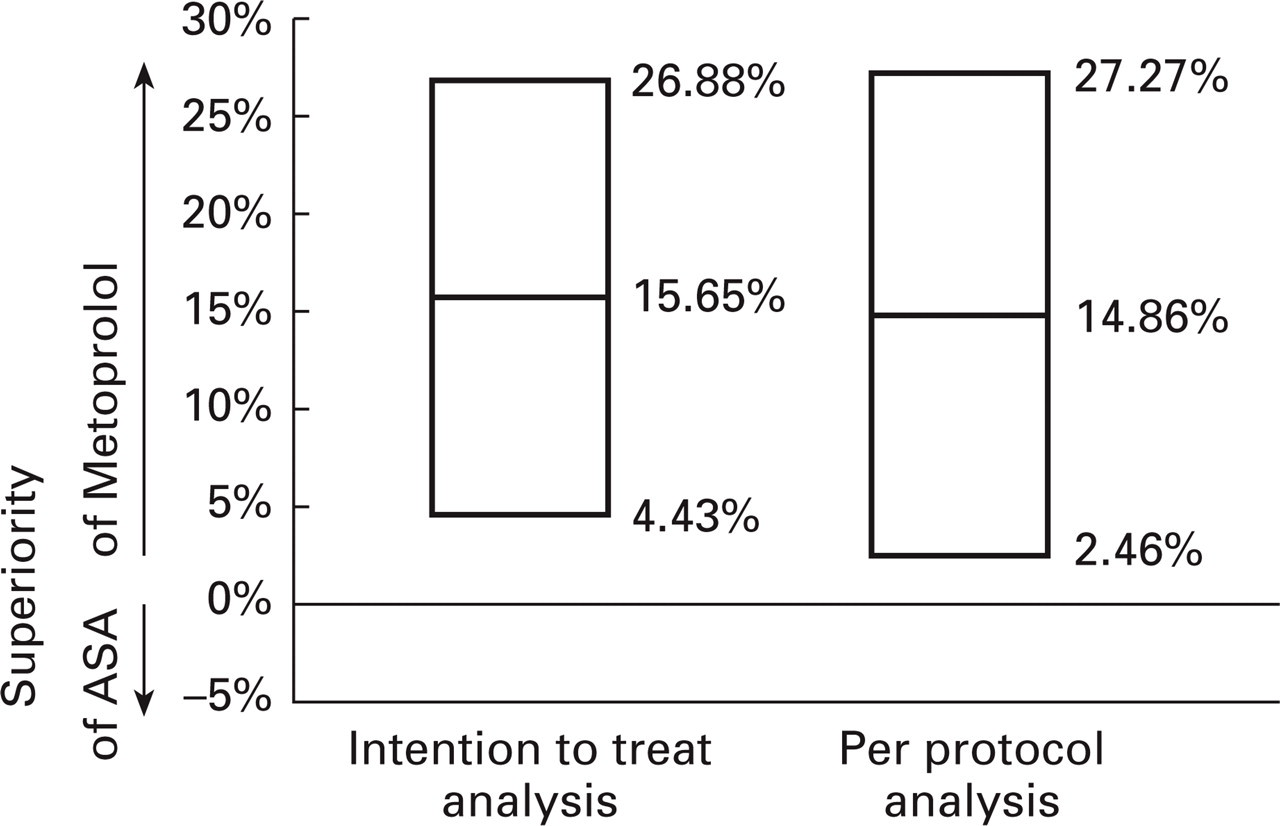
Difference of response rates showing the superiority of metoprolol. The mean difference is shown for the intention-to-treat and per-protocol analysis. 90% confidence intervals do not cross zero and therefore prove the superiority of metoprolol over ASA.
Both treatment groups showed improvement regarding the mean and median number of migraine attacks. The median number of attacks decreased from three to two in the ASA group, whereas in the metoprolol group a decrease from three to one was observed (Table 3). The mean number of migraine attacks decreased from 3.36 to 2.37 in the ASA group and from 3.55 to 1.82 in the metoprolol group. There was no effect of placebo treatment during the first 4 weeks on the frequency of attacks. The concomitant medication to treat the acute symptoms of migraine attacks showed no difference for both treatment groups in frequency and nature of the study drugs. Pain intensity of migraine attacks was equally distributed among both treatment groups at baseline. During the course of the study the number of patients rating their pain intensity as mild increased in the metoprolol group from 15% to 22%, whereas the number of patients in the ASA group slightly increased from 13% to 15%. A change in the duration of migraine attacks could be observed. The median attack duration in the ASA group was 8 h, with the lower quartile of 5 and the upper quartile of 16 on day 0. On week 20 the median of duration was 7 (4–13) h. The metoprolol group showed a decrease from 8 (4 to 15) to 5 (3, 4–10) h. Additional medication used for acute therapy was multiplied with factors from 1 to 3. The median use of medication did not change throughout the study in both treatment groups. There was a tendency to more moderate and severe vomiting in the ASA-group.
Frequency of migraine attacks by visit and treatment (ITT all)

Adverse events (AEs)
Table 4 gives an overview of the reported adverse events during the study period. Forty-two patients in the ASA group and 53 patients in the metoprolol group had at least one adverse event (difference not significant). More adverse events regardless of causality were recorded in the metoprolol group. Especially skin (9), psychiatric (16), vascular (extracardiac) (5), respiratory system disorders (6) and body as a whole general disorders (11) were observed in the metoprolol group. In the ASA group patients suffered mainly from gastrointestinal system disorders (30). Table 5 summarizes all adverse events according to body system. In cases of multiple recordings of the adverse event (due to the duration of the event) this adverse event was considered only once. All adverse events were assessed regarding causality by the investigator. A better tolerance could be observed in the ASA group. Cases considered by the investigators as probably, possibly or definitely related to study medication were more frequent in the metoprolol group (73) than in the ASA group (37). Gastrointestinal disorders were almost similar in frequency in both groups (ASA 22, metoprolol 16), whereas psychiatric disorders (ASA 0, metoprolol 12), autonomic nervous system (ASA 0, metoprolol 8) and general disorders (ASA 1, metoprolol 9) were more frequent in the metoprolol group. No unexpected and unlabelled adverse drug reactions were reported during this trial. The investigators classified serious adverse events in two cases. Hepatitis B was diagnosed in one patient in week 20. A coloscopy was performed because of darkened stools in one patient as inpatient procedure. The result of the coloscopy was without pathological findings but the patient discontinued the study.
WHO classification (symptoms) ( ITT all)

Number of patients with at least one adverse event (ITT All)

Clinical laboratory evaluation
To obey inclusion criteria it was necessary to exclude a thrombocytopenia before study entrance. A clinical laboratory evaluation during the study period was not foreseen in the protocol. No patient showed thrombocytopenia before admission to the study. There have been no obvious differences in vital signs and in physical findings between the treatment groups. There have been also no obvious changes in vital signs and in physical findings between the start of treatment and the end of the study in both groups. There was only a slight and expected change in median heart rate in the metoprolol group from 76 beats per minute to 72. The median for body weight remained unchanged with 68 kg in the metoprolol group throughout the study, whereas a slight gain of body weight was observed in the ASA group from baseline values of 66 kg to 67 kg at the end of the study.
Discussion
Epidemiological data regarding the incidence of migraine show that 12–14% of all adult women and 8% of all men suffer from migraine when classified by the IHS (International Headache Society) criteria (19–22). The attacks can occur at any time and can last for up to 3 days, combined with considerable pain. The duration and frequency of attacks impairs the quality of life, leading to frequent absence from work. The impaired social functioning is not only an economic factor, but influences private life as well (23–24). The use of symptomatic treatment for the acute migraine attack is driven by the patient him/herself, whereas prophylactic therapy of migraine most commonly is initiated by the physician. If migraine attacks are infrequent and eased rapidly by analgesics, non-steroidal anti-inflammatory drugs, ergotamine or triptans, there is no need for prophylactic therapy. Prophylactic therapy of migraine should be considered when the frequency of migraine headaches exceeds three per month, treatment of the acute attack has failed, the attacks are severe and the patient is psychologically unable to cope with the attacks (17). Many agents were investigated in the prophylactic treatment of migraine with sometimes conflicting results. The first priority is the use of an agent with the best benefit:risk ratio.
The aim of this study was to investigate the efficacy and tolerance of acetylsalicylic acid compared with metoprolol. Both treatment groups were comparable regarding demographic data and prognostic factors. The migraine type and migraine history, as well as pain and concomitant symptoms at baseline, showed that both treatment groups were well balanced. A 4-week placebo phase was planned before active treatment was started with 300 mg ASA and 200 mg metoprolol. The primary criterion for the confirmatory analysis of efficacy was to determine equivalence between the treatments regarding the reduction in number of migraine attacks. Both treatment groups showed a reduction of migraine attacks during the study period. This was true for the primary endpoint (number of responders) for the mean number of migraine attacks per month. The median frequency reduction was significantly higher with metoprolol compared with ASA. In a preliminary analysis of the study (4) we observed a higher responder rate in both treatment groups. However, the final analysis with the protocol-defined strict response rate did not confirm these rates, although the differences between the treatment groups remained the same. In the ITT-analysis 29.6% of all ASA patients showed a reduction in attacks of greater than 50% and could be considered as responders according to the definition of the protocol.
This confirms the results of a previous study in which one-third of all patients responded to treatment with ASA (3). Indirect comparisons with the placebo rate in other studies on migraine prophylaxis, however, indicate that the placebo responder rate is the in the range of 30% (25–27). This would indicate that ASA is not superior to placebo in migraine prophylaxis. In the metoprolol group 45.2% of all patients responded to treatment. The results of the ITT-analysis could be confirmed with the results from the per protocol analysis. Metoprolol was slightly superior regarding reduction of pain intensity of migraine attacks. This did not result in a decrease in numbers and potency of drugs taken for the treatment of migraine attacks. Otherwise, no differences were found between the two treatment groups regarding other concomitant symptoms such as nausea, phono- and photophobia. The 4-week placebo phase did not result in a reduction in the frequency of migraine attacks. However, the duration of a single attack, as well as the concomitant symptoms, was influenced by treatment with placebo.
A major shortcoming of the present trial is the absence of a placebo group. This trial was initiated at a time when it was difficult to convince ethics committees in Germany that placebo groups in migraine trials are of major importance. As mentioned above, in most trials with a comparable design, the number of responders in terms of a 50% reduction in migraine frequency with placebo is around 30%. Therefore only the inclusion of a placebo group would have answered the question of whether ASA is effective in migraine prophylaxis.
Most of the earlier studies were not powered to answer the question of whether aspirin is effective in migraine prophylaxis. As early as 1977, O'Neill and Mann (2), in a small open trial, observed that daily treatment with 1300 mg of ASA reduced the frequency of migraine attacks by 50% and more. In other small studies, low-dose ASA (160 mg/daily and 250 mg/daily) did not affect migraine frequency (28). Two multicentre trials, however, indicated efficacy of ASA in the prophylaxis of migraine. In 1988 ‘The British Doctors Trial’ showed that a daily dose of 500 mg ASA reduced the frequency of migraine by an average of 30% (29). In the second trial, 325 mg ASA (every other day) was given to 22 071 male American physicians between the ages of 40 and 84 years for a period of 60 months, 661 of whom were migraineurs. In this trial, attack frequency in persons suffering from headache was reduced by 20% (3). These trials, however, did not use the same strict criteria for treatment success as the present trial.
ASA might be one of the few drugs that can be used for the prophylactic treatment of migraine in children. In a small double-blind trial comparing ASA (2–5 mg/kg every evening) with the calcium entry blocker flunarizine (5–10 mg), Pothmann (30) treated 30 children aged between 7 and 17 years for a period of 3 months. The final results showed no difference between the two drugs in reduction of attack frequency or symptoms. In this trial, ASA reduced the frequency of migraine attacks in 73.3% of the cases by more than 50% (flunarizine 71.4%). Neither the intensity nor duration of the attacks were affected by ASA or flunarizine.
Only a few trials have compared ASA's prophylactic benefits with other drugs. In a small open trial Baldratti et al. (31) compared the efficacy of ASA (13.5 mg/kg) with propanolol (1.8 mg/kg). In this trial both drugs were equally effective and reduced frequency, duration and intensity of the attacks to the same extent. Other studies were not able to reproduce these results. In a double-blind cross-over trial ASA (500 mg daily) was statistically less effective when compared to 200 mg propanolol daily (5). Similar results were obtained in our much larger multicentre trial.
The incidence of adverse events was lower with ASA than with metoprolol. Fifty-one adverse events were documented in the ASA group compared with 99 events in the metoprolol group. This difference was even more pronounced when comparing the adverse drug reaction, i.e. all those events classified by the investigator as probably, possibly or definitely related to the treatment. Thirty-seven drug-related side-effects were recorded in the ASA group and 73 drug reactions were recorded in the metoprolol group. The frequency of gastrointestinal disorders was almost similar in both groups (ASA 22, metoprolol 16). ASA was given in an acid-resistant preparation. The events classified as psychiatric disorders (ASA 0, metoprolol 12) or autonomous nervous system (ASA 0, metoprolol 8) were present only in the metoprolol group as expected and known from other clinical trials (5–12). If one compares the reasons for premature termination of study throughout the course of the trial, both groups were comparable regarding lack of efficacy (ASA 2, metoprolol 3), withdrawal of consent, lack of medication compliance and lack of visit compliance. In contrast, early termination because of adverse events during the active treatment phase happened more often with metoprolol (13) than with ASA (5). One of the advantages for the use of ASA in migraine therapy would be the good tolerance and the safety profile which allows a safe and uncomplicated intake over a long period. Only a few contraindications (haemorrhagic diathesis, gastrointestinal ulcers) limit the population who benefit from the efficacy of ASA.
In conclusion, this trial showed that 200 mg metoprolol daily are superior to 300 mg acetylsalicyclic acid. Using a strict responder criterion, ASA showed a responder rate of about 30%, which is comparable with the placebo rate in other trials. We therefore can not exclude that ASA might not be beneficial in migraine prophylaxis. However, only the inclusion of a placebo group would have answered the question of whether ASA is effective for the prevention of migraine.
Footnotes
Acknowledgements
The study was supported by a grant from Bayer AG Leverkusen. Monitoring was performed by EPA Euro Pharma, Kronberg. The protocol was written by the principal investigator.
