Abstract
Time-to-event (or time-to-relief) analysis has been recommended for comparison of onset of headache relief among migraine medications (1). In randomized clinical trials (RCTS) this analysis has been used for comparison of rizatriptan with sumatriptan (2, 3), naratriptan (4) and zolmitriptan (5). The superiority of rizatriptan vs. the other triptans for ‘onset of effect’ in these RCTs has been ascribed to the shorter Tmax (time to maximum concentration of drug in plasma) for rizatriptan (1 h) than for sumatriptan (1.5 h), zolmitriptan (1.5 h), and naratriptan (2.5 h) (6, 7).
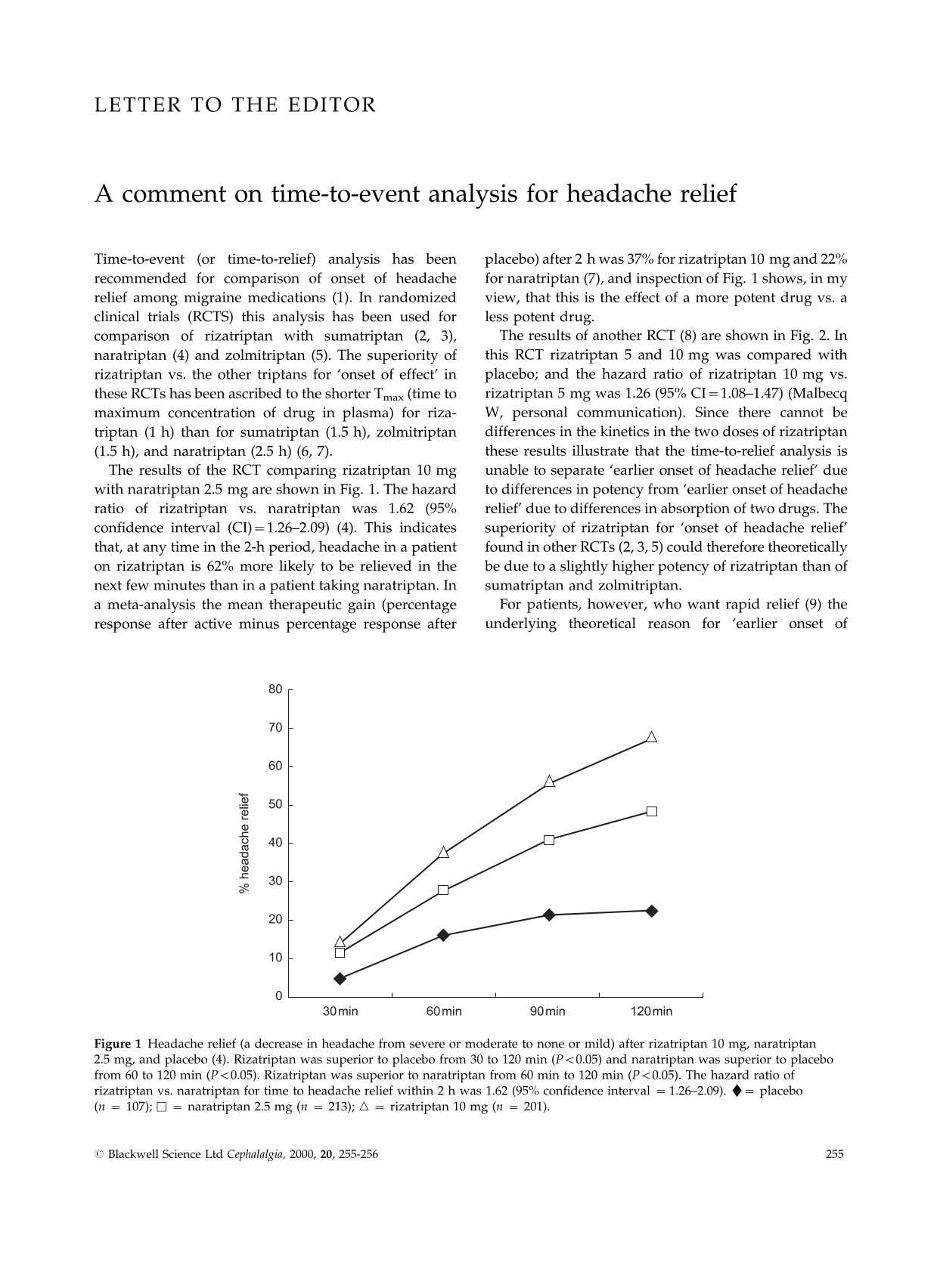
The results of the RCT comparing rizatriptan 10 mg with naratriptan 2.5 mg are shown in Fig. 1. The hazard ratio of rizatriptan vs. naratriptan was 1.62 (95% confidence interval (CI) = 1.26–2.09) (4). This indicates that, at any time in the 2-h period, headache in a patient on rizatriptan is 62% more likely to be relieved in the next few minutes than in a patient taking naratriptan. In a meta-analysis the mean therapeutic gain (percentage response after active minus percentage response after placebo) after 2 h was 37% for rizatriptan 10 mg and 22% for naratriptan (7), and inspection of Fig. 1 shows, in my view, that this is the effect of a more potent drug vs. a less potent drug.
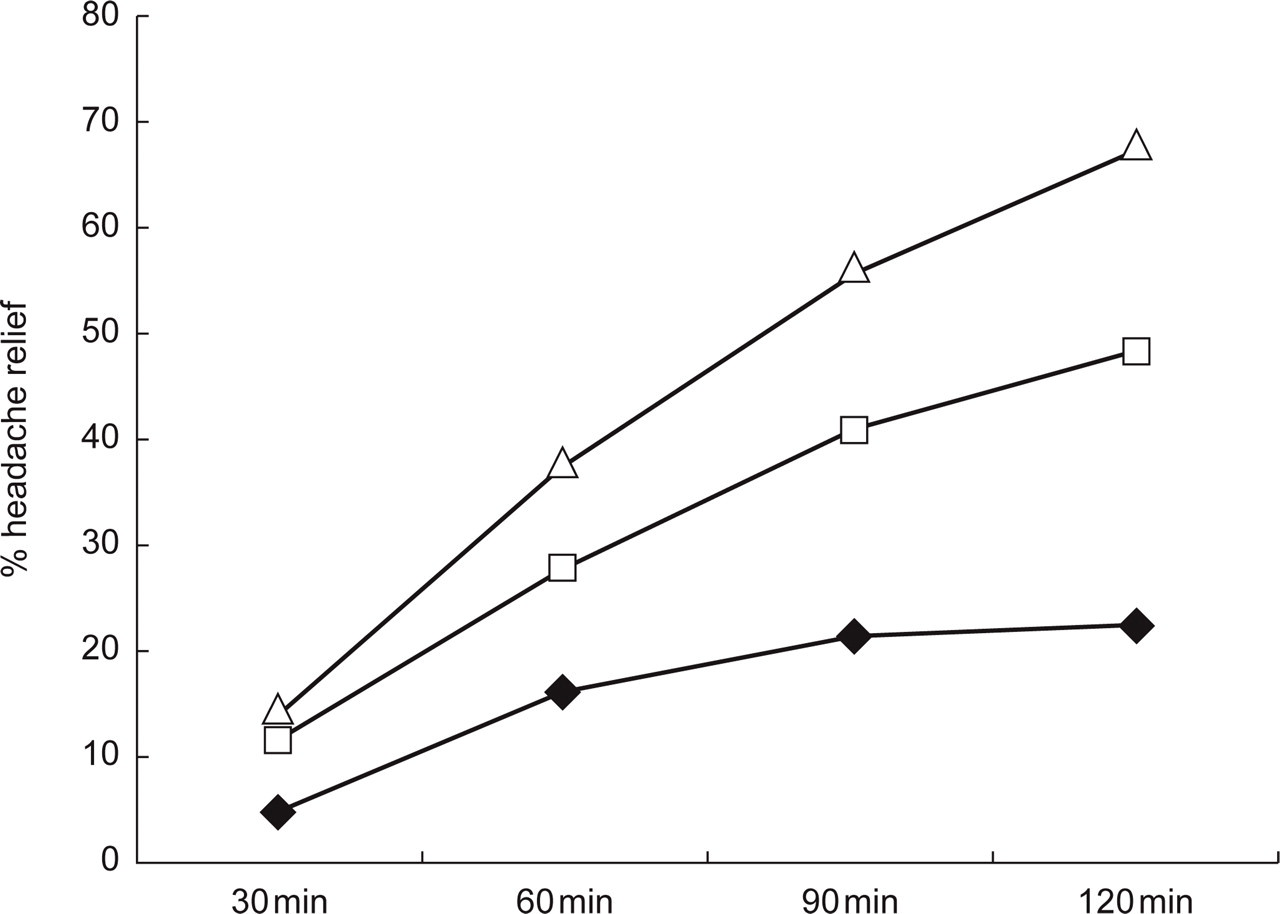
Headache relief (a decrease in headache from severe or moderate to none or mild) after rizatriptan 10 mg, naratriptan 2.5 mg, and placebo (4). Rizatriptan was superior to placebo from 30 to 120 min (P < 0.05) and naratriptan was superior to placebo from 60 to 120 min (P < 0.05). Rizatriptan was superior to naratriptan from 60 min to 120 min (P < 0.05). The hazard ratio of rizatriptan vs. naratriptan for time to headache relief within 2 h was 1.62 (95% confidence interval = 1.26–2.09). ♦= placebo (n= 107); □= naratriptan 2.5 mg (n= 213); ▵= rizatriptan 10 mg (n= 201).
The results of another RCT (8) are shown in Fig. 2. In this RCT rizatriptan 5 and 10 mg was compared with placebo; and the hazard ratio of rizatriptan 10 mg vs. rizatriptan 5 mg was 1.26 (95% CI = 1.08–1.47) (Malbecq W, personal communication). Since there cannot be differences in the kinetics in the two doses of rizatriptan these results illustrate that the time-to-relief analysis is unable to separate ‘earlier onset of headache relief’ due to differences in potency from ‘earlier onset of headache relief’ due to differences in absorption of two drugs. The superiority of rizatriptan for ‘onset of headache relief’ found in other RCTs (2, 3, 5) could therefore theoretically be due to a slightly higher potency of rizatriptan than of sumatriptan and zolmitriptan.
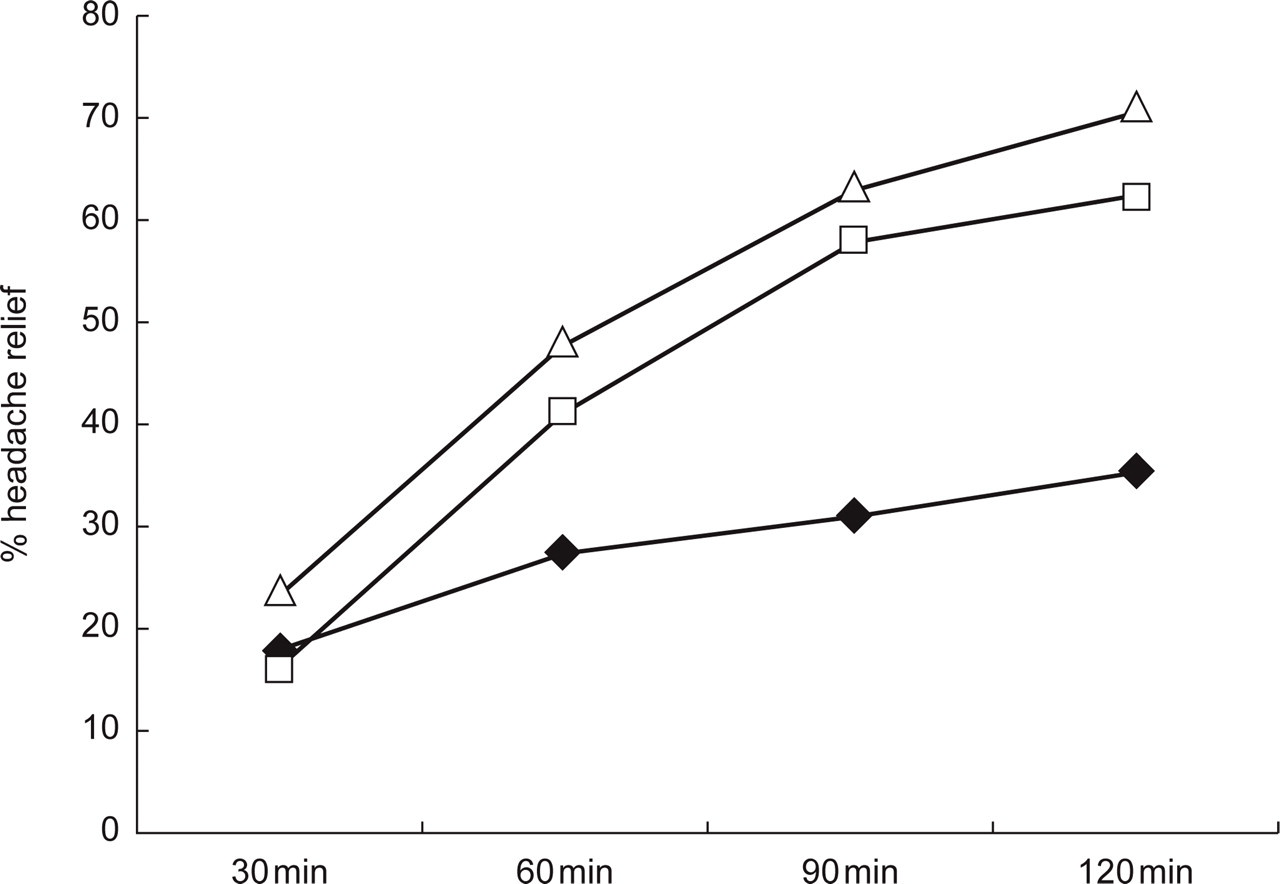
Headache relief after rizatriptan 5 and 10 mg, and placebo (8). Rizatriptan 10 mg and 5 mg were superior to placebo from 60 to 120 min (P < 0.05). Rizatriptan 10 mg was superior to rizatriptan 5 mg after 30 and 120 min (P < 0.05). The hazard ratio of rizatriptan 10 mg vs. rizatriptan 5 mg for time to headache relief within 2 h was 1.26 (95% confidence interval = 1.08–1.47). ♦= placebo (n= 304); □= rizatriptan 5mg (n= 458); ▵= rizatriptan 10 mg (n= 456).
For patients, however, who want rapid relief (9) the underlying theoretical reason for ‘earlier onset of headache relief’ is not important, and I find analysis of time-to-event clinically relevant.
