Abstract
Memory clinics were introduced in the US in the mid 1970s with the aim of identifying patients in the early stages of dementia and to act as a resource and database for research into memory impairments [1]. Since then they have spread around the world and the number and range of assessments offered at such clinics has increased in the context of demographic changes, improved assessment techniques and emerging treatments for dementia [2].
The first memory clinic in Victoria was set up in 1988 at the then Mount Royal Hospital (later called Northwest Hospital and now known as the Melbourne Extended Care and Rehabilitation Service) and data on the first 100 patients seen were published in 1992 [3]. In 1998 Cognitive Dementia and Memory Service (CADMS) clinics were established throughout Victoria and replaced the few independently operating clinics, adopting a standard model for the assessment of memory impairment [4].
This paper reports the characteristics and diagnoses of patients seen during 9 years’ functioning of this first Victorian memory clinic, in the period after the first 100 patients were seen and before the CADMS initiative came into effect. The utility of four of the instruments used in distinguishing patients with and without dementia has been assessed. Data will be presented for the initial assessment only. Some limited data on 299 of these patients were presented in an earlier publication [5].
Method
The clinic was staffed by a psychiatrist (DA), a geriatrician (LF/DL), a social worker, nurse, occupational therapist, speech pathologist, neuropsychologist and a secretary. It operated on a weekly half-day basis as previously described [3]. Referrals were accepted from General Practitioners (GPs), specialists and other health and community services. Most patients were seen on two occasions a few weeks apart during the initial assessment. Two new patients were seen each week by a doctor while an interview with their carer(s) was undertaken separately at the same time by a nurse, social worker or occupational therapist, and two patients who had seen the doctor a few weeks before underwent neuropsychological/speech pathology testing followed by a family conference/feedback session [3]. This paper reports on patients first seen between 19th December 1989 and 29th September 1998.
The main assessment tool was the Cambridge Mental Disorders in the Elderly Examination (CAMDEX) [6], which is a standardized instrument designed to enable the diagnosis of dementia to be made. It consists of several elements including a structured psychiatric interview with the subject and a standardized schedule for recording observations of the mental state and appearance. Also included are a structured interview with an informant and a scale for objective evaluation of a broad range of cognitive functions. Included within this cognitive subsection of the battery the Cambridge Cognitive Examination (the CAMCOG), are the Mini Mental State Examination (MMSE) [7] and Abbreviated Mental Test Score (AMTS) [8]. Addition of a small number of additional questions enabled calculation of the Organic Brain Score (OBS) of the Brief Assessment Scale [9]. Although widely used in research, it is unclear whether the CAMDEX is more efficient than shorter scales such as the MMSE at distinguishing demented from non-demented subjects [10]. One of the clinic doctors (DA, LF, DL) administered all elements of the CAMDEX except for the informant interview, which was administered by the nurse, social worker or occupational therapist.
The CAMDEX includes 19 direct questions to the patient about depression, 3 direct questions about sleep disturbance and 3 questions to the informant about loss of interest, guilt or depression seen in the patient. While most of these questions elicit a dichotomous yes/no answer, 6 have a possible score of 0–2 and the question about suicide can be scored from 0 to 3. By adding scores for the 25 items together a depression scale score (maximum 33) was generated.
The CAMDEX was supplemented by several informant questionnaires. The Informant Questionnaire on Cognitive Decline in the Elderly (IQCODE) [11] is a 26-item tool which asks an informant to compare the current cognitive state of the patient with that of 10 years previously. Individual items are graded: much better; a bit better; not much change; a bit worse and much worse. The response to each item is assigned a score from 1 to 5. The final total is derived from the average score for all questions; an average greater than 3.6 is regarded as indicative of the presence of dementia [11].
The Burden Interview Schedule [12] is a 29-item scale to assess the level of burden placed on the carer by the patient. Scales to measure activities of daily living (ADL) were also included as a means of assessment in terms of performance (PADLs) in domains such as bathing, dressing, toileting, transfer, continence and feeding. Instrumental tasks (IADLs) [13] assessed included: ability to use a telephone, to shop, to prepare food, to housekeep, to do the laundry, to travel, to self-administer medication and to handle finances. Ability in performance and instrumental indices were rated on scales from complete independence to complete dependence.
A physical examination was performed by a doctor together with standard physical investigations as appropriate. These were performed to assess general physical health and to detect any potentially reversible causes of memory impairment [14]. Haematological investigations included a full blood count; erythrocyte sedimentation rate; urea and electrolytes; liver and thyroid function tests; random blood glucose; vitamin B12; folate and syphilis serology. Other investigations included urine testing; computerized tomographic head scanning; chest radiograph and electrocardiogram. The results of these investigations were used in reaching the diagnosis, but do not form the main focus of this report. Results were not recorded on computer and the utility of these investigations may be the subject of a future report following further data coding and analysis.
At the second assessment visit patients underwent detailed cognitive testing by a neuropsychologist or speech pathologist prior to a family conference/feedback meeting. Reports from the neuropsychologist/ speech pathologist were used in diagnosis.
To document dementia severity the Clinical Dementia Rating scale (CDR) [15] was completed by the doctor and allied health professionals involved, at the end of the assessment process. This scale examines the domains of memory, orientation, judgement and problem solving, community affairs, home and hobbies, and personal care. Impairment in any of these areas is assigned a corresponding score: no impairment = 0; questionable impairment = 0.5; mild, moderate and severe impairment are assigned scores of 1, 2 and 3, respectively. Using complex rules, the scores on these six domains can be processed to indicate the presence of no (0), questionable (0.5), mild (1), moderate (2) or severe (3) dementia. By totalling the scores for all six domains it is also possible to achieve an overall ‘CDR Box score’ out of 18; the higher the total, the more severe is the degree of dementia. As CDR score sheets were not retained in the early years, box scores are available only for later patients.
The presence and extent of psychological distress in the informant carer was evaluated using the 30-item General Health Questionnaire (GHQ-30) scored in the simplest way recommended by its originators who note that the ‘best’ cut point for detection of a ‘case’ of neurotic disorder scoring the GHQ-30 this way is 4/5 [16]. The responses were categorized and totalled to produce an overall result. In a survey of 756 Australian adults, higher GHQ-30 scores were associated with progressively greater likelihood of being diagnosed as a ‘case’ of neurotic disorder. In that survey the mean scores for men (3.13) and women (3.18) were not significantly different [17].
As a result of the assessments and investigations described above, it was possible to derive diagnoses of mental and behavioural disorders (including organic disorders) according to the International Classification of Diseases: Diagnostic Criteria for Research, tenth version (ICD-10) [18]. These were made by a psychiatrist (DA) and geriatrician (LF) together using all available information including the results of neuropsychological or speech pathology assessment, but excluding the use of scale scores (CAMCOG, etc.) so as to permit subsequent assessment of the specificity and sensitivity of these scales. The National Institute of Neurological and Communicative Disorders and Stroke/Alzheimer's Disease and Related Disorders Association (NINCDS-ADRDA) [19] criteria for Alzheimer's Disease (AD) were applied to the data from each patient permitting each to be classified as having no, possible, probable or definite (autopsy confirmed) AD.
To determine whether the longer CAMCOG was more useful than the briefer MMSE and AMTS, or the informant rated IQCODE at differentiating those patients with a consensus diagnosis of dementia from those without, a series of receiver operator curves (ROCs) were constructed, plotting sensitivity against one minus specificity for each of these instruments. These plots were performed only for those patients with a consensus diagnosis of dementia versus those felt to be cognitively normal or having only a functional psychiatric syndrome.
In order to have sufficient subjects for statistical analysis it was necessary to combine the group with functional psychiatric disorders with the ‘normal’ group, as so few ‘normal’ patients were seen in the clinic. We believe it is reasonable to assess the performance of the instruments at discriminating demented patients from patients who were normal and had functional psychiatric illness as this is an important and common clinical issue. Those with organic cognitive impairments other than dementia were excluded from this analysis as many of them had significant impairment on cognitive instrument scores, but did not meet dementia criteria in relation to impairments of daily function not rated by those instruments.
Results
Patient demographics
Data were available on 577 patients. Of these, 226 (39.2%) were male and 351 (60.8%) were female. The age range was 43–96 years with a mean of 72.9 and median of 74 years (SD = 9.35). Three hundred and three patients (52.5%) were married and 200 (34.7%) were widowed. Single, divorced and separated individuals made up the remaining 70 (12.1%) (no data available for four patients).
Only 373 (64.6%) patients were born in Australia. Italian-born subjects made up the next largest subgroup: 68 (11.8%). Other Europeanborn subjects amounted to 98 (18.6%) of the cohort. English as a first language was spoken by 422 (73.1%) of patients. An interpreter was used for 48 (8.3%) assessments and 34 (5.9%) patients were interviewed in Italian by an Italian speaking geriatrician (DL).
Data collected over the life of the clinic did not permit identification of all referral sources, but the vast majority of referrals (over 80%) came from local GPs. Only eight patients (1.4%) were inpatients when seen.
Diagnoses
ICD-10 diagnoses are listed in Table 1. Exactly 64% of the patients were classified as having dementia using ICD-10 criteria. Only 28 (4.9%) were cognitively intact and psychiatrically well.
Main ICD-10 psychiatric diagnoses
Causes of cognitive impairment with some potential to improve
For some patients with the following diagnoses there was a theoretical potential for their cognitive impairment to improve if appropriate management were implemented: dementia due to abuse of alcohol (21); clinical AD with low B12 (10); AD with alcohol abuse (2); AD with meningioma (1); organic (non-alcoholic) amnesic disorder (24); mild cognitive disorder (MCI) associated with a systemic physical disorder (17); organic hallucinosis/delusional disorder/mood disorder (5); alcohol harmful use/dependence/amnesic disorder (14); late onset psychiatric disorder due to alcohol (2); harmful use of sedatives (1); schizophrenia and delusional disorders (4); mood disorders (52); neurotic, stress related and somatoform disorders (12); malingering (3). Thus, in theory, 168/577 (29%) of patients seen could have had a cognitive impairment with some potential to improve or recover. However, assessment of cognitive scores revealed that a number of these individuals did not have very severe cognitive impairment if any. For those for whom a MMSE score was available, 38/51 (75%) of those with mood disorders; 3/4 (75%) of those with organic hallucinosis/delusional disorders/mood disorders; 5/17 (29%) with dementia due to abuse of alcohol; 11/24 (46%) with organic amnesic syndrome; 12/17 (71%) with MCI associated with physical disorder; 5/11 (45%) with alcohol harmful use/dependence/amnesic disorder; 2/4 (50%) with schizophrenia and related disorder; 9/12 (75%) with neurotic, stress related and somatoform disorder; 3/6 (50%) others without AD had an MMSE of 24 or greater. The individuals with AD complicated by other disorders were generally quite impaired – only 1/12 had an MMSE of 24 or more and 7/12 (58%) had an MMSE below 17. Thus, 63% (99/158) of those who had the potential to improve and for whom an MMSE score was available scored 24/30 or more on the MMSE.
Cognitive screening tests and other instruments
The results from the cognitive screening tests, functional assessment tools, questionnaires and staging protocols used are shown in Table 2.
Cognitive screening, functional assessment, staging and questionnaire results
Table 3 shows how the scores on the different instruments correlated with one another. The directly applied cognitive instruments (CAMCOG, MMSE, AMTS and OBS) had the highest intercorrelations and also correlated well with the CDR and CDR Box scores. They had significant, but lower correlations with the IQCODE which correlated highest with carer burden score and significantly with carer GHQ-30 score, IADL and PADL.
Spearman correlations for instruments used in clinic
The mean depression score (ex 33) for 345 dementia patients in whom sufficient items of the scale had been completed to permit calculation of a score was 4.9 (SD = 3.9). There was no significant difference between the mean score of those with AD (mean = 4.5, SD = 3.5), vascular dementia (mean = 5.5, SD = 4.4) or other dementias (mean = 5.3, SD = 4.4) (one way ANOVA).
Predictive utility of direct cognitive assessment tools
The ROC curves for the CAMCOG, MMSE, AMTS and IQCODE are shown in figure 1. The area under the curve for each plot (higher score = better discrimination, maximum possible = 1.0) was: CAMCOG 0.92 (SE = 0.018; 95% CI = 0.88–0.96); MMSE 0.87 (SE = 0.024; 95% CI = 0.83–0.92); AMTS 0.85 (SE = 0.024; 95% CI = 0.80–0.90), IQCODE 0.82 (SE = 0.036; 95% CI = 0.75–0.89). A table showing the sensitivity, specificity, positive and negative predictive values and per cent correctly classified for each of these four instruments at a variety of cut points is available from the authors.
Receiver operator curve for detection of dementia versus no dementia (normal or functional psychiatric disorder) for four cognitive assessment instruments. +, Cambridge Cognitive Examination; ▪, Mini-Mental State Examination; ♦, Abbreviated Mental Test Score; , Informant Questionnaire on Cognitive Decline in the Elderly.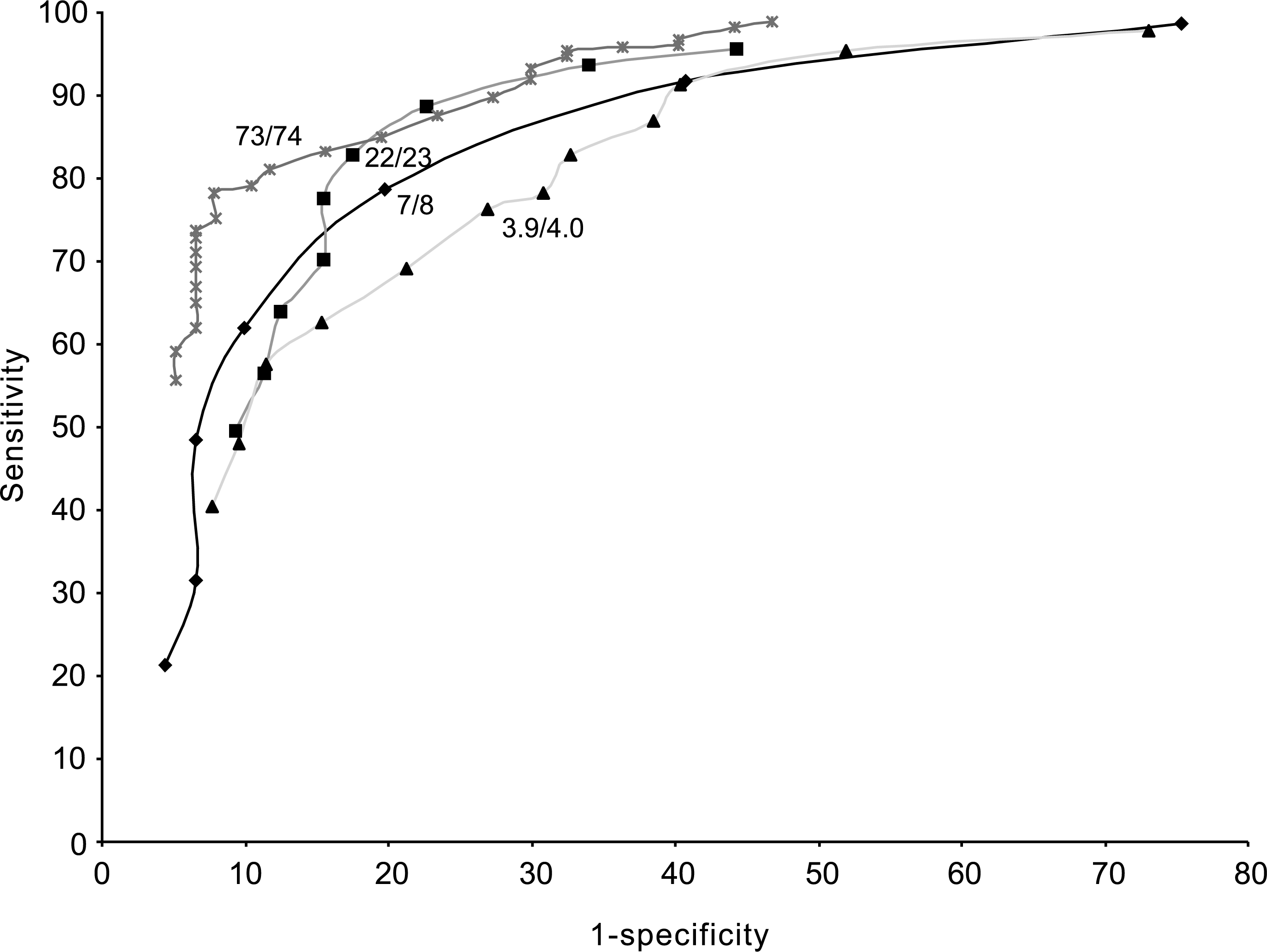
Discussion
No previous Australian study has reported on so large a cohort of memory clinic patients. Ours is one of the largest such series in the world literature [1, 20]. The operation of this memory clinic was like that of many others. The patients assessed were of similar age and sex to those in other reports [1, 20, 21]. Two-thirds of those seen had dementia, and AD was the commonest single primary diagnosis, though our figure of 40.4% is somewhat lower than quoted elsewhere [21, 22]. This may be due to our rigorous classification of all patients with any evidence of cerebrovascular disease, B12 deficiency or other possible causes of dementia as not having ICD-10 AD. There is a certain degree of ambiguity in ICD-10, which specifies that patients with AD (F00) should have ‘no evidence for any other possible cause of dementia’ and yet specifies a subcategory (F00.2) of ‘Dementia in Alzheimer's Disease, atypical or mixed type’ and states that ‘mixed Alzheimer's and vascular dementia is included here’. There was a low frequency of vascular dementias in our sample (7.5%). The ICD-10 requires the presence of neurological signs to confirm such a diagnosis and several patients with infarcts on brain CT nevertheless lacked frank focal neurological signs. We placed all patients with evidence of cerebral infarction from history, physical examination or special investigations, who did not meet ICD-10 vascular dementia criteria, into the ‘unspecified dementia’ category, together with patients who had a clinical presentation suggestive of AD, in the context of B12 levels slightly below the normal range or other confounding possible or multiple causes of dementia rendering the attribution of the dementia syndrome to a single cause impossible.
Few (4) patients were thought to have primary frontal dementias. We did not attribute any case of dementia to dementia with Lewy bodies (DLB) [23] as this diagnosis was not included in the 1992 edition of ICD-10 which we used, but some of the possible AD patients would likely have met criteria for DLB.
The 106 patients with other mental disorders due to brain damage, dysfunction, physical disease or substance abuse were a somewhat diverse group not satisfying criteria for a diagnosis of dementia, ranging from individuals with mild cognitive disorder with no apparent physical cause, to 36 subjects with amnesic symptoms due to alcohol, vascular disease or head injury.
Sixty-eight subjects had a functional psychiatric disorder, of which the largest number had a depressive disorder. Many of these were referred on to their local old age psychiatry services for management, as the clinic lacked the resources to treat them and follow them up. Some of these patients had been seen at the request of old age psychiatry services for assessment of cognition. Others were referred back to their GPs, who were given advice about further treatment. At this geriatric hospital clinic it was uncommon for cognitively intact individuals to present with pure anxiety disorders. The three individuals classed as malingering all had inconsistent atypical presentations in the context of compensable or superannuable work related injuries, accidents or stress.
Patients assigned to the ‘normal’ category were those in whom no significant psychiatric or medical abnormalities were found. Often these individuals had a family history of dementia or had anxious personality traits without meeting ICD-10 criteria for a psychiatric diagnosis.
Although 29% of the cohort had the theoretical potential for cognitive impairment to improve, many of these had either very mild impairment with limited potential to improve, or quite marked cognitive decline in the context of AD complicated by an additional cause of brain injury. We doubt that more than 10% of our cohort had the potential for their cognitive function to improve significantly with treatment.
It is notable that the vast majority of patients seen had cognitive or psychiatric disorders which were clearcut and of considerable clinical significance. The ‘worried well’ were conspicuous by their absence from the clinic.
The directly applied cognitive screening instruments had correlations between 0.81 and 0.93 with each other, but they correlated less well with an informant measure of cognitive decline (– 0.57 to −0.60). The informant measure of cognitive decline had a significant correlation with carer GHQ-30 and burden scores, as well as with CDR dementia severity ratings and activity impairment, emphasizing that informant reports measure something rather different from directly applied cognitive assessment tools and may be influenced by the emotions of the carer. However, the lower correlations with directly applied cognitive screening tools may also mean that informant scales provide additional clinically useful information in the diagnosis of dementia.
Symptoms of depression were not prominent in this cohort apart from the 9% of patients whose main diagnosis was a mood disorder, and there was no difference in frequency of depressive symptoms between the different classes of dementia. However, GHQ-30 scores in carers were high, the mean score being over twice that found in a large community survey in Australia [17], and higher than the most popular GHQ-30 cutpoint (4/5) for detection of a ‘case’ of neurotic disorder [16].
A comparison of 95% confidence intervals demonstrated no clear advantage for the 107-item CAMCOG over the 30-item MMSE, the 10-item AMTS or the 26-item IQCODE in discriminating ICD-10 dementia from normality or functional psychiatric disorder, though the additional information derived from the CAMCOG was found valuable by clinicians in their assessment of individual patients. For example, the ability to determine individual domains of impairment was often useful for directing further specialist neuropsychological assessment, as well as providing families with feedback on areas of strength and weakness in individual patients. The consensus among clinic staff was that while all instruments used provided some valuable data, the sheer length of the CAMDEX was an impediment to any expansion of the clinic because of the time taken to complete it (usually 90–100 min per patient).
Over a 9-year period the clinic sustained a high level of referrals (waiting times of several weeks for appointments were typical) until it was replaced by the state funded CADMS clinic. While the cost-effectiveness of such clinics is quite unclear, the service appeared popular with patients, families and GPs and the staff found the assessment tools useful, if time-consuming, to administer. As new treatments for AD burgeon, the memory clinic remains one method of coordinating assessments and initiating the delivery of appropriate treatment and supports for people with cognitive impairments and their families.
Footnotes
Acknowledgements
Thanks to patients, families and staff as well as Sue Graham, Elizabeth Ames and Marilyn Cain.
