Abstract
In recent years, the advent of the atypical antipsychotic medications has revolutionized the pharmacological treatment of psychotic illness. The efficacy of these newer medications, such as risperidone and olanzapine, is at least comparable to that of conventional medications such as chlorpromazine and haloperidol, while being associated with fewer adverse events [1, 2]. In particular, the atypical agents have a lower propensity to cause extrapyramidal side-effects (EPS) at therapeutically effective doses [3]. Further, clozapine, the first of the newer medications to be developed, has established superior efficacy for treatment-resistant patients [4, 5]. For these reasons, atypical antipsychotic medications have become the preferred first-line of treatment for patients with schizophrenia [6, 7].
While anecdotal reports suggest a dramatic shift towards prescribing of the atypical medications, little reliable information exists concerning the extent to which use of these medications has been adopted in psychiatric practice. Frangou and Lewis [8] recently reported that 26% of schizophrenic patients in a south-London psychiatric service were receiving atypical antipsychotics, while in the US approximately 50% of new prescriptions for antipsychotic medications are for atypicals [2]. A review of the most recent data available [9–13] indicated that atypical antipsychotic medications were routinely prescribed by clinicians in Australia, risperidone most commonly, followed by olanzapine and clozapine. However, the use of traditional medications, both oral and depot, remained widespread. Thioridazine was the most widely used oral conventional medication, while fluphenazine decanoate and flupenthixol decanoate were the most commonly used depot medications. It was also apparent that significant variation in prescribing practice occurred across regions, particularly with respect to the medications favoured within each category.
Survey research is expensive and time consuming as a means of elucidating drug utilization, with the risk that data may quickly become outdated where prescribing practices are undergoing rapid change. An alternative method employs prescription data to yield an index of drug utilization in terms of the number of defined daily doses per thousand population per day (DDDs/1000/day). This measure is the international unit of drug utilization approved by the World Health Organization (WHO) for drug use studies and has been adopted by the Commonwealth Department of Health and Aged Care Drug Utilization Sub-Committee [14, 15]. Expression of drug utilization in terms of DDDs/1000/day permits comparisons to be made independent of differences in price, preparation and quantity per prescription. For a chronically administered drug, the DDD/1000/day statistic indicates how many people per 1000 of the population have in theory received a standard dose (i.e. the defined daily dose) of a particular medication or category of medication daily [16]. The DDD/1000/day methodology has previously been used to elucidate trends in the utilization of various classes of drugs in Australia, including benzodiazepines, non-steroidal anti-inflammatories, antibiotics and, more recently, antidepressants [15, 17–19].
The aim of the present research was to employ the DDD/1000/day methodology to examine trends in the utilization of antipsychotic medications in Australia over the period July 1995 to December 2001. It was expected that the data would indicate a significant increase in the utilization of atypical medications–as a result of the introduction of risperidone (1993) and olanzapine (1997) onto the Australian market–and a concomitant decrease in the utilization of oral conventional and depot medications over the time period considered.
Method
For any given strength and dose form of a preparation, the number of DDDs/1000/day is given by the formula [16]:
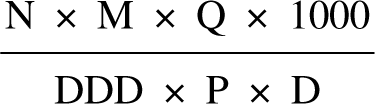
All prescriptions for antipsychotic medications forwarded to the Australian Health Insurance Commission (HIC) for subsidy under the Pharmaceutical Benefits Scheme (PBS) between July 1995 and December 2001 were included in the analysis. Currently, there are no PBS listed antipsychotic agents priced below the patient co-payment (i.e. not attracting a subsidy). Prescriptions for medications not available through the PBS, prescriptions dispensed through state and territory run public (and some private) hospitals and prescriptions for ‘doctor's bag’ items were not included [15]. Exclusion of these data was not expected to alter the substance of the results. The number of prescriptions dispensed for each strength and dose form of each medication, as well as total quantities dispensed, were obtained from the HIC. Data were provided in six-monthly intervals and were grouped by state and territory.
Data concerning actual quantities dispensed were available only for the period January 1996 to December 2000. Therefore, in calculating the DDD/1000/day statistic for the periods July 1995 to December 1995 and January 2001 to December 2001, actual quantity data from the nearest adjacent time periods (i.e. January 1996 to June 1996 and July 2000 to December 2000, respectively) were employed. In practice, there was little change over time in actual quantities dispensed for each dose and strength form of each medication. There was also little difference between actual quantities dispensed and maximum quantities as stipulated in the Schedule of Pharmaceutical Benefits [20], so that in the absence of information as to actual quantity (Q) prescribed, an estimate of utilization might still be obtained by substitution of maximum quantity in the formula given above.
Information concerning the major speciality of the prescribing doctor is also maintained within the HIC database. It was therefore possible to determine, for each dose and strength form of each preparation (excluding clozapine), and for each time period, the proportion of total prescriptions written by general practitioners (GPs) as opposed to specialist providers, assumed in the main to be psychiatrists. These data were similarly available for the period January 1996 to December 2000. Data concerning prescriptions for quetiapine, listed on the PBS in November 2000, were therefore not included in this analysis.
Data for public hospital use of clozapine, listed as a highly specialized (Section 100) drug in the Schedule of Pharmaceutical Benefits since September 1993, were obtained from the Highly Specialized Drugs Secretariat of the Commonwealth Department of Health and Aged Care. These data were available up to and including September 2001, so that calculation of the DDD/1000/day statistic for clozapine for the period July 2001 to December 2001 was in fact based on the period July to September, with appropriate adjustment of the time period (D) entered into the equation.
Defined daily doses for the appropriate dose form (i.e. oral or parenteral) of each medication were obtained from the WHO Collaborating Centre for Drug Statistics Methodology Guidelines for ATC classification and DDD assignment [21] and are given in Table 1. The mass of each strength and dose form of each medication was as given in the Schedule. Population data were obtained from Australian Bureau of Statistics estimates [22]. Data for the September 2001 quarter were not available at the time of writing. Therefore, data for the June 2001 quarter were used in calculating the DDD/1000/day statistic for the period July 2001 to December 2001.
World Health Organization defined daily doses (DDDs) for PBS-listed antipsychotic medications (WHO, 2000) [21]
It should be noted that the WHO DDD is a technical unit of use and the daily doses for each medication defined by the WHO do not necessarily reflect average doses prescribed in Australia [16]. Additional limitations of the method are that all drugs dispensed are not necessarily consumed and that the DDD/1000/day figure is calculated for the total population, while drug use may be concentrated in certain age groups or in a particular sex. Caution must be exercised where the DDD/1000/day statistic is used to estimate prevalence of use, since the validity of such estimates depends on the extent to which use of the agent in question has been continuous over the time period considered.
In order to compare utilization of oral conventional, depot and atypical medications–and of particular medications within each of these categories–total script numbers and quantities associated with each strength and dose form of each medication were summed across states and territories for each of the 13 time-periods considered. The number of DDDs per 1000 population per day was then calculated for each strength and dose form of each medication, at each time period, before being summed across strength and dose forms within medications, and in turn across medications within each category and across all medications. A similar procedure was followed in order to calculate the proportion of scripts for particular medications and categories of medication written by GPs as opposed to specialists.
Results
Table 2 shows change in the utilization (DDDs/1000/day) of each medication, of each category of medication and of all medications, between July 1995 and December 2001.
Utilization (DDDs/1000/day) of oral conventional, depot and atypical antipsychotic medications in Australia between July 1995 and December 2001
Overall use of antipsychotic medications
Dispensing of prescriptions for antipsychotic medications through community pharmacies in Australia increased from an estimated 3.41 DDDs/1000/day (424 420 prescriptions) during the latter half of 1995 to an estimated 5.69 DDDs/1000/day (698 750 prescriptions) during the latter half of 2001, an increase of 66.9%. Over the same period, the population of Australia increased from an estimated 18 122 900 persons (September 1995) to an estimated 19 383 424 persons (June 2001), an increase of 7.0%. The number of persons estimated to have received a standard dose daily of any antipsychotic medication increased from 61 771 to 110 385.
Use of oral conventional, depot and atypical medications
Dispensing of prescriptions for atypical medications increased from 0.27 DDDs/1000/day during the latter half of 1995 to 3.83 DDDs/1000/day during the latter half of 2001 (+ 1318.5%). The number of persons estimated to have received any atypical agent (daily) increased from 4962 to 74 236 during this time. Little change in the use of the atypical medications occurred prior to the latter half of 1997, after which the rate of increase accelerated markedly.
Over the same period, dispensing of prescriptions for oral conventional medications decreased from 1.76 DDDs/1000/day to 0.91 DDDs/1000/day (–48.3%), while those for depot medications decreased from 1.38 DDDs/1000/day to 0.96 DDDs/1000/day (–30.4%). Little change in the utilization of the oral conventional medications occurred prior to the first half of 1998, after which a gradual decline commenced. The number of persons estimated to have received standard doses of oral conventional and depot medications each day decreased from 31 816 to 17 611, and from 24 993 to 18 539, respectively, over the time period considered.
During the latter half of 1995, oral conventional medications accounted for 51.5%, depot medications 40.5%, and atypical medications 8.0%, of total prescriptions for antipsychotic medications dispensed through community pharmacies in Australia (percentage split based on DDDs/1000/day). During the latter half of 2001, atypical medications accounted for 67.3%, oral conventional medications 16.0%, and depot medications 16.7%, of total prescriptions.
Use of particular medications
Little change occurred in the use of pericyazine, which remained the least commonly used of the oral conventional medications, while utilization of each of the other oral conventional agents declined: thioridazine 0.55–0.11 (–80.0%); trifluoperazine 0.32–0.16 (–50.0%); and haloperidol 0.53–0.32 (–39.6%). Utilization of chlorpromazine declined between July 1995 and June 2000 (–26.9%), after which a slight increase in the use of this agent occurred.
Thioridazine and haloperidol were the most frequently prescribed oral conventional medications, at least until the first six months of 2001, during which time a rapid decline in the utilization of thioridazine occurred, from 0.39 to 0.17 DDDs/1000/day (–56.4%). While thioridazine and haloperidol accounted for 32.2% and 29.2%, respectively, of total prescriptions for oral conventional medications during the latter half of 2000, thioridazine accounted for only 12.4% of total prescriptions during the latter half of 2001 (haloperidol 35.3%; chlorpromazine 24.7%; trifluoperazine 17.6%; pericyazine 10.1%).
Dispensing of prescriptions for fluphenazine decanoate decreased markedly, from 1.08 DDDs/1000/day during the latter half of 1995 to 0.47 DDDs/1000/day during the latter half of 2001 (–56.5%). The number of persons estimated to have received a standard dose daily of this medication decreased from 19 578 to 9118. Utilization of zuclopenthixol decanoate increased from 0.01 DDDs/1000/day during the first six months of 1997 to 0.18 DDDs/1000/day during the latter half of 2001. Utilization of flupenthixol decanoate also increased over the time period considered (0.07–0.15), while utilization of haloperidol decanoate gradually decreased (0.22–0.15;–38.0%).
Notwithstanding the large decrease in its use, fluphenazine decanoate remained the most frequently prescribed depot medication during the time period considered. Prescriptions for this medication accounted for 49.2% of total prescriptions for depot medications during the latter half of 2001, while prescriptions for zuclopenthixol decanoate, haloperidol decanoate and flupenthixol decanoate accounted for 18.9%, 16.0% and 15.9%, of total prescriptions, respectively.
Utilization of olanzapine increased dramatically, to an estimated 2.49 DDDs/1000/day during the latter half of 2001, while utilization of risperidone and clozapine increased much less rapidly. Risperidone was prescribed more frequently than clozapine, the magnitude of this difference increasing over time. A slight increase in the use of quetiapine, available as a PBS medication since November 2000, occurred between January 2001 and December 2001.
During the first six months of 1997, risperidone accounted for 66.0% of total prescriptions for atypical medications, clozapine 34.0%. During the first six months of 1998, risperidone accounted for 35.1% of prescriptions for atypical medications, olanzapine 41.8% and clozapine 23.1%. During the second half of 2001, olanzapine accounted for 65.0% of prescriptions for atypical medications, risperidone 18.7%, clozapine 10.3%, and quetiapine 6.0%. An estimated 48 265 persons received a standard dose of olanzapine daily at this time.
Additional analysis
Data for the period July to December 2001 were re-analysed using a DDD for fluphenazine decanoate of 1.8 mg/day (25 mg/fortnight) and a DDD for olanzapine of 14 mg/day. Results of this analysis indicated that the proportion of total prescriptions for depot medications accounted for day by prescriptions for fluphenazine decanoate decreased from 49.2% to 35.0% (zuclopenthixol decanoate: 24.2%; haloperidol decanoate: 20.4%; flupenthixol decanoate: 20.4%), while the proportion of total prescriptions for atypical medications accounted for by prescriptions for olanzapine decreased from 65.0% to 57.0% (risperidone: 23.0%; clozapine: 12.6%; quetiapine: 7.4%). The proportion of total prescriptions for antipsychotic medications accounted for by prescriptions for depot medications decreased from 16.7% to 15.7%, while the proportion of total prescriptions for antipsychotic medications accounted for by prescriptions for atypical medications decreased from 67.3% to 65.3%.
Proportion of prescriptions written by GPs as opposed to specialist providers
In 1996, 82.1% of scripts for oral conventional and 83.5% of scripts for depot medications were written by GPs, while in 2000, 84.2% of scripts for oral conventional medications and 81.2% of scripts for depot medications were written by GPs. There was also little variation in the proportion of scripts for particular oral conventional and depot medications written by GPs as opposed to specialists, with the exception that the proportion of scripts for zuclopenthixol decanoate written by GPs remained somewhat lower (65%-75%) than for the other depot medications. The proportion of scripts for risperidone written by GPs increased from 45.6% in 1996 to 67.4% in 2000, while the proportion of scripts for olanzapine written by GPs increased from 32.1% in 1996 to 63.8% in 2000.
Discussion
The results of the present study indicate a remarkable change in antipsychotic prescribing practice in Australia over the past few years. Utilization of atypical antipsychotic medications–measured in DDDs/1000/day–increased more than 12-fold between July 1995 and December 2001, while the use of both oral conventional and depot medications declined. The number of individuals receiving a standard dose daily of any atypical medication increased from an estimated 4962 in the second half of 1995 to an estimated 74 236 during the second half of 2001, while the proportion of total prescriptions for antipsychotic medications accounted for by prescriptions for atypical medications increased from 8.0% to 67.3%. While some increase in the use of atypical medications was to be expected, the magnitude of the observed increase was surprising.
A proportion of the observed increase in the use of atypical medications–and decrease in the use of oral conventional and depot medications–can be attributed to patients switched from oral conventional or depot medications to atypical medications. In the study by Keks et al. [11], 88% of patients receiving an atypical medication had previously been on a conventional medication. However, the rate at which use of atypical medications increased far exceeded the rate at which use of oral conventional and depot medications decreased, so that a greater proportion of the increased use of atypicals must be accounted for by an increase in the number of patients receiving an atypical as their first antipsychotic medication. The fact that utilization of oral conventional medications decreased more markedly than that of depot medications suggests that switching, where this has occurred, has been primarily from oral conventional to atypical medications. Presumably, clinicians are less inclined to risk non-compliance by switching a patient who is stable on depot medication [23].
It is also possible that the present results reflect, in part, an increase in the range of conditions for which the atypical medications are employed. Currently in Australia, the use of aytpical antipsychotic medications within the PBS is indicated only for the treatment of schizophrenia. However, evidence is accumulating [24, 25] for the efficacy of the atypical medications in the treatment of other conditions–including bipolar disorder and major depression, and in the treatment of elderly patients presenting with late-onset psychosis or dementia–and it is clear that clinicians in Australia are currently prescribing atypical medications for these ‘off-label’ indications [11, 26]. An increase in the range of conditions for which atypical medications are employed would explain why overall use of antipsychotic medications increased, though other possible explanations for this finding, such as an increase in the proportion of cases reaching services or in the proportion of cases detected, cannot be excluded. A change in the population prevalence of schizophrenia is unlikely [27].
The results of the present study suggest that the use of depot medications in Australia is not as high as has been suggested [15]. Nevertheless, an estimated 18 539 persons across Australia were still receiving depot medications during the latter half of 2001. Until atypical medications are available in depot form, there is likely to remain a proportion of patients for whom depot medication is considered the only viable treatment [28]. With respect to the use of particular depot medications, fluphenazine decanoate was prescribed far more often than any other depot medication over the time period considered, notwithstanding a marked decline in its use. The increased use of flupenthixol decanoate and of zuclopenthixol decanoate may reflect a perception that these newer depot medications offer more favourable side-effect profiles than those of fluphenazine and haloperidol, though evidence for such an advantage from controlled studies has begun to emerge only recently [29]. The rapid decline in the use of oral conventional agent thioridazine, which occurred during the first half of 2001, is likely to reflect the response of clinicians to advice concerning adverse effects associated with the use of this medication [30].
A conspicuous finding of the present study concerns the rapid uptake of the atypical antipsychotic olanzapine in the Australian market since approval for the marketing of this agent was granted in March 1997. During the latter half of 2001, prescriptions for olanzapine accounted for 65.0% of total prescriptions for atypical medications dispensed through community pharmacies in Australia. By contrast, available evidence indicates that risperidone and olanzapine are prescribed equally often in the UK, while in the US use of risperidone is favoured [2, 8].
Few direct comparisons of the efficacy of different atypical medications have been conducted, and those that have compared risperidone and olanzapine using a randomized double-blind procedure have produced conflicting findings [31, 32]. However, it is clear that both medications are efficacious in the treatment of patients with schizophrenia and related psychotic disorders and generally well tolerated [1, 33]. The incidence of EPS may be higher with risperidone [31], but only when used at higher than optimal doses (i.e. < 6 mg) [32]. The only findings on which these studies agree are that risperidone is associated with greater prolactin elevation and olanzapine with greater weight gain [34]. However, evidence that the use of olanzapine is associated with significant weight gain is available from a number of other studies [35, 36]. There is also some evidence for an association between use of olanzapine and treatmentemergent Type-II (non-insulin dependent) diabetes mellitus, not necessarily related to weight gain [37]. Finally, evidence is emerging that the use of risperidone is more cost-effective than that of olanzapine, at least in overseas practice [1, 38]. In sum, the overwhelming preference for olanzapine among clinicians prescribing antipsychotic medication in Australia is surprising, and not justified on grounds of efficacy, side-effect profiles or cost.
As has been noted, a limitation of the present method is that the WHO DDDs do not necessarily reflect average doses prescribed in Australia. Comparison of the DDD values for each medication (given in Table 1) with recommended doses, as given in the Psychotropic drug guidelines [39], indicated that these values are generally in accord with doses recommended for Australian practice, though dose ranges for some medications are large. However, the extent to which either the DDD or the recommended dose for a particular medication is approximated in clinical practice is a separate question. Because the DDDs for olanzapine (10 mg) and fluphenazine decanoate (1 mg) appeared somewhat lower than the evidence from survey research would suggest, data for the period July 2001 to December 2001 were re-analysed using mean daily doses more consistent with those reported in recent studies [9, 11, 29, 40]. The pattern of the results was essentially the same. Analysis of the proportion of prescriptions for particular medications written by GPs, as opposed to specialist providers, indicated that, during the years 1996–2000, the proportion of scripts for oral conventional and depots medications written by GPs was consistently high, in the order of 80–85%. The proportion of scripts for the atypical medications written by GPs was considerably lower, though this figure increased significantly at each time point considered. Presumably, most GPs would be reluctant to prescribe a drug about which little is known–particularly in the case of a new class of medication–unless recommended by a specialist in the first instance [41]. However, as experience in the use of the newer medications is gathered, GPs will increasingly take responsibility for initiating treatment in their patients. For this reason, and given that the overwhelming proportion of scripts for antipsychotic medications are written by GPs, programs designed to foster collaboration between GPs and specialists in the delivery of mental health services will be crucial in ensuring that patients receive the best possible treatment [42].
In conclusion, the findings of the present study confirm that atypical antipsychotic medications have replaced conventional medications as the first-line pharmacological treatment for psychotic illness in Australia. While this change in prescribing practice is gratifying, the overwhelming preference for the use of olanzapine among clinicians prescribing antipsychotic medication in Australia is surprising, and of some concern given the significant increases in body weight which can accompany use of this medication. The findings of the present study also indicate that the DDD/1000/day methodology offers a simple and cost effective means by which to monitor change in antipsychotic prescribing practice over time. Such findings could be used to indicate the need for programs to correct prescribing practices that deviate too markedly from perceived ‘best practice’ in a given region at a given point in time.
Footnotes
Acknowledgements
This research was conducted while the first author was in receipt of a Research Training Fellowship from the NSW Institute of Psychiatry. Thanks also to Peter McManus for kindly offering advice concerning application of the methodology employed in the present study whenever this was required.
