Abstract
Depression is a frequent and disabling disorder [1, 2] where antidepressants and psychotherapy are the mainstay of treatment [3]. However, there is an increasing trend by patients towards the use of ‘natural alternatives’ to currently prescribed antidepressants to treat depression [4, 5]. The reasons for this trend are incompletely understood [5–7], but the perception by the consumer that currently prescribed pharmacological agents are more harmful than complementary and the rise of alternative medicine (CAM) plays a part [6, 8].
Complementary treatments for depression include substances such as St Johns Wort, Kava-Kava, tyrosine, tryptophan and 5-hydroxytryptophan (5-HT) [9, 10]. St Johns Wort (hypericum) has been the subject of metaanalysis suggesting it has an antidepressant effect, but tryptophan and 5-HT have been less extensively studied. In view of the community interest in ‘natural’ treatments, we thought it worthwhile to examine the evidence regarding tryptophan and 5-HTP for depression.
Tryptophan and 5-HT have a plausible physiological basis for acting as antidepressants because they are precursors of serotonin synthesis. 5-HT is synthesized from the amino acid tryptophan. The body absorbs tryptophan, converts it to 5-HT and then into serotonin both centrally and peripherally [3]. Both tryptophan and 5-HT are transported across the blood–brain barrier [3]. A normal Western diet contains about 0.5 g of tryptophan daily, of which only 2–3% is used in central serotonin production [11, 12]. An increase in dietary tryptophan increases the amount transported across the blood–brain barrier. Increase in the other amino acids transported by the same carrier reduces the transport of tryptophan [13, 14]. Therefore, there are some pharmacological and pharmacokinetic reasons why depressed patients administered 5-HT or tryptophan might experience improvement.
However, clinical trials in which patients have been administered tryptophan or 5-HT have given conflicting results and reached differing conclusions [10, 12, 15–17]. They also have side-effects (commonly nausea and gastrointestinal distress) [14]. Additionally, tryptophan ingestion has been associated with Eosinophilia–Myalgia Syndrome, which affected nearly 1500 users in 1989 and led to over 30 deaths. The reason for the outbreak, whether it was directly due to tryptophan itself or due to an impurity within the tryptophan, is still not known [18–21].
Methods
Trials were searched for in electronic general (MEDLINE, Psych-LIT, and EMBASE) and specialized databases (Cochrane Controlled Clinical Trials Register, Cochrane Collaboration Depression, Anxiety and Neurosis Controlled Trial Register) from 1966 to 2000; by checking reference lists of relevant articles; by hand-searching relevant specialist journals; and by contacting relevant authors where appropriate. Publications in all languages were sought. Search terms used were ‘tryptophan’, 5-hydroxytryptophan ′, ‘5-HTP’, ‘5-HT’ and ‘depression’. Trials were included if they were randomized, included patients with unipolar depression or dysthymia, compared preparations of 5-HT or tryptophan with placebo, and included clinical outcomes assessed by scales assessing depressive symptoms.
We independently extracted the data onto collection forms. Inclusion criteria were applied to all potential studies independently and a coefficient of agreement (Kappa) was calculated for them. Disagreement was resolved by consensus. Trial quality was scored according to risk of bias.
Studies were scored according to the method of treatment assignment (whether correct, blinded, and method of randomization described or group similarity documented), control of selection bias after treatment assignment (intention to treat analysis and < 15% loss to follow-up), blinding (blinding of outcome assessor and patient/ care-giver), and outcome assessment (all patients had standardized assessment). Standardized assessment required that studies utilize a clinically recognized and tested depression rating scale. This was most commonly the Hamilton Depression Rating Scale. Preparation, dose and route of administration were also recorded to facilitate comparison between studies.
Comparison: 01 L-Tryptophan and 5-HTP versus placebo for the treatment of depression. Outcome: 01 Numbers of the responders.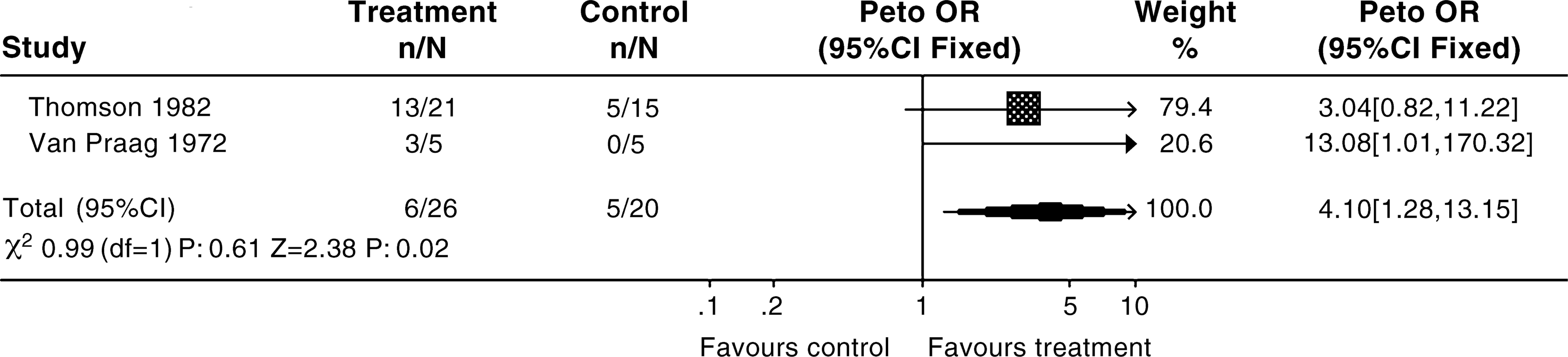
Subjects with bipolar affective disorder were excluded from the analysis because it is thought to be a different condition neurobiologically, and because of a theoretical potential of 5-HT and tryptophan to trigger upward mood swings in patients with bipolar affective disorder receiving serotonergic substances [22].
Studies of tryptophan and 5-HT were grouped for meta-analysis, which was undertaken by various techniques (Peto odds ratio, odds ratio (OR), relative risk and risk difference). When overall results were significant the relative risk reduction and number needed to treat were calculated. Additionally, the number needed to harm and confidence interval around these measures was calculated.
Results
One hundred and eight clinical trials were located. Eleven trials met the criteria for evaluation. Table 1 displays the exclusion process and reasons for exclusion of the other studies. The 11 remaining trials, including two non-English language trials, were evaluated:
– Three were subsequently excluded on the basis of methodologic weakness. – Six were excluded as they were crossover trials from which data could not be extracted for the first period.
Exclusion process
Only 2 remaining trials, (total combined number of patients was 64) met inclusion criteria [22, 23] Table 2. Details regarding the two included studies and nine excluded studies are presented in Tables 2 and 3, respectively.
Characteristics of included studies
Characteristics of 9 excluded studies
Because of the small number of patients included, which increases the risk of publication bias, conclusions about the efficacy of 5-HT and tryptophan are unreliable. The results suggested that 5-HT and tryptophan may be better than placebo at alleviating symptoms of depression (Peto OR = 4.10, 95% CI = 1.28–13.15). Four patients on active treatment reported side-effects (dizziness, nausea and diarrhoea). No deaths related to the use of 5-HT or tryptophan were reported.
Discussion
Evidence based medicine centres on a thorough evaluation of studies to ensure that reliable, trustworthy research is influencing clinical practice [24]. In this review a large body of evidence was subjected to very basic criteria for assessing reliability and validity, and was found to be largely of insufficient quality to inform clinical practice. There is a temptation to adopt a less stringent approach to assessing clinical evidence, however, this may result in incorrect conclusions being drawn about treatment efficacy.
The results of this meta-analysis suggest that 5-HT and tryptophan may have a positive effect in depression. However, well-designed studies are required before the true efficacy of these compounds is known.
The possible association between these substances and the potentially fatal Eosinophilia–Myalgia Syndrome remains an element of concern. Because currently used antidepressants are effective and usually well-tolerated, the clinical usefulness of 5-HT and tryptophan must remain limited until more information is available.
Footnotes
Acknowledgements
We thank A. Spinks for her assistance in data entry, and L. Chen and A. Bardossy for translation of non-English language articles.
