Abstract
Major depression is a common disorder which can be treated using several classes of antidepressant agents, including selective serotonin re-uptake inhibitors (SSRIs), monoamine oxidase inhibitors (MAOIs) and tricyclic antidepressants (TCAs). Although all currently available antidepressants appear to be equally effective, they can be distinguished by their side-effect profiles. About 30% of patients do not respond to the first antidepressant administered even when it has been given at the therapeutic dose and for an adequate period [1,2,3]. Additionally, up to 20% of patients are unable to tolerate the initial antidepressant medication. There are several treatment options for depressed patients who have failed to respond to their first-line medication: they can be switched to another antidepressant drug from the same class or to one from a different class [1]. Alternatively, they can be given an augmenting agent such as lithium or a combination of anti-depressant drugs [2]. The choice of initial and subsequent therapy is important because the prospect of recovery from depression recedes with increasing duration of the illness [2].
Selective serotonin re-uptake inhibitors are well established as safe and effective antidepressants. They are not only first-line drugs for the treatment of major depression, but may also play a major role in the treatment of non-responders to initial therapy. Sertraline is an SSRI that has been shown to be effective and well tolerated in the treatment of major depression [4,5,6,7,8]. The starting dose is 50 mg administered as a single daily dose. This is usually the optimal therapeutic dose for most patients, but the dose may be gradually increased in a step-wise fashion to a maximum of 200 mg/day, depending on response [9].
Moclobemide is a reversible and selective inhibitor of MAO type A [10]. It is an effective first-line anti-depressant and is comparable in terms of efficacy to non-selective MAOIs, TCAs and SSRIs. However, moclobemide is better tolerated than non-selective irreversible MAOIs [10]. The recommended therapeutic dose of moclobemide is 300–600 mg daily in two divided doses.
The aim of this open study was to evaluate the efficacy and tolerability of sertraline in patients with major depression who had failed to respond to an adequate trial of moclobemide.
Method
Patients
To be eligible to enter the study, patients (outpatients or inpatients) who were at least 18 years of age had to meet the DSM-IV criteria for a major depressive episode (single or recurrent) and have discontinued treatment with moclobemide within the last 6 weeks due to lack of efficacy after an adequate trial period. This was defined as no improvement after at least 4 weeks of treatment with moclobemide at a dose of at least 600 mg/day (or 300–450 mg/day in patients who could not tolerate the higher dose; n = 5). Patients also had to have a total score of at least 18 but less than or equal to 30 on the 17-item Hamilton Rating Scale for Depression (HAMD).
Patients were excluded if they were considered at serious risk of suicide (a score of 4 on question 3 of the 17-item HAMD or a score of 5–6 for question 10 on the Montgomery-Asberg Depression Rating Scale, MADRS). They were also excluded if they had a current history of seizure disorder, organic brain disease, anorexia nervosa, bulimia nervosa or purgative abuse or had been dependent on any drug or alcohol within the previous 6 months. Women of childbearing potential had to have a negative pregnancy test before entering the study and to use adequate contraception both during the study and for at least 1 month after study completion.
Patients entering the study were not permitted to have taken any other antidepressant after discontinuation of moclobemide. Patients with a history of non-response to sertraline or any other SSRI were excluded from the study, as were patients requiring concomitant psychotropic drugs, electroconvulsive therapy, intensive psychotherapy or drugs known to interact with sertraline. All concomitant medications taken during the study were recorded.
Study design
This was an 8- to 16-week multicentre, open study evaluating sertraline (50–200 mg) as a treatment for major depression in patients who had not responded to an adequate trial of moclobemide. Patients were recruited from various psychiatric services in Victoria and Queensland. The study was approved by local ethics committees and performed in accordance with Good Clinical Research Practice Guidelines. Before entering the study, each patient provided written informed consent.
Patients entering the study underwent a brief physical examination and provided a medical and psychiatric history. Any patients receiving antidepressant therapy had to discontinue that medication and undergo a wash-out period equivalent to five half-lives of the drug or its active metabolite. Sertraline was provided as 50 mg capsules and taken once daily at an initial dose of 50 mg/day. After 4 weeks of treatment, if there was no change in the HAMD score and the investigator considered the response was insufficient, the dose of sertraline was increased to 100 mg/day. Where necessary, the dose of sertraline was further titrated upwards in increments of 50 mg to a maximum of 200 mg/day, with 2 weeks at each dose level. By the end of the study, each patient had been maintained on a fixed dose of sertraline for a total of 8 weeks.
If intolerable side effects occurred or persisted at any time between weeks 4 and 8, the dose could be reduced by 50 mg/day. Additionally, if intolerable side effects occurred at the lowest dose (50 mg/day) or after the visit at week 8, the patient was withdrawn from the study.
Assessments
Patients were assessed at baseline and at the end of weeks 1, 2, 3, 4, 6, 8, 10 (optional), and 12, 14, or 16 (if applicable) of treatment. There was also a follow-up visit 30 days after the final dose of sertraline or at the time treatment was discontinued. For consistency, the same investigator assessed the patient at each visit and at the same time of day, wherever possible.
Efficacy was assessed using the following four measures: the 17-item HAMD [11], the Clinical Global Impression (CGI) scales of severity of illness and global improvement [12], the MADRS [13], and the Beck Depression Inventory (BDI) which is a 21-item questionnaire completed by the patient [14].
Tolerability was assessed by recording all adverse events observed by the investigators and reported spontaneously by patients.
Investigators also provided a personal evaluation of the overall efficacy and tolerability of sertraline for each patient.
Analysis
The primary outcome measures were the 17-item HAMD and the CGI scores. Response rate (and 95% confidence intervals [CI]) was calculated as the percentage of patients who had a successful response to treatment, which was defined as at least a 50% reduction in HAMD total score compared with the baseline score and a final HAMD score of 17 points or less. Treatment success was also defined as at least ‘minimally improved’ (CGI score of 1, 2 or 3) on the global improvement scale.
Response rates were calculated for both the intention-to-treat (ITT) population (all patients who received at least one dose of sertraline) and the evaluable population (patients who received at least four weeks' sertraline treatment). Both available data and last observation carried forward (LOCF) analyses were performed. A paired t-test was used to assess the statistical significance of the change from baseline in HAMD.
Results
Patients
A total of 63 patients from six study sites in Australia entered the study. As one patient did not receive sertraline, the intention to treat (ITT) population comprised 62 patients. There were 48 patients in the evaluable (per protocol) population. Thus, one patient was excluded from the ITT analysis and a further 14 patients from the per protocol analysis. Reasons for exclusion from the per protocol analysis included failure to meet entry criteria (n = 7); failure to complete at least 4 weeks of therapy (n = 4); and protocol violations (n = 3). The protocol violations consisted of two patients taking prohibited medications and a third patient increasing the dose of sertraline to 100 mg within 1 week of therapy.
The demographic and baseline characteristics of the 62 patients in the ITT population are summarised in Table 1. The mean duration since diagnosis was approximately 6 years. This was the first major depressive episode for 31 (50%) patients, and 52 (84%) patients had been free of major depressive episodes during the 12 months prior to the current episode. The mean duration of the current episode was 26 days (range = 35–2389 days). Most patients (92%) had no secondary psychiatric disorder, but 33 (53%) had a family history of psychiatric disorder, most commonly depression (40%).
Demographic and baseline characteristics of patients in the intention-to-treat population
Of the 62 patients in the ITT population, 39 (63%) reported having side effects with prior moclobemide therapy. The most common side effects were headache (29%), nausea (16%), drowsiness (15%) and insomnia (15%).
Forty-four (71%) patients were treated with sertraline 50 mg/day and only 18 (29%) received a higher dose of sertraline (17 patients were treated with 100 mg/day and one patient received 150 mg/day). Most patients (54, 87%) commenced sertraline therapy within two weeks of stopping moclobemide. Concomitant medication to provide night sedation was used by 17 (27%) patients. Temazepam was the medication most commonly used.
Efficacy of sertraline
Of the 62 patients in the ITT population, 48 (77%; 95% CI = 65–87%) were considered to be responders as defined by ≥ 50% reduction in HAMD total score and a final HAMD score of ≤ 17 points. Of the evaluable patient population, 45 (94%; 95% CI = 83–99%) responded to treatment. In addition, 54 patients in the ITT group (87%; 95% CI = 76–94%) were considered treatment responders as assessed by the CGI global improvement scale (‘very much improved’ = 39; ‘much improved’ = 12; ‘minimally improved’ = 3), while 100% of the evaluable patients achieved this endpoint (95% CI = 93–100%). Figures 1 and 2 show the changes in mean HAMD and CGI scores over time, respectively.
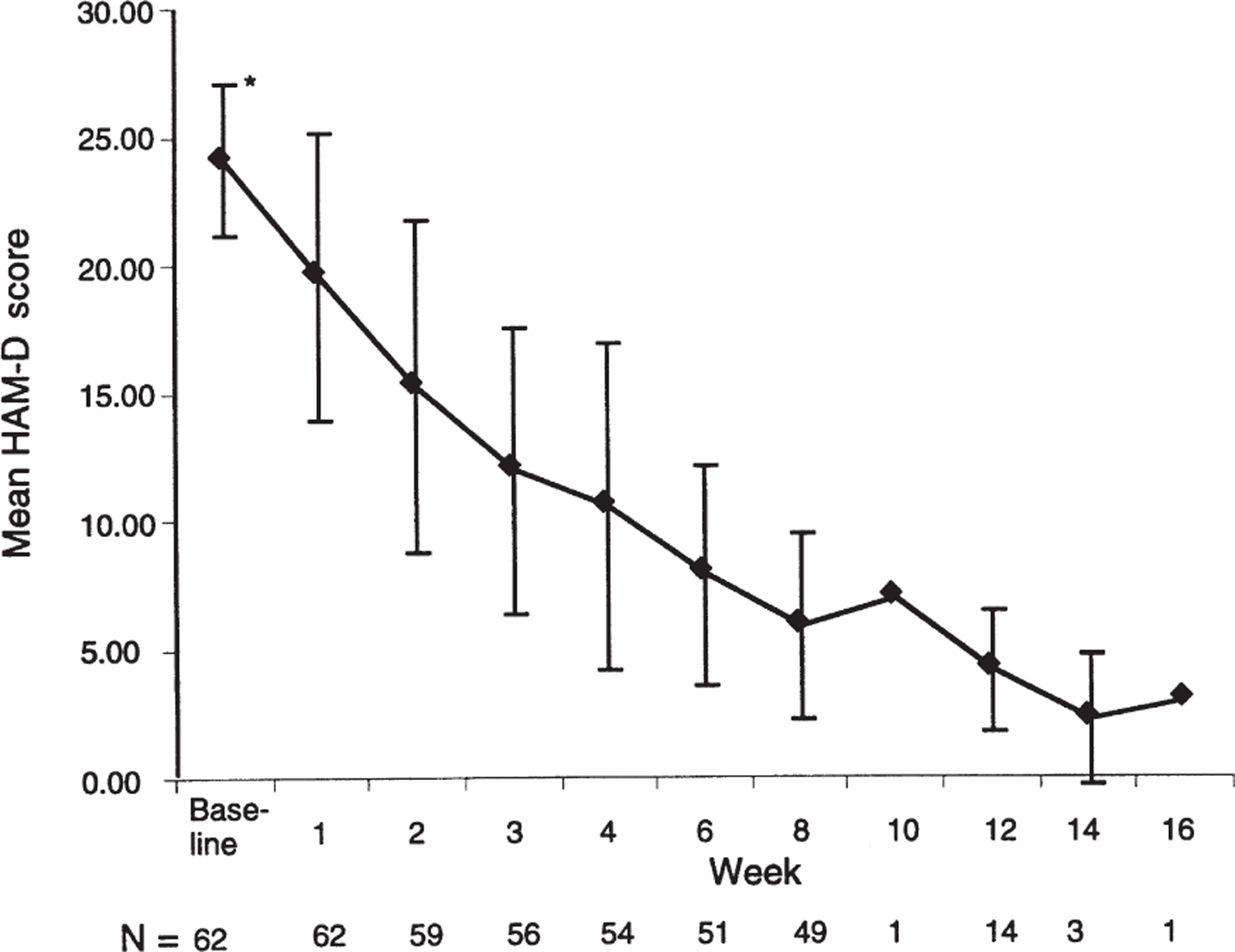
Mean Hamilton Rating Scale for Depression (HAMD) score at each timepoint. Error bars indicate ± ISD.
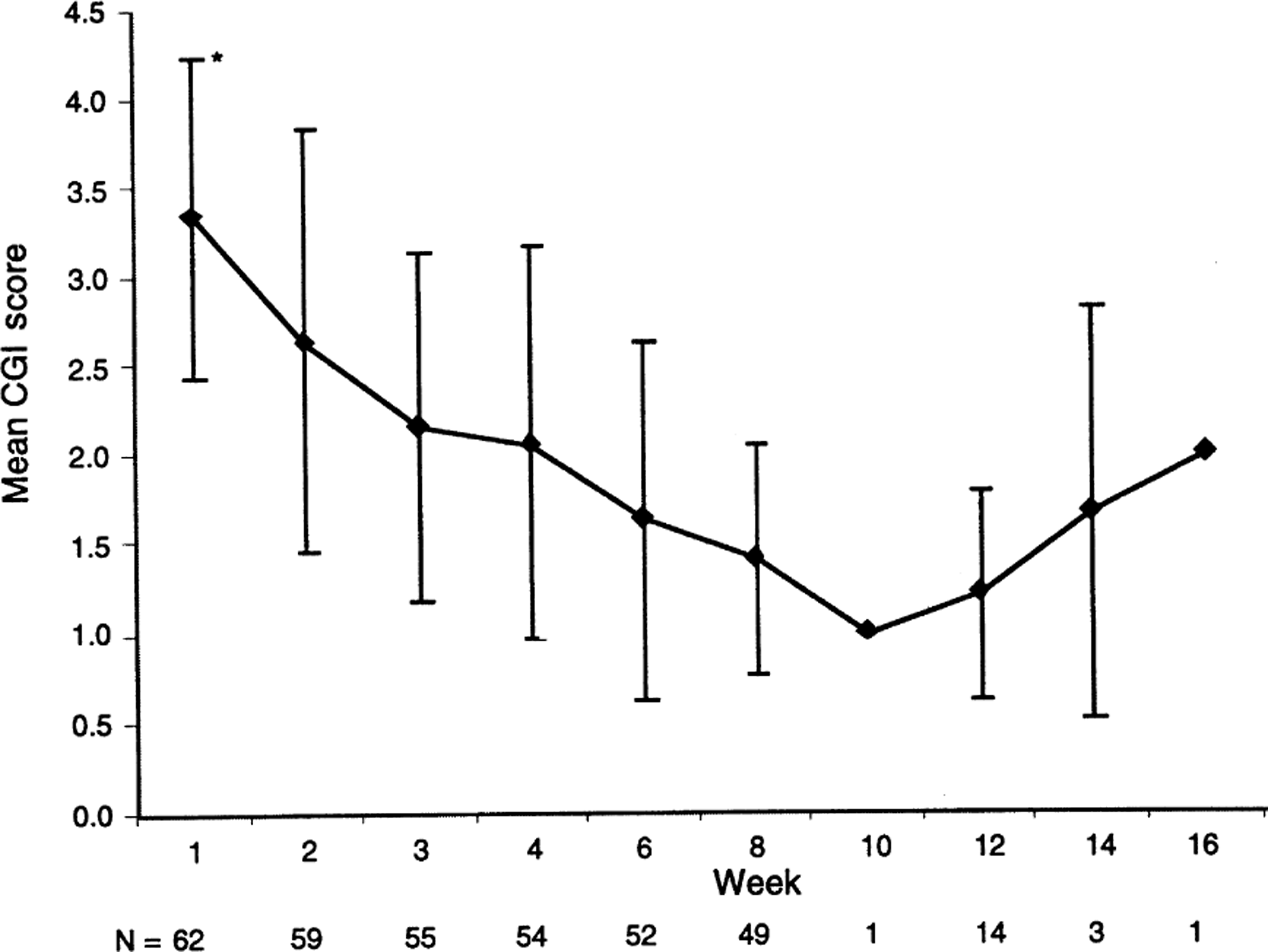
Mean Clinical Global Impression (CGI) score at each timepoint. Error bars indicate ± ISD.
The response rates were almost identical for the subgroup of 43 ITT patients whose optimal dose of sertraline was 50 mg/day (HAMD, 77%; CGI, 88%).
The mean HAMD total score decreased significantly from baseline to endpoint (p < 0.0001; Table 2). There were also clinically and statistically significant reductions in the secondary outcome measures of mean MADRS and BDI scores (p < 0.0001) (Table 2). The changes from baseline to endpoint in the individual items of anxiety and sexual interest on the HAMD subscales (LOCF analysis) are shown in Table 3. There was a reduction in all these items and sexual function was improved in both males and females.
Efficacy of sertraline therapy in the ITT and evaluable populations
Hamilton Rating Scale for Depression (HAMD) subscales (ITT population, LOCF analysis)
The investigators' subjective ranking of the overall efficacy of sertraline was considered to be excellent in 37 (77%) patients, good in nine (19%) and fair in two (4%).
Adverse events and tolerability
Of the 62 patients who received at least one dose of sertraline, 52 (84%) reported at least one adverse event during the study but only three (5%) withdrew from the study due to an adverse event. The most common adverse events noted during sertraline therapy were headache, nausea, diarrhoea and insomnia (Table 4). The incidence and type of adverse events reported were similar between patients who received sertraline 50 mg/day and those whose optimal dose was higher (100–150 mg/day).
Most frequent∗ adverse events recorded during sertraline therapy
The investigators' subjective rating of the overall tolerability of sertraline was excellent in 37 (60%) patients, good in 17 (27%), fair in five (8%), poor in two (3%) and unevaluable in one (2%).
Discussion
This study shows that sertraline is an effective and well-tolerated treatment in patients with major depression who have not responded to previous treatment with moclobemide for their current depressive episode. Improvements were seen on all primary and secondary outcome measures of depressive symptoms and global functioning. These improvements were both clinically and statistically significant.
The results of this open study are consistent with those of previously reported randomised controlled studies in which sertraline was found to be effective and well tolerated as a treatment for major depression [5,6,7,8,15].
Sertraline was well tolerated in the present study, and only 5% of patients withdrew because of adverse events. In general, the tolerability of moclobemide and SSRIs are similar although moclobemide tends to cause fewer gastrointestinal effects and does not appear to interfere with sexual function [10], which is a common side effect of SSRIs, TCAs and MAOIs [1]. In the present study, most patients were women and 10% of patients overall reported sexual dysfunction as an adverse effect. However, analysis of the HAMD subscales showed that sexual function was improved in both males and females.
One of the main limitations of this study is that it was an open label design. The lack of patient and investigator blinding is a potential source of bias but reflects more closely a real-life setting. This is also the case with the exclusion of patients with a history of non-response to sertraline, which is intended to reflect clinical practice.
There is a wide range of treatment options for patients who have had an inadequate response to the first-line antidepressant administered. The initial approach is often to switch to alternative monotherapy using an antidepressant from either the same or a different class. Recent open-label studies have suggested that 40–70% of patients unresponsive or intolerant to one SSRI, can respond to another SSRI [16,17,18]. Additionally, good therapeutic response rates to sertraline have been seen in patients who could not tolerate, or were resistant to TCAs [19,20]. Thus, sertraline may be effective in depressed patients irrespective of their previous response to other antidepressants.
In conclusion, although treatment decisions should always be made on an individual basis, the results of this study show that depressed patients with a history of non-response or intolerance to moclobemide can generally be treated successfully with sertraline.
Footnotes
Acknowledgements
This study was funded by Pfizer.
