Abstract
Objective:
Understand the perspectives of children who survived acute lymphoblastic leukemia (ALL) and their parents to adapt a guideline-based, family-based, intensive health behavior and lifestyle intervention treatment for this population.
Method:
Nine children 8–17 years of age (Mdn = 12 years [interquartile range, 10–16], median years off treatment = 5 [2–7]) who survived ALL and 11 parents participated in focus groups to assess perceptions of weight, weight-related behaviors, and perceived barriers to family-based behavioral weight loss treatment (FBT). Responses were analyzed thematically, and resultant adaptations were guided by the Framework for Reporting Adaptations and Modifications-Enhanced.
Results:
Topics and themes identified included mental and physical health concerns (e.g., treatment-related medical complications, body esteem), a perception of excess weight as protective, the continuing influence of eating habits established during cancer treatment (e.g., instrumental feeding practices, snacking), and potential barriers to activity (i.e., physical limitations, lack of sport experience). Resultant adaptations to FBT were contextual (e.g., virtual delivery) and related to the content, including an emphasis on weight management in the context of survivorship; education about late effects, overweight and obesity; increased emphasis on structured eating patterns and instrumental eating; provider recommended physical activity; and tailored emotion-focused and body esteem content.
Conclusions:
Focus groups for children who survived pediatric ALL provided insights that aided the adaptation of FBT for this population. A pilot trial of FBT for children who survived ALL and their parents is underway to evaluate acceptability, feasibility, and preliminary efficacy.
Implications for Impact Statement
This study elicited perspectives of children who survived acute lymphoblastic leukemia and their parents to inform the adaptation of an intensive health behavior and lifestyle treatment for this population. The adaptation, which is delivered entirely virtually and includes an increased emphasis on emotion-focused content and structured and instrumental eating patterns, may offer a promising approach to decrease the risk for chronic, treatment-related medical complications within this population.
Pediatric acute lymphoblastic leukemia (ALL) has a 5-year survival rate over 90% (R. L. Siegel et al., 2022). Children who survive ALL are at considerable risk for chronic, treatment-related medical complications known as “late effects,” including cardiovascular, pulmonary, and endocrine conditions (Mulrooney et al., 2019).
The risk for these late effects is exacerbated by excess weight. Rates of overweight and obesity (OW/OB) among children with ALL range as high as 40%–69%, surpassing rates within the general U.S. pediatric population of approximately 35% (Fryar et al., 2020; Zhang et al., 2014). The therapeutic regimens endured by children with ALL have been implicated in these elevated rates, however, accumulating evidence suggests that poor diet quality and inadequate physical activity are widespread, modifiable factors driving these conditions (Hansen et al., 2014). Importantly, food preferences are thought to be largely stable as early as 2–3 years of age, an age range which corresponds to the ages at which ALL diagnoses are most common (1–4 years; Nicklaus et al., 2005; D. A. Siegel et al., 2017). Thus, the poor-quality food habits developed among these children during cancer treatment may persist long after treatment is over. An intensive health behavior and lifestyle treatment (IHBLT) targeting OW/OB may help reduce the likelihood of late effects among children who survived ALL.
Guidelines for pediatric OW/OB proposed by the American Academy of Pediatrics, the American Psychological Association, and the U.S. Preventative Services Task Force recommend a family-based, multicomponent (e.g., addresses behavior change, diet, and physical activity) IHBLT delivered at a dose of ≥26 contact hours, with the number of contact hours directly proportional to the likelihood of achieving a significant reduction in body mass index (BMI; American Psychological Association, Clinical Practice Guideline Panel, 2018; Hampl et al., 2023; US Preventive Services Task Force et al., 2017). To our knowledge, only three interventions exist to treat OW/OB among children who survive cancer, none of which achieved a significant reduction in measures of child relative weight (e.g., BMI z-scores, BMI percentile; Huang et al., 2014; Stern et al., 2013; Wright et al., 2013). All three were multicomponent interventions and involved family participation, however, each was comprised of <10 contact hours. Currently considered the gold standard treatment for childhood obesity, family-based behavioral weight loss treatment (FBT) is an IHBLT designed to be delivered over ≥26 contact hours that uses a traffic light system, behavioral skills training, and socioenvironmental facilitation to help families improve dietary intake and physical activity behavior. FBT represents a promising approach to decrease the risk for late effects within this population (Hayes et al., 2018).
Although guideline-based, FBT was not developed for children who survived ALL. The incorporation of feedback from patients is a necessary component of treatment adaptation (Petkovic et al., 2020). After 2–3 years of intensive treatment with prolonged hospitalizations, chemotherapy, and steroids, children who survived ALL and their families may offer unique insight into FBT-related topics. Thus, the aim of the current study was to understand, for the first time, the perspectives of children who survived ALL and their caregivers and utilize this information to adapt FBT for this population. Our adaptation represents the first guideline-based, FBT for children who survived ALL.
Method
The Washington University Institutional Review Board approved the study protocol, procedures, and consent forms.
Participants
Medical record reviews conducted by the research team identified eligible children from the Childhood Cancer Survivorship Program at St. Louis Children’s Hospital. Eligible children were 6–18 years old at the time of enrollment, had a diagnosis of ALL, were at least 6 months postfinal cancer treatment and in remission, and met criteria for OW/OB (a BMI greater than the 85th percentile for age and sex). Children with a diagnosis of autism spectrum disorder and who were nonverbal or with a diagnosis of Down syndrome were excluded. Only English-speaking caregiver/parent–child dyads were eligible.
Procedure
Investigators contacted a caregiver/parent of each eligible child by phone to elicit interest and confirm eligibility. A member of the research team (Jessica Jakubiak) conducted informed consent.
Seven structured focus groups took place, and were audio recorded via Zoom, a Health Insurance Portability and Accountability Act (HIPAA)-compliant video communication platform, in October and November 2021. Groups were separated by age: three parent groups, two with 13–17-year-olds, and two with 7–12-year-olds. Groups lasted between 30 and 120 min and consisted of 2–7 participants. The group facilitator (Jessica Jakubiak) was an advanced doctoral student and a trained FBT interventionist.
Three semi-structured sets of questions based on participant age were used, with follow-up questions added to clarify responses as necessary. The question sets were generated with pediatric oncologists Allison A. King and Robert J. Hayashi, and behavioral health specialists, including Denise E. Wilfley. Questions addressed health concerns for the child who survived ALL, perceptions of weight, the impact of cancer treatment on eating and physical activity habits, and potential barriers to participation in FBT (see the
A brief demographic survey was completed by participating parents following their focus group. Each family received a $50 electronic gift card for their participation.
Focus Group Discussion Analytic Plan
Braun and Clarke’s (2006) thematic analytic approach was used to identify themes within the focus group discussions because of its usefulness in identifying the perceptions of participants whose views are relatively unknown. The group facilitator (Jessica Jakubiak) and two research assistants (Megan Guan and Sabir Khan) analyzed verbatim transcripts of each focus group according to Braun and Clarke’s (2006) six-phase thematic coding guide (a) coders familiarized themselves with the transcripts, and (b) developed an initial code set based on the questions posed and organized quotes accordingly. Over two subsequent coding passes, data-derived codes were added. (c) Code-specific quotes were organized into candidate themes which (d) coders then refined. To enhance transferability, we only included those themes identified by more than one participant. When two individuals from the same family endorsed a particular theme, at least one individual from a separate family endorsed the same theme. (e) Coders named and defined candidate themes and (f) an analytic narrative was agreed upon and presented to pediatric oncologists Allison A. King and Robert J. Hayashi, and behavioral health specialists, including Denise E. Wilfley, who helped inform resultant adaptations.
Adaptation Analytic Plan
The Framework for Reporting Adaptations and Modifications-Enhanced (FRAME; Wiltsey Stirman et al., 2019), a theoretical framework to systematically guide the process of adapting an evidence-based program, was employed to inform our adaptations. FRAME encompasses eight elements to comprehensively evaluate and report adaptations. (a) When the modification occurred, (b), whether the modification was planned, (c) who was involved in modification decisions, (d) what was modified, (e) at what level of delivery/for whom the modification was made, (f) what was the nature of modifications made, (g) the degree to which the modification is fidelity-consistent, and (h) the overall goal and contextual factors that contributed to the decision to modify (Wiltsey Stirman et al., 2019).
Results
Participants
Participant Demographics
Note. IQR = interquartile range.
As detailed in Figure 1, nine of the 40 (23%) potentially eligible families participated in the focus groups. Those who did participate were primarily non-Hispanic, Caucasian individuals (89%) who were a median of years 5 off treatment (interquartile range, IQR 2–7).Participant Flow Chart
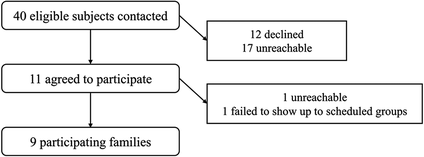
Every family requested to be contacted about the intervention when enrollment began.
Focus Group Discussions
Health Concerns
No children mentioned a specific concern for late effects, however, this was a theme endorsed by six of 11 parents who discussed late effects generally, cardiac disease, endocrine abnormalities, and fertility issues. Seven of the 11 parents and six of the nine children specifically discussed concern regarding their child’s weight and/or a concern for late effects exacerbated by excess weight.
Mental Health Concerns
Concerns about mental health were a prominent theme spontaneously introduced by nine of the 11 parents and four of the nine children. Four parents specifically cited “emotional issues,” impulsivity, and worry about their child processing their experience with ALL. Self-blame was a theme expressed by four parents and one child. Finally, three parents and two children spontaneously spoke of body image concerns and a lack of body acceptance with examples including hair loss, steroid-related “moon face,” and dramatic changes to height and weight. These comments align with previous literature indicating that subsets of children who survive cancer have poorer body image than healthy controls, particularly those with chronic health conditions (i.e., OW/OB; Belle et al., 2022).
Weight Perceptions
Two of the 11 parents and one of the nine children associated excess weight with increased energy, better health, and a higher likelihood of survival from a cancer diagnosis. Five parents and four children expressed concern regarding the child’s weight. However, on the parent-reported demographic survey, when presented with categories to describe their child’s weight status, only one parent reported that their child was significantly overweight. All other responses were split between a little overweight (n = 4) and a healthy weight (n = 4) despite a median BMI percentile of 97 (IQR 93–98).
Eating Habits
Encouragement from their child’s treatment team to increase caloric consumption during treatment was described by five of the 11 parents. Emotional and instrumental feeding practices (i.e., use of food as a reward to motivate, promote, and reinforce good behavior) were reported by four parents, including the use of favorite foods as rewards and/or comfort after medical procedures as well as food choices being a source of perceived control. “Constant” snacking was discussed within every parent group and by one child. Five parents reported that such behaviors problematically influence their child’s current eating patterns.
Physical Activity
Four of the 11 parents spoke of concern about their child’s posttreatment involvement in competitive sports given the amount of time they took off from sports while peers continued to participate and progress. Additionally, six parents described several perceived physical limitations of their children, including stiffness, lack of agility, and being “a little bit slower” than peers. No child expressed these concerns.
Adaptations
Proactive, preimplementation adaptations were made to FBT content and context, with the goal of improving fit, feasibility, and retention for children who survived ALL and their families. Figure 2 details the specific adaptations made within each of the eight FRAME elements, as well as our rationale for such modifications (e.g., sociopolitical, organizational, provider, and recipient factors). Table 2 illustrates themes identified within our focus groups and the specific adaptations directly informed by each. Note. FRAME = framework for reporting adaptations and modifications-enhanced. Note. FBT = family-based behavioral weight loss treatment; CBT = cognitive behavior therapy.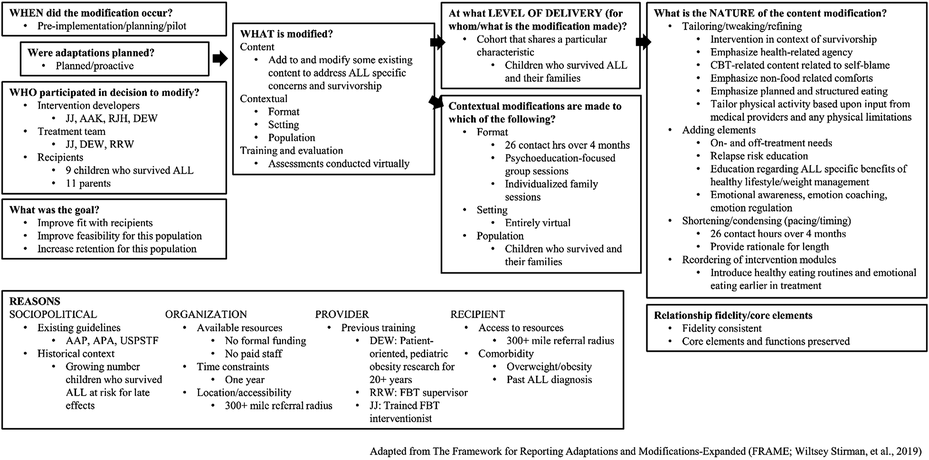
Themes, Representative Quotes, and Adaptations
FBT emphasizes dietary modification, increased physical activity, and behavior change skills to improve health and manage weight. FBT utilizes traffic light eating and activity plans in which RED, YELLOW, and GREEN labels guide families toward consuming low energy-dense, high nutrient-dense foods consistent with individual, familial, and cultural preferences and increasing moderate-to-vigorous physical activity and decreasing sedentary activity. FBT also includes training in a variety of behavioral skills including self-monitoring, stimulus control, planning, goal setting, and problem-solving. Parents are taught positive parenting methods to shift health behavior choices, including the use of limit setting and reinforcement, the importance of modeling healthy behaviors, and how to create a healthier home environment. FBT facilitates socioenvironmental support to optimize the durability and generalizability of improved health habits across multiple contexts (e.g., home, school, with friends) and helps participants manage negative peer interactions (e.g., teasing) that may hinder healthy behaviors (Wilfley et al., 2017). To maintain fidelity, all existing FBT content is included, though some are condensed.
Modifications were made to the way FBT is introduced. Education regarding associations between medical complications and a healthy lifestyle and weight management is included, and FBT is presented not as a distinct, novel treatment focus, but opportunity to make behavioral changes that may enhance long-term survival. The positive, proactive nature of FBT is emphasized throughout the intervention. Moreover, participant input regarding how cancer influenced FBT topics is elicited throughout to normalize the experiences of these families.
Several traditional FBT topics are emphasized and/or presented earlier in treatment, including planned and structured eating patterns as well as emotional eating and alternative coping strategies. Existing cognitive behavior therapy (CBT) techniques are extended to address self-blame and additional emotional awareness and emotion regulation strategies are incorporated. Body esteem content is tailored for this population, to target treatment-related physical changes and body appreciation, including its role in survival.
Additional content was also added to the intervention. Differences between on and off-treatment needs and behaviors are addressed, normalized, and validated while simultaneously advocating for change. Child participation requires physician approval through which potential physical limitations will be elicited. The individualized nature of FBT is utilized to emphasize healthy body movement dependent on an individual child’s capabilities.
Intervention adaptations must consider the influence of culture within different populations. As previously noted, focus group participants were primarily non-Hispanic and Caucasian, which may limit our understanding of the experiences of the broader population of child survivors of ALL. Drawing from reviews of culturally tailored weight management interventions among minority populations, we expanded an existing FBT resource designed to assess eating habits to help us better understand ways in which each family’s culture, ethnicity, and socioeconomic context may influence health-related beliefs and behaviors (e.g., weight perceptions, household-make up and social supports, societal stressors; Peña et al., 2012).
Finally, to improve feasibility the program was condensed to include the recommended 26 contact hours over a 4-month period, in line with previous FBT trials and parental input (e.g., “6 months seems like a long time”; Wilfley et al., 2017). The adaptation is delivered and evaluated entirely via Zoom to eliminate travel time, a potentially substantial barrier to treatment given the large referral radii (i.e., 300 miles) of specialty oncology centers. Additionally, given our large eligible age range (5–18 years), treatment groups will be separated by age: parents, 13–17-year-olds, and 7–12-year-olds. Parent and child groups will occur simultaneously to consolidate the number of contact hours. Additionally, the intervention will include both group and individual components to normalize the experiences of the families and facilitate social support while allowing for individualization.
Discussion
Themes reflective of the experiences and preferences for an IHBLT for children who survived ALL and their parents were identified through focus group discussions and used to inform an adaptation of FBT specific to this population. Adaptations were contextual (e.g., virtual delivery) and content-related (e.g., reordering of modules, adding elements).
This study has several strengths. Given the family-based nature of FBT, we thought it particularly pertinent to elicit feedback from both children who survived ALL and their parents. To our knowledge, this is the first study to employ the use of focus groups to do so. Additionally, the inclusion of families representing a wide range of time off treatment (2–11 years) and child age (8–17 years) will facilitate our findings’ transferability to the entire population of children who survived ALL. Moreover, the standard by which sample sizes for qualitative research are conducted is saturation, which is believed to exceed 90% with 4–6 focus groups (Guest et al., 2017). Our study included seven. Additionally, a multidisciplinary team was closely involved in the focus group design, generation of question sets, and resultant adaptations to FBT. We hope that this allowed for a broad understanding and application of the experiences of these children and their families. Finally, the study employed the use of two established methodologies to systematically guide both the qualitative analysis (Braun & Clarke, 2006) and the development of the resulting adaptations (Wiltsey Stirman et al., 2019).
Several weaknesses must also be noted. Despite data to show that the highest rates of OW/OB in children with ALL occur among Hispanic and non-Hispanic black children, our sample was primarily white and non-Hispanic (89%; Brown et al., 2016). However, drawing from reviews of culturally tailored weight management interventions among minority populations, we expanded an existing FBT resource to better understand ways in which each family’s culture, ethnicity, and socioeconomic context may influence health-related beliefs and behaviors (Peña et al., 2012). Additionally, families willing to engage in focus group discussions regarding an IHBLT likely represent those for whom this topic is of particular interest and their opinions may not transfer to the wider population of children who survived ALL and their families. Similarly, given that participating children were recruited from the Childhood Cancer Survivorship Program at St. Louis Children’s Hospital which sees patients who are at least 2 years off treatment, their opinions may not reflect those whose treatment experiences were more recent nor those families who are not actively engaged in follow-up care, for whom late effects may not be perceived as concerning.
We hope that the information acquired from these focus groups and the resultant adaptations made to FBT will allow us to determine whether this approach will result in the same improvements in eating and activity behaviors among children who survived ALL that has been achieved with other groups, including the reversal of some of the poor diet quality and inadequate physical activity habits potentially developed during treatment (Epstein et al., 2007; Wilfley et al., 2017). FBT is an evidence-based practice, and an adaptation for families of children who survived ALL will facilitate a relevant and much-needed intervention. Success in improved eating and activity habits and sustained weight loss in this population could lead to a reduction in late effect-related morbidity and an improvement in overall survival.
