Abstract

Case Description
Three medical students rented a townhouse in a West Virginia summer golf resort over spring break. After the first night of their stay, 1 student complained of pain in the back of his right calf. Over the next 2 d, a papule developed at the site of the discomfort. It ulcerated the next day. The pain and the papule were associated with a fever of 38.5°C (101.3°F), malaise, headache, and photophobia (Figure 1). The other students described the lesion as a black, tick bite eschar and diagnosed their classmate’s condition as Lyme disease without a bull’s-eye target rash.
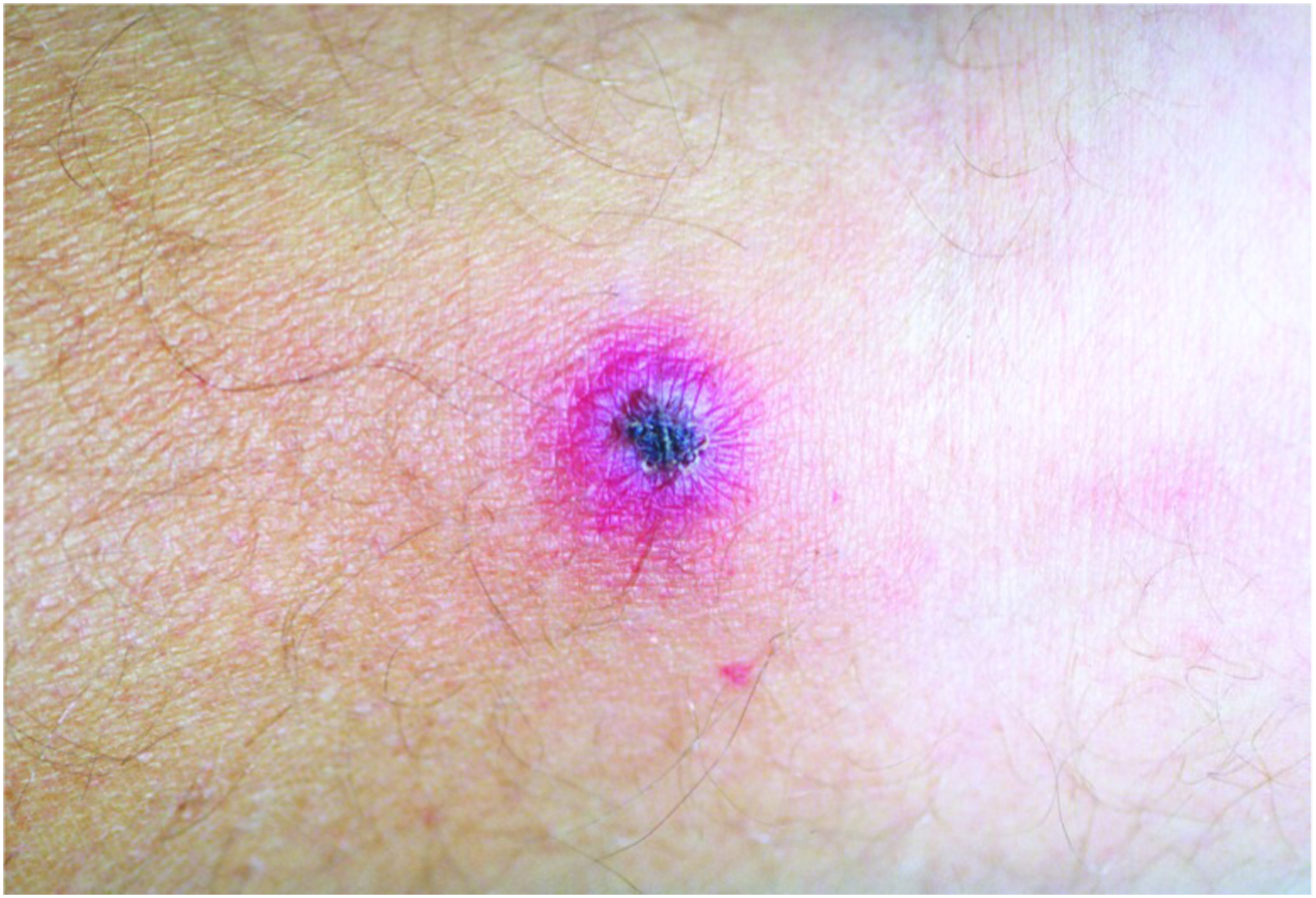
A black scab surrounded by reddish-purplish erythema, similar to lesion in the case described here. Photo is in the public domain; no copyright permission required. 1
Since the patient had a history of severe dermatologic reactions to tetracyclines, including doxycycline, his classmates advised a provider at a nearby urgent care facility to start amoxicillin therapy for Lyme disease, 500 mg orally three time a day for 14 d. Over the next 2 d, the patient’s condition worsened, his temperature rose to 39.2°C (102.6°F), and he developed a papulovesicular rash on his anterior chest and shoulders. He was admitted to a regional tertiary care center.
What is the diagnosis? How would you treat this patient?
Diagnosis
Rickettsialpox
Causative pathogen
Rickettsia akari
Management
Admitting physicians in the emergency department (ED) obtained additional history that there were no wildlife, rodent, or tick exposures, but the townhouse had a year-round resident indoor/outdoor cat living in the basement next to the heater. The eschar was described as painful on palpation. It had a black base surrounded by a violaceous ring with a clear exudate expressed on compression (Figure 1). A tender right popliteal lymph node was also palpable. There were about 20 maculopapular erythematous lesions, many with small, central weeping vesicles scattered across the patient’s anterior chest and shoulders.
Laboratory testing showed normal values for electrolytes, creatinine, hematocrit, and leukocytes. The platelet count was 85,000/mm3 (normal, 150,000–450,000/mm3). Routine blood cultures were sterile.
Despite a negative exposure history, ED physicians were concerned about cutaneous anthrax because of the black eschar (Figure 1). They submitted swab samples from the base of the eschar, from the weeping lesions on the chest wall, and from a lymph node aspirate to the state public health laboratory. An infectious disease consultant recommended discontinuing amoxicillin and starting oral ciprofloxacin, 500 mg twice a day for 10 d, for inoculation anthrax.
Alerted to the possibility of anthrax, state laboratorians conducted Gram and special staining, direct immunofluorescent antibody testing, and polymerase chain reaction (PCR) assays on the specimens submitted under biological safety level 3 conditions. Since Rocky Mountain spotted fever was endemic in the region and also characterized by fever, eschar, and rash, state laboratorians tested all specimens for Rickettsia rickettsii.
A preliminary diagnosis of rickettsialpox was made based on microscopic, immunohistochemical, and PCR results. Five days later, the diagnosis was serologically confirmed as positive based on significantly higher reciprocal immunoglobulin (Ig)M antibody titers to R akari than to R rickettsii antigens. Cultures of the submitted specimens also grew R akari sensitive to doxycycline, azithromycin, and chloramphenicol.
Ciprofloxacin was discontinued, and the patient was treated with oral azithromycin, 500 mg daily for 10 d. Within 48 h, the patient became afebrile, his other symptoms resolved, and he was discharged to complete his antibiotic therapy for rickettsialpox as an outpatient. All lesions healed without scarring.
Discussion
Rickettsialpox is caused by the gram-negative, intracellular, spotted fever-group rickettsial bacterium R akari, with a host rodent reservoir in the common house mouse (Mus musculus). 1 Rickettsialpox is transmitted to humans by bites from infected house mouse mites, Liponyssoides sanguineus (Figure 2).

The adult house mouse mite, Liponyssoides sanguineus, the mite vector of rickettsialpox. Source: United States Centers for Disease Control and Prevention. Public Health Image Library (PHIL). PHIL ID 5447. Public domain. No copyright permission required. Available at
Life Cycle and Feeding Behavior
The house mouse mite’s life cycle begins with a larval form hatched from an egg that undergoes 2 nymph stages before maturing into an adult. Nymphs and adult mites feed on R akari–infected house mice every 3 to 4 d for 1 to 2 h before dropping off engorged. 2 After feeding, infected adult females may lay rickettsia-infected eggs. These eggs complete the mite’s life cycle. R akari bacteria are subsequently passed by the molting process through the mite’s developmental stages from egg to adult. 2
Epidemiology
The disease ecology of rickettsialpox is characterized by a worldwide epizootic cycle of asymptomatic maintenance of R akari transmitted by house mouse mites in the preferred rodent reservoir of house mice. Although the common house mouse, M musculus, is considered the only true rodent reservoir host for mite-transmitted rickettsialpox, other rodent reservoirs have been suspected outside of the United States but remain unconfirmed. Suspected rodent reservoirs include the Korean vole, Mongolian gerbil, and Egyptian gerbil.1,2
Although nymph and adult mites typically feed on house mice every 3 to 4 d, they can go for week-long periods without feeding. This feeding-starvation behavior has resulted in cluster outbreaks of rickettsialpox in apartment complexes in large US cities, including New York, Boston, Cleveland, Philadelphia, and Pittsburgh, following mouse extermination programs that encouraged house mouse mites to seek alternative human hosts (Figure 2).1,3
Investigators agree that rickettsialpox is an internationally underreported infection for several reasons, including the following: 1) the worldwide distribution of house mice and their mites; 2) additional suspected rodent reservoirs of R akari; 3) a high seroprevalence of R akari infection in domestic animals, especially dogs, in major metropolitan areas; 4) a high incidence of undiagnosed, empirically treated cases of rickettsialpox susceptible to doxycycline and a broad range of other antibiotics; and 5) an unknown prevalence of patients with subclinical rickettsialpox who recover without antibiotic therapy. 1 -3
Clinical Manifestations
The diagnosis of rickettsialpox may be based on a history of exposure to house mice and a triad of fever, eschar, and vesicular rash. The incubation period is 7 to 14 d. The prodromal symptoms of rickettsialpox begin 2 to 3 d after a painless bite by a house mouse mite, L sanguineus (Figure 2). Common symptoms include malaise, headache, photophobia, stiff neck, myalgia, and high fever of 38.3 to 40.0°C (101–104°F). 1 Additional prodromal symptoms may include nausea, vomiting, conjunctival injection, cough, and pharyngitis. 1
The mite bite eschar is the classic hallmark of infection. It follows the house mite bite by 2 to 3 d and precedes the rash by 1 to 3 d. The eschar is umbilicated with surrounding reddish-purplish erythema, crusts over in 1 to 2 d, and heals later without significant scarring within 2 to 3 wk (Figure 1). 1
The rash phase is delayed, beginning several days after the onset of the prodrome and starts with maculopapules that progress to papulovesicular lesions. These lesions eventually darken, scab over, crust, and fall off within 10 to 12 d without scarring (Figures 3 and 4). 1 Patients usually have 20 to 40 papulovesicular lesions, distributed primarily on the trunk and upper extremities, sparing the palms and soles. This is in contrast to other spotted fever-group rickettsioses, such as Rocky Mountain spotted fever, that cause lesions on palms and soles.
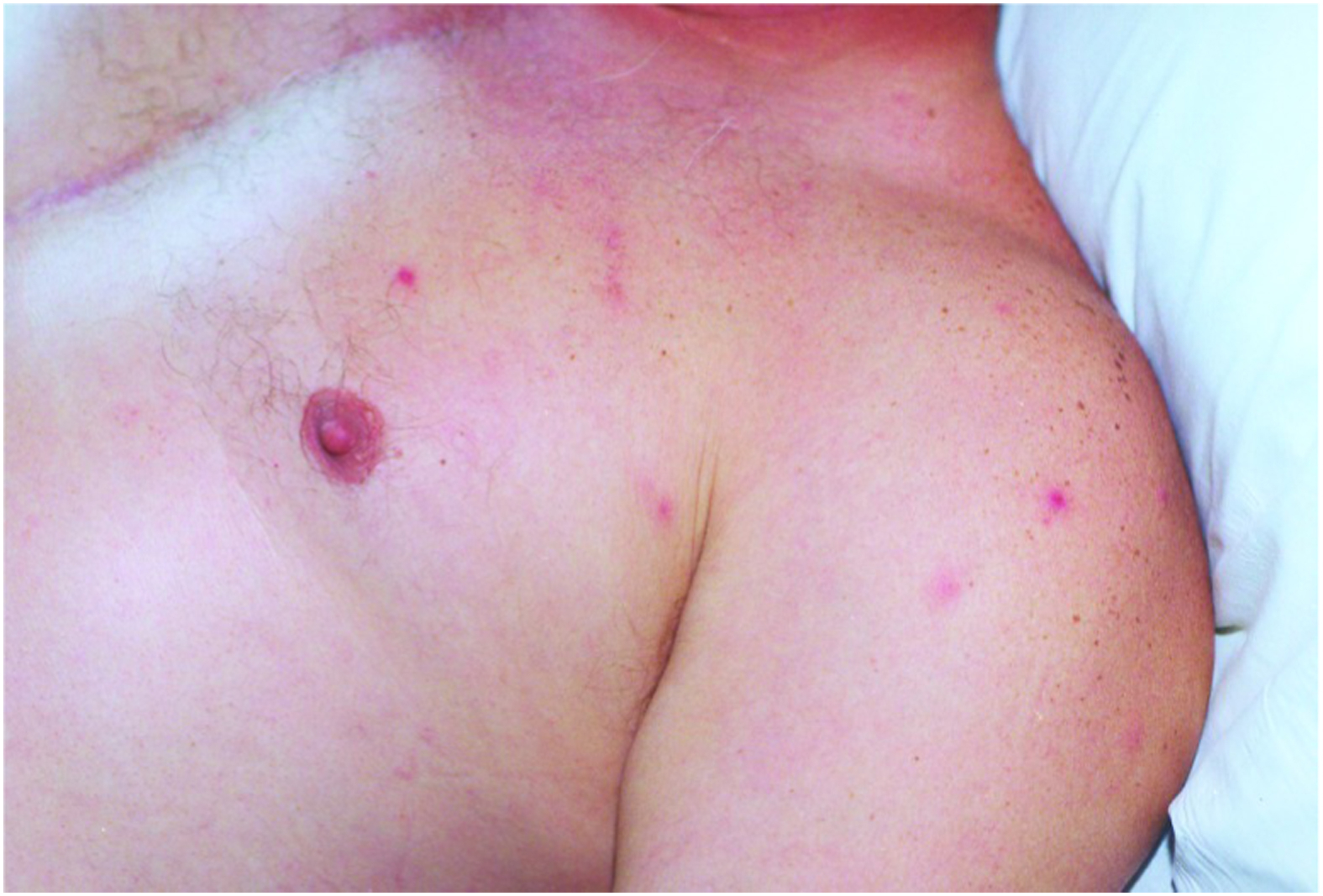
Papulovesicular lesions on the upper trunk of a patient with rickettsialpox. Source: Krusell A, Comer JA, Sexton DJ. Rickettsialpox in North Carolina: a case report. Emerg Infect Dis. 2002;8(7):727–728. United States Centers for Disease Control and Prevention. Public domain. No copyright permission required. Available at
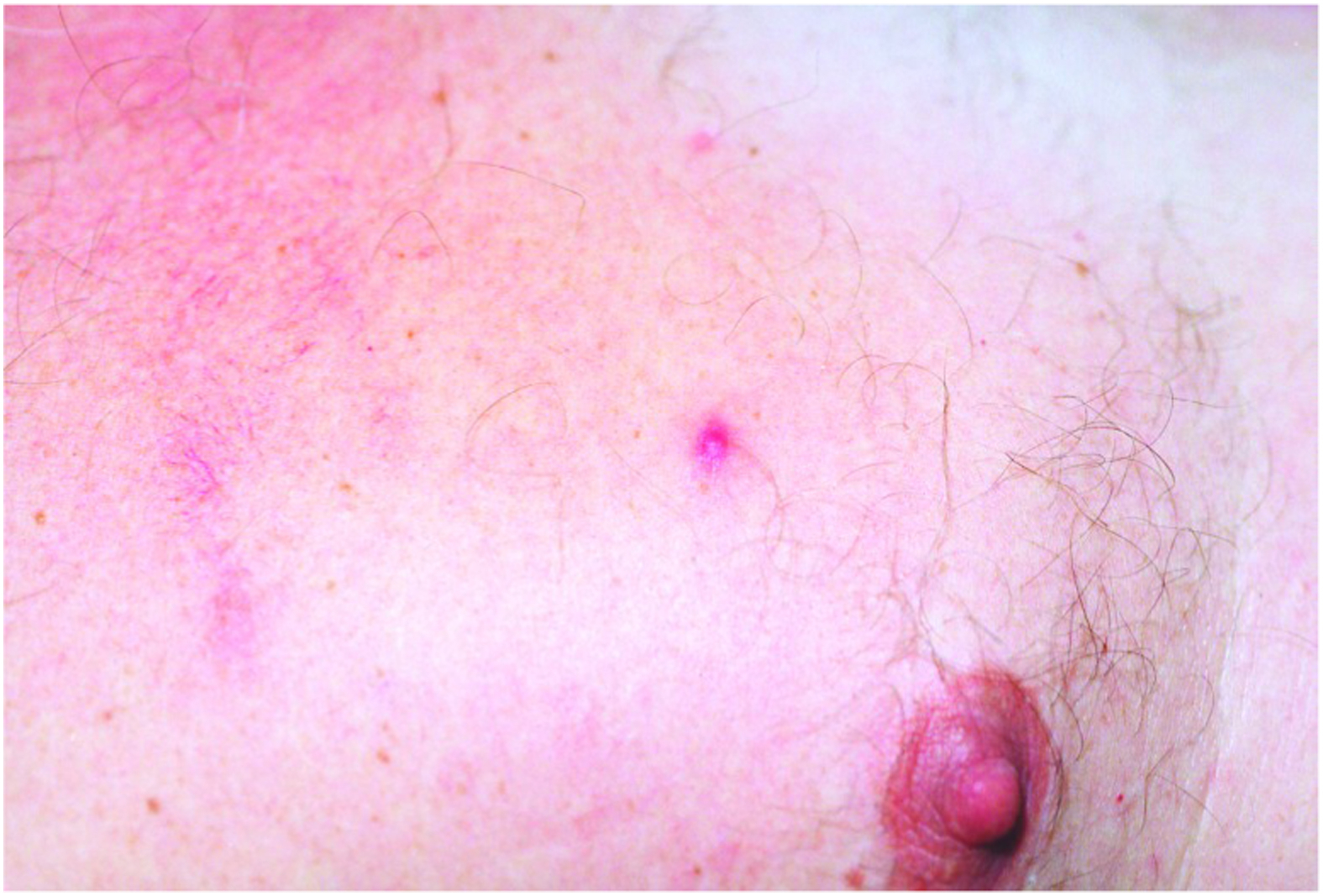
Closer view of papulovesicular lesions on a patient with rickettsialpox. Source: Krusell A, Comer JA, Sexton DJ. Rickettsialpox in North Carolina: a case report. Emerg Infect Dis. 2002;8(7):727–728. United States Centers for Disease Control and Prevention. Public domain. No copyright permission required. Available at
Differential Diagnosis
The cutaneous manifestations of rickettsialpox may be distinguished from those of chickenpox and other infections causing vesicular rashes (smallpox, herpes zoster, and herpes simplex) by the appearance of ≥1 eschars at mite bite sites before the rash starts. The cutaneous manifestations of rickettsialpox also begin with papules and not vesicles as in chickenpox. Rickettsialpox has a single crop of vesicles, while chickenpox has successive crops of vesicles. 1
The incubation period, prodromal manifestations, and eschars of rickettsialpox resemble those of inoculation anthrax, larval trombiculid mite–transmitted scrub typhus, Rickettsia parkeri, and other endemic fevers transmitted by tick bites, including spotted fevers in the United States, African tick bite fever in East Africa, Queensland tick typhus in Australia, and numerous others. Inoculation or cutaneous anthrax is characterized by a dark eschar that is not followed by a vesicular rash. Cutaneous anthrax can be rapidly excluded in the diagnosis of R akari infection by Gram staining, direct immunofluorescent antibody testing, or PCR on swab samples of vesicular fluid from the base of the eschar.
In mite-borne scrub typhus and other tick-borne rickettsial cases, an eschar forms at the arthropod bite site within 3 to 7 d. Fever, chills, severe headache, regional lymphadenopathy, and truncal maculopapular and then vesiculopustular rash follow the appearance of the eschar. Scrub typhus and other tick-borne rickettsial diseases with eschars can also be excluded based on their geographic distributions as well as serologic and molecular testing of samples obtained from eschar swabs, draining vesicles or pustules, and lymph node aspirates.
Laboratory Diagnosis
Transient leukopenia, thrombocytopenia, and elevated aminotransaminase levels are common abnormal laboratory findings. 1 Although these laboratory findings are shared across many tick- and mite-borne zoonotic illnesses, appropriate history taking, including location, timeframe, geographic area, attention to rash, and other specific signs and symptoms, may narrow down the etiology of the specific zoonotic illnesses, which closely mimic each other. Positive laboratory diagnostic findings can begin at the bite site where eschar swabs and biopsies may reveal R akari antibodies by direct immunofluorescent antibody testing. Swabs of the eschar and weeping vesicles also permit detection of R akari DNA by PCR (Figures 1 and 4). During the febrile phase, blood cultures may isolate R akari. Serologic tests may detect IgG and IgM antibody titers to rickettsial antigens 7 to 15 d after onset of the illness. Cross-reactivity leading to positive serology also occurs in patients with prior Rocky Mountain spotted fever caused by R rickettsii. 1
Treatment
Rickettsialpox may resolve on its own without antibiotic therapy in uncomplicated cases and require only supportive treatment. However, antibiotic therapy is recommended in all cases to shorten the course of illness and to prevent complications, including interstitial pneumonia, chronically elevated transaminase levels from liver damage, and central nervous system involvement.
Although R akari is sensitive to many antibiotics, doxycycline is the preferred antibiotic for treatment. The initial oral loading dose is 200 mg, followed by an oral maintenance dose of 100 mg every 12 h for 7 d.1,3,4 Erythromycin, azithromycin, and tetracycline may also be effective in treating rickettsialpox but are not recommended as first-line treatments. Chloramphenicol is an effective alternative antibiotic but is also not recommended because of its bone marrow toxicity.
Prevention and Control
Prevention and control strategies include house mouse rodent control and house mite vector extermination programs.
Footnotes
Acknowledgements
Disclaimer: This is a fictitious case based on a published case report. 1
