Abstract
Introduction
Improper use of camp stoves in enclosed spaces has resulted in fatalities from carbon monoxide (CO) poisoning. Prior research has focused on the CO output of stoves burning white gas, unleaded gas, or kerosene. Stoves burning an isobutane/propane fuel have not been investigated and are the focus of this study.
Methods
Three stoves utilizing isobutane/propane fuel were used to heat a pot of water inside a 3-season tent under controlled settings. Multiple runs with each stove were performed, and CO measurements, in parts per million (ppm), were recorded at 1-min intervals for a total of 15 min using a RAE Systems gas monitor. Data are reported as mean with SD. Repeated measures analysis of variance was utilized to examine changes over time. Statistical significance was set at P<0.05.
Results
There was a statistically significant main effect of time and CO level, F (14, 168)=7.6, P<0.001. There was a statistically significant difference between-subjects effect of stove group F (2, 12)=8.6, P=0.005, indicating that CO levels were different depending on the stove. Tukey’s post-hoc analyses revealed that stove A had the highest CO levels. The average level of stove A was statistically significantly higher than that of stove B and stove C, with a mean CO level difference of 79 ppm (95% CI, 3–156), P=0.043 and 117 ppm (95% CI, 40–194), P=0.004, respectively.
Conclusions
Stoves utilizing isobutane/propane fuel can produce unsafe CO levels and should not be used in enclosed spaces.
Introduction
Carbon monoxide (CO) is a by-product of carbon combustion and acts as an asphyxiant gas. In the body, CO combines with hemoglobin to form carboxyhemoglobin (COHb), which is unable to transport oxygen. It has a hemoglobin binding affinity 200 to 250 times that of atmospheric oxygen. CO is difficult to detect, as it is tasteless, odorless, and colorless. Toxicity from CO represents an important cause of morbidity and mortality, with greater than 1500 deaths per year in the United States attributed to CO poisoning between 1999 and 2014. 1 Approximately 400 of these were considered accidental and, therefore, potentially avoidable. While most CO poisonings occur in urban environments from home heaters and gas leaks, risk also exists in backcountry settings. Improper portable camp stove use in enclosed spaces has previously been described in the literature and resulted in multiple fatalities. 2 A California review of unintentional CO-related deaths over a 9-year period, between 1979 and 1988, found 10 deaths attributed to camping equipment. 3 Additionally, a Centers for Disease Control and Prevention report from 1999 detailed 6 total deaths from 2 different incidents involving the use of camping stoves inside a closed tent. 4
A prior study compared the CO output of a multifuel stove using white gas, unleaded gas, or kerosene in an enclosed space. 5 All 3 fuel types rapidly generated high levels of CO. A difference in CO output was noted between fuel sources with white gas, producing the least amount of CO, and kerosene, producing the most. More recently, a study investigated the effect that varying the type of fuel and type of tent had on CO accumulation, showing that in a 3-season tent, unleaded gas produced statistically significant higher levels of CO than white gas. 6 While no statistically significant difference between the fuel types was observed in the 4-season tent, the absolute CO levels were higher than those in the 3-season tent. This is likely the result of the construction of 4-season tents, which are typically constructed from thicker fabric and have less ventilation in order to stand up to the harsher environment during winter.
Canister stoves using an isobutane/propane fuel mixture are popular among backcountry enthusiasts due to their low weight, portability, and ease of use. A literature review found no studies investigating the CO output of these stoves. The aim of this study was to investigate the CO emissions of isobutane/propane burning canister stoves.
Methods
The University of Louisville institutional review board (IRB 20.0742) reviewed and approved the study protocol. Three different canister stoves utilizing isobutane/propane fuel were selected for the study. The Primus Power Trail Stove (Fenix Outdoor International AG, Stockholm, Sweden), Jetboil MightyMo Stove (Johnson Outdoors Inc, Racine, WI), and MSR PocketRocket 2 Stove (Cascade Designs, Inc, Seattle, WA) were chosen based on name brand recognition and similar stove design and are referred to as stove A, stove B, and stove C, respectively. The power output of the stoves was obtained from the manufacturer and in outputs measured in kilojoules per hour (kJ/h) was 14,243, 10,550, and 8651 kJ/h, respectively. The study stoves were purchased in new condition specifically for this study and paired with new isobutane/propane canisters of the same brand name. The fuel canisters all used an 80/20 mixture of isobutane/propane. Each stove underwent 2 trial burns of 15 min to ensure proper functioning prior to data collection.
The Alps Mountaineering Zephyr 3 (Alps Brands, New Haven, MO), a 3-season tent, was chosen because it was readily available to the authors. The tent was in like-new condition. The tent walls were mesh, and the floor and fly were made from 75D 185T polyester, which is a relatively common fabric thickness and thread count for 3-season tents. The tent had a floor area of 3.72 m2 and a height of 1.04 m. The tent, fly, and footprint were assembled per manufacturer’s instructions with all flaps, zippers, and openings on the tent placed in a closed position. The tent was set up in a large firehouse bay with the bay doors open to allow adequate ventilation for researcher safety. Each stove was placed in the center of the tent. After ignition, the stove was set to the maximum output. A 2-L pot of ambient temperature water was placed on the burner for the duration of the trial. The water was exchanged between trials, and the temperature was not standardized. The tent doors were closed for the duration of each trial. The trials were performed on a single day in late April between 0800 and 1800. The local ambient temperature was between 20.0°C and 26.7°C, and the average atmospheric pressure was 99.8±0.1 kPa according to the US National Weather Service. The trials were performed at approximately 141 m above sea level. There was no appreciable crossbreeze during each trial. CO measurements, in parts per million (ppm), were recorded at 1-min intervals for a total of 15 min using a RAE Systems QRAE PGM-2500 Gas Monitor (Honeywell, San Jose, CA). The sampling tubing end placed 0.61 m above the stove/pot set up was chosen to attempt to minimize introduction of excess water vapor into the tubing. The gas monitor was equipped with a hydrophobic filter to prevent water vapor intake into the CO sensor. The gas monitor was calibrated prior to the start of the study in ambient air using the internal calibration function on the monitor.
Stoves were individually placed inside the tent for the duration of each trial. The trial proceeded in a stove A, stove B, stove C order to complete 1 round and was repeated for 5 rounds. The order of the stoves was chosen arbitrarily and did not vary between rounds. The stoves were allowed to cool between each trial. The tent was ventilated between each stove use until the CO monitor returned to zero. We report CO levels over time, in 1-min intervals with means and SDs. Repeated measures analysis of variance was utilized to examine changes over time. Tukey’s post-hoc testing was used. When reporting differences between stoves, we report both CIs and P values. Statistical significance was set at P<0.05. IBM SPSS version 27 was utilized for data analysis.
Results
Table 1 provides stove CO levels. There was a statistically significant main effect of time and CO level, F (14, 168)=7.6, P<0.001. There was a statistically significant between-subjects effect of stove group F (2, 12)=8.6, P=0.005, indicating that CO levels were different depending on the stove. Tukey’s post-hoc analyses revealed that stove A had the highest levels. The average level of stove A was statistically significantly higher than stove C, with a mean difference of 117 ppm (95% CI, 40–194), P=0.004, and stove B, with a mean difference of 79 ppm (95% CI, 3–156), P=0.043. Figure 1 visualizes CO level over time by stove group.
Descriptive statistics for stove carbon monoxide levels
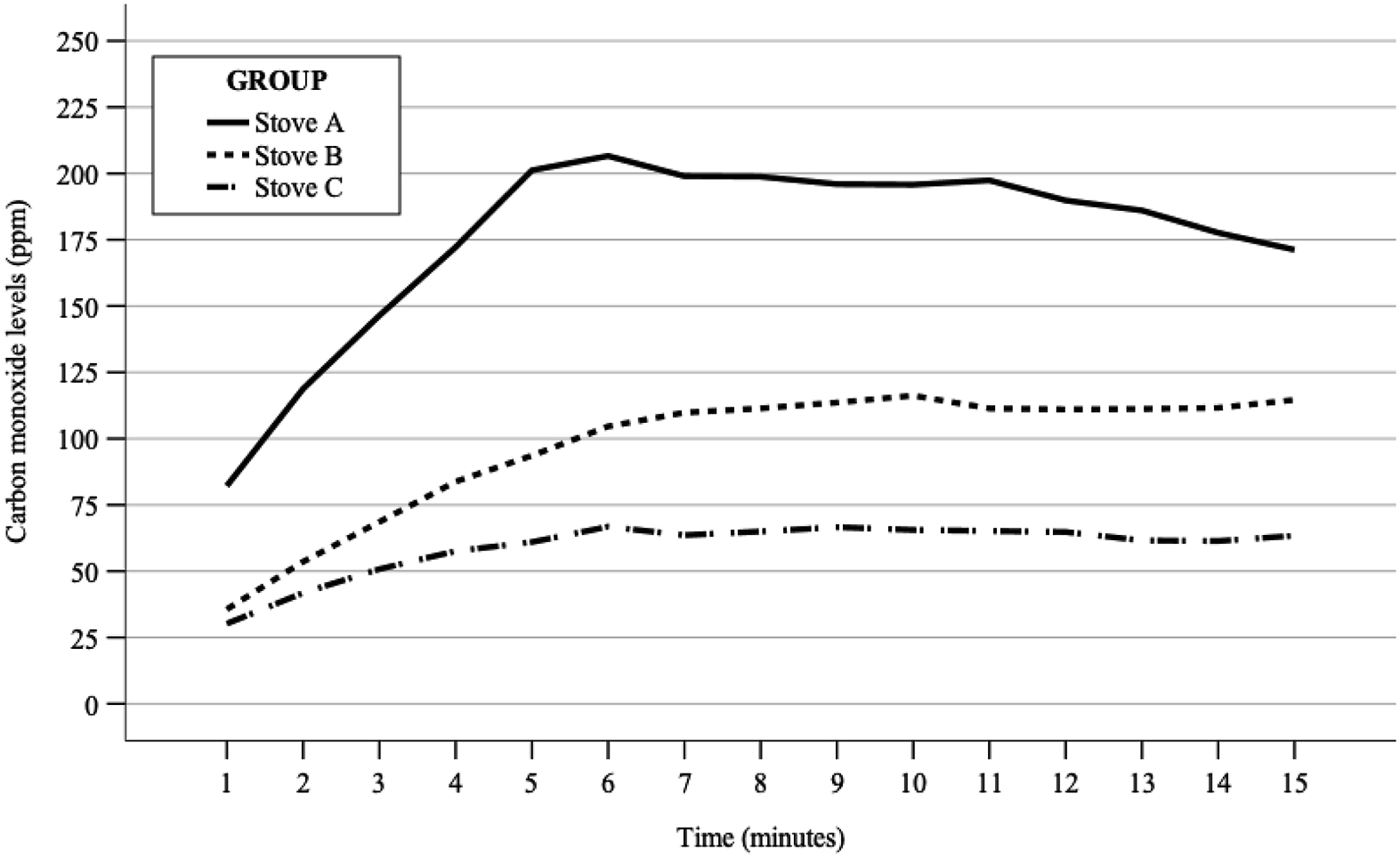
Average carbon monoxide levels for each stove at 1-min intervals. ppm, parts per million.
Discussion
Isobutane/propane stoves can generate high levels of CO inside an enclosed 3-season tent. Based on our results, a higher kJ/h output appears to correlate with increased generation of CO and is a possible explanation for the differences seen between stoves despite their similar design and fuel source.
A study investigating CO accumulation of white gas, unleaded gas, and kerosene showed maximum CO levels of 348±76, 464±32, and >999 ppm, respectively. 5 Comparison to this study would suggest that isobutane/propane produce less CO than the other fuel types. However, the comparison is imperfect because the study used CO measurements taken from inside a cardboard box that had a volume of 0.4 m3, which is smaller than the volume of the tent used in this study. Additionally, the box lacked any ventilation. A more robust comparison may be obtained from a study investigating stoves burning white gas and unleaded gas in a 3-season and 4-season tent. 6 The 3-season tent used in the study had a floor area of 4.55 m2 and a height of 1.34 m, which is larger than the tent used for this study. In the study, white gas produced a mean peak CO level of 61 ppm (95% CI, 31–90) and unleaded gas produced a mean peak CO level of 103 ppm (95% CI, 78–128). Contrasted to this study, stove B and stove C showed CO levels comparable to white and unleaded gas in a 3-season tent. However, stove A did show generally higher levels of CO. The difference in tent size alone is unlikely to account for this difference, given the performance of stove B and stove C. The high power output of stove A is a possible explanation of the variation. However, a direct power output comparison cannot be made, as the manufacturer does not publish the power output of the stove used in the reference study. Extrapolating from the results of this study, isobutane/propane burning stoves would be expected to produce higher CO levels in a 4-season tent than in a 3-season tent. Consistent with both prior investigations, the levels of CO were demonstrated to rise rapidly followed by plateaus within 5 to 6 min, suggesting a steady state between the generation of CO and rate of diffusion through the tent.
Determination of whether the CO levels generated in this study would be potentially toxic to a human exposed to the study environment is necessary to draw conclusions about the use of isobutane/propane stoves inside enclosed spaces. A COHb level of 15% is generally accepted as the level at which most subjects have onset of detectable symptoms, although this level can be variable. 2 Prior work has produced the Coburn-Forster-Kane equation, which is used to estimate (COHb) levels based on an atmospheric CO reading. 7 Using this equation, a COHb level of 15% can be achieved by exposure to 100 ppm CO. 2 The duration of exposure needed to achieve the 15% threshold is variable depending on individual susceptibilities and degree of exertion. Thus, exposure to a CO level greater than 100 ppm creates the potential of developing clinical toxicity. The peak COHb reached depends on a combination of the atmospheric CO level, duration of exposure, and intensity of physical exertion during the exposure. With high atmospheric CO and/or intense physical exertion, a person can develop clinical symptoms even with an exposure of a few minutes. Conversely, lower levels of atmospheric CO can still produce clinical toxicity with a longer exposure. In the United States, the Occupational Health and Safety Administration sets exposure limits when working around CO. Workers cannot be exposed to greater than 50 ppm averaged over an 8-h period, and a concentration of 100 ppm or greater mandates immediate evacuation from the environment. In our study, stove A and stove B reached levels above 100 ppm within 2 and 6 min, respectively. This demonstrates that isobutane/propane canister stoves can rapidly generate levels of CO in excess of 100 ppm and are not safe for use in an enclosed tent. Many factors contribute to the ultimate level of CO accumulation inside an enclosed space. The effect that changing these variables has on CO accumulation was not measured in this study. As a result, while stove C did generate CO levels less than 100 ppm, it cannot be deemed safe to use in a tent based on the results of this study alone.
Limitations
This study was limited by several factors. The study did not include a direct comparison with other fuel types. The permeability of the tent fabric to CO was not measured. The measurements were taken from a single point in the tent, and uniform distribution of CO was not determined. The study was conducted with a single tent over the course of 1 d in stable environmental conditions. The exact tent volume is unknown. Only a single stove of each type was tested. Stove power output was not directly measured but obtained from the manufacturer specifications. Many variables that may contribute to power output fluctuations in real world use were not measured, such as cylinder pressure.
Conclusion
This study demonstrates using isobutane/propane canister stoves in a 3-season tent has the potential to generate CO levels greater than 100 ppm within a few minutes. Use of these stoves in this manner has the potential to cause toxicity and possibly death. Despite differences in study design and fuel type, our results are consistent with prior studies in showing that stove use is not safe in an enclosed space. The use of backpacking stoves should occur in adequately ventilated spaces to minimize the exposure to CO. Future work could attempt to quantify the degree of ventilation required to safely operate stoves in an enclosed space and explore the relationship between stove power output and CO generation.
Footnotes
Acknowledgment
Special thanks to the Buechel Fire Department for use of their facilities and equipment.
Author Contributions: study concept and design (JTB, JTT, MRH); acquisition of the data (JTB, JTT, RL, TE); analysis of the data (JTB, JTT, RL, JS); drafting of the manuscript (JTB, JTT, RL, JS); critical revision of the manuscript (all); approval of final manuscript (all).
Financial/Material Support: None.
Disclosures: None.
