Abstract
Considering the challenges in meeting the high nutritional demand during ultramarathons, the aim of this study was to analyze the nutritional strategies and glycemic response of an athlete with type 1 diabetes (DM1) during participation in a 217-km ultramarathon. A 36-y-old male athlete who was diagnosed with DM1 15 y earlier was studied during participation in the Brazil 135 ultramarathon. Food consumption and blood glucose were recorded during the race, and nutritional intake was calculated after the race. The athlete completed the race in 51 h 18 min. He consumed a total of 15.0 MJ (3593 kcal), 532 g carbohydrate, 166 g protein, 92 g lipid, and 14 L of water during the race. Glycemic values ranged from 3.6 to 18.2 mmol·L-1. Most glycemic values (47%) ranged from 3.9 to 10 mmol·L-1, whereas 5% were <3.9 mmol·L-1, 16% were >10 to 13.9 mmol·L-1, and 32% were >13.9 mmol·L-1. This case report describes the dietary profile of an athlete with DM1 during a 217-km ultramarathon. Although the athlete implemented strategies that differed from those recommended in the literature, food and nutrient intake and the glycemic management strategy adopted allowed him to successfully finish the race. These results suggest that past personal experiences can be considered and that nutritional recommendations for athletes with DM1 should be individualized.
Introduction
Ultraendurance events have become more popular, and participation in ultramarathons has increased worldwide. 1 Driven by the motivation to reach new achievements and limits, ultramarathons have been undertaken by many individuals, including those with type 1 diabetes mellitus. 2 Current exercise management guidelines provide pragmatic recommendations for individuals with type 1 diabetes, such as adjusting insulin doses and increasing carbohydrate intake before, during, or after exercise. 3
Although maintenance of on-target blood glucose levels is challenging for individuals with type 1 diabetes, with adequate strategies of exogenous insulin administration and nutritional intake, it is possible to manage glucose homeostasis and obtain good exercise performance. 4 In addition to daily challenges, maintaining normal glucose levels during different training protocols, especially during prolonged exercise events 4 and traveling, may present a major challenge for athletes with type 1 diabetes, 5 considering that factors other than insulin levels have an impact on glycemia, such as macronutrient intake and psychophysiological stress responses. 4
Some athletes with type 1 diabetes ingest low to moderate levels of carbohydrates during training and rest periods4,6,7; however, this strategy becomes inefficient during long-distance events when high levels of carbohydrate (70–90 g·h-1) are ingested, aiming to prevent hypoglycemia and support performance. 4 When carbohydrate is consumed during exercise, there is an alteration of substrate utilization to energy provision, with a lower contribution of endogenous carbohydrate and a higher rate of exogenous carbohydrate oxidation. 8
Moreover, other macronutrients can also affect blood glucose.9,10 For example, protein intake can also lead to hyperglycemia delayed by gluconeogenesis, together with increased secretion of glucagon. 10 Considering this sustained postprandial hyperglycemic effect of this macronutrient, protein intake associated with carbohydrate after exercise has been considered an interesting strategy for muscle glycogen replacement associated with blood glucose homeostasis. 11 In addition, dietary fat can also affect blood glucose9,10; it contributes to initial hypoglycemia owing to a delay in gastric emptying, and to later hyperglycemia induced by impaired insulin sensitivity and increased hepatic glucose production.9,10
Although the number of athletes with type 1 diabetes who complete ultramarathon races has increased, 4 knowledge about the dietary pattern of these athletes during competition is scarce in the literature. 12 This study aims to analyze the nutritional strategies and glycemic responses of an athlete with type 1 diabetes during participation in a 217-km ultramarathon.
Case Report
A 36-y-old male athlete who was diagnosed with type 1 diabetes 15 y earlier and had 14 y of running training, including participation in 38 marathons (best marathon time: 3 h 38 min) and 4 ultramarathons (100–217 km), was studied during participation in the Brazil 135 Ultramarathon. This athlete was considered well trained for this sport, having performed a weekly training regimen of 40 to 100 km. He had a body mass of 71.1 kg, height of 1.66 m, and a body fat percentage of 10%. Body mass and height measurements were performed with the participant barefooted and wearing light clothing, using a platform scale (Welmy, Sao Paulo, Brazil) to a precision of 0.1 kg and 0.5 cm, respectively. Skinfold thickness (chest, abdominal, and thigh) was measured using a caliper (Sanny, Sao Paulo, Brazil) to a precision of 1 mm, and body fat percentage was calculated by means of standard equations.13,14 The study was approved by the research ethics committee of São Paulo State University (no. 037/2008), and the athlete gave written consent to participate.
In this race, runners cover 217 km and arrive at the finish line within 60 h (Figure 1). During the race, athletes can eat and drink ad libitum. To evaluate food consumption during the ultramarathon, the participant completed a form prepared by the researchers that recorded the time, food consumed, and quantity. With these data, the amounts of energy, carbohydrate, lipid, and protein consumed during the race could be calculated using Avanutri software (version 3.1.1) or from the nutritional information on the product labels.
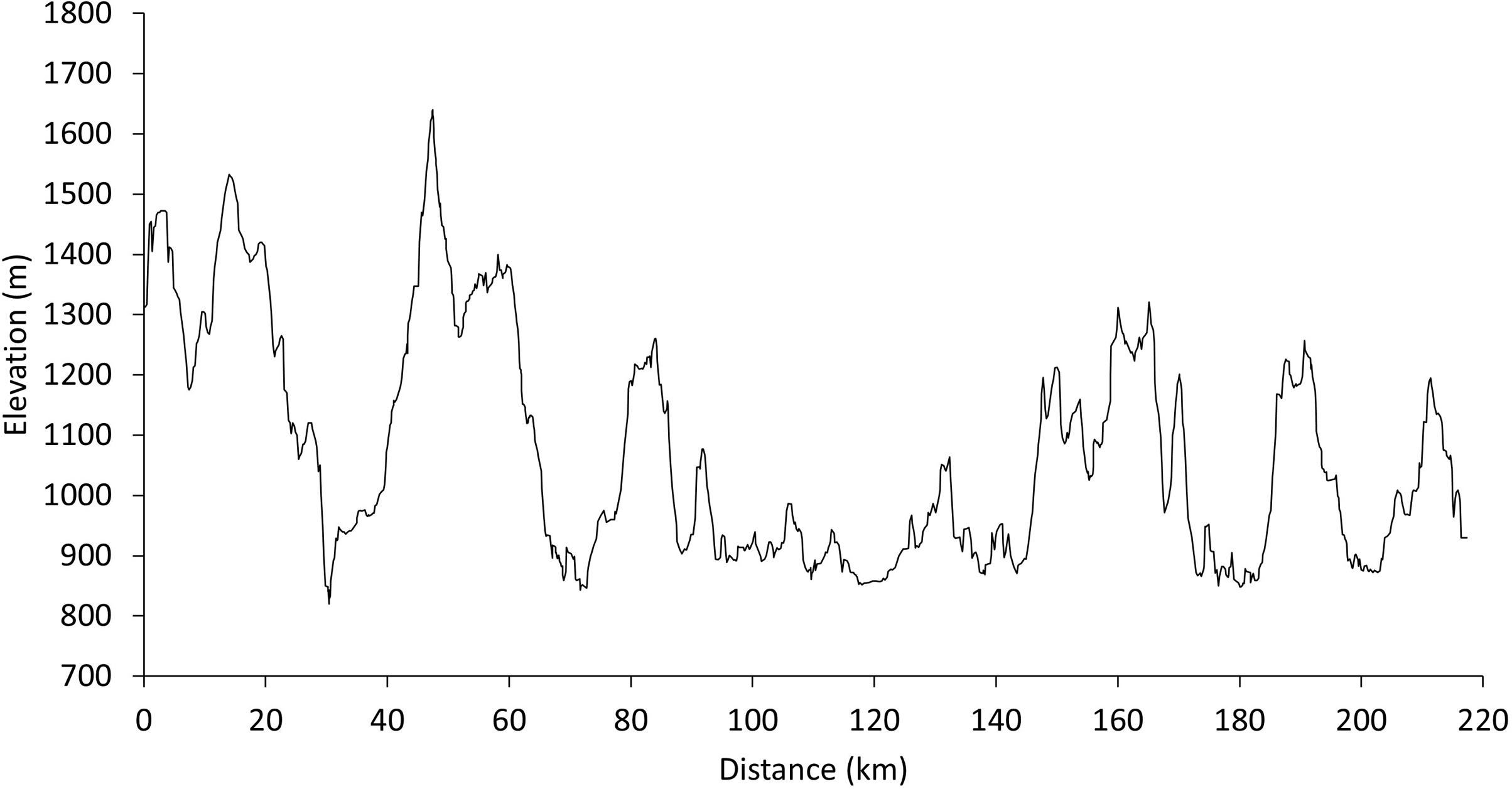
Elevation profile throughout the Brazil 135 Ultramarathon route.
Records of blood glucose levels (Accu-Chek 360 glucose meter, Roche Diagnostics, Sao Paulo, Brazil) and insulin administration were recorded by the athlete during the race. Repeat measures were made of body mass at 78, 175, and 217 km to evaluate hydration status. The athlete was free to sleep whenever he wanted.
Records of split and overall race times were obtained via the official race reports. Thus, we calculated split and overall velocities of the athlete. Moreover, critical velocity was determined 1 wk before the competition. 2 This exercise intensity occurs approximately midway between the anaerobic threshold and maximal oxygen uptake. 15
Table 1 describes the type and amount of food consumed, in addition to the calorie and macronutrient intake, according to the specific race time. During the competition, 14 L (263 mL·h-1) of water was consumed.
Food and nutrient consumption during the race
D1, day 1; D2, day 2; D3, day 3.
As shown in Figure 2, the athlete’s blood glucose ranged from 3.6 to 18.2 mmol·L-1 during the race. Most glycemic values (47%) ranged from 3.9 to 10 mmol·L-1, whereas 5% were <3.9 mmol·L-1, 16% were >10 to 13.9 mmol·L-1, and 32% were >13.9 mmol·L-1. The athlete took a total of 10 U basal insulin (4 U at 2 h postrace and 6 U at 6 h postrace) plus 40 U of bolus insulin (10 U before the race, 10 U at 116 km, and 20 U at 2 h postrace) via insulin pen.
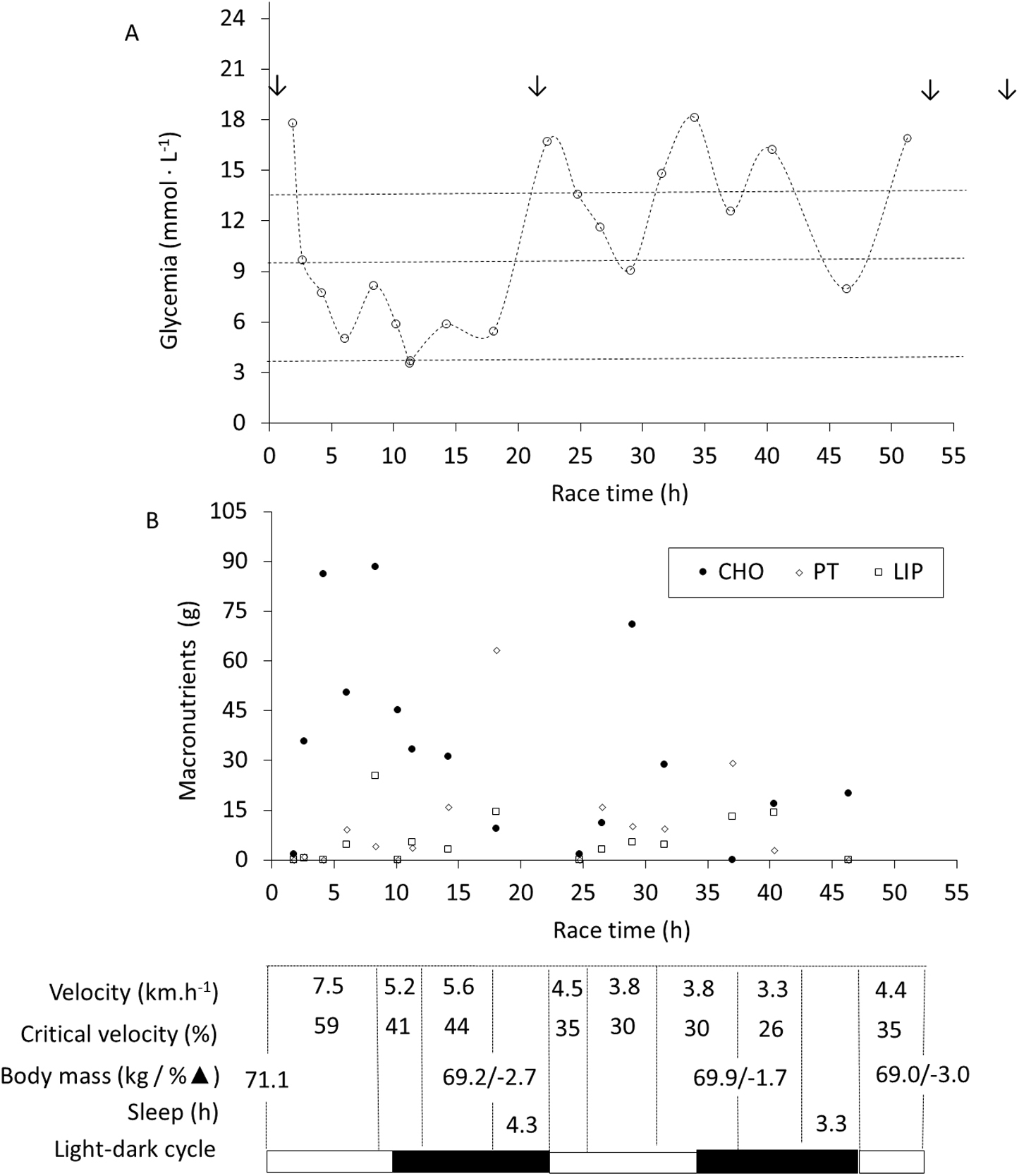
Glycemic response and macronutrients consumed during the race. Dotted lines delimit the blood glucose targets proposed for athletes with type 1 diabetes mellitus during the competition 4 : <3.9 mmol·L-1, 3.9 to 10 mmol·L-1, >10 to 13.9 mmol·L-1, and >13.9 mmol·L-1. Arrows represents bolus insulin infusion. BM, body mass; CHO, carbohydrate; LIP, fat; PT, protein.
Sixty-two athletes participated in the race. Forty-four runners (66% of participants), including our volunteer, completed the competition. Their race time ranged from 27 h and 59 min to 59 h and 23 min. The studied athlete completed the race in 51 h and 18 min (mean of 4.2 km·h-1, 33% of critical velocity) in 27th place. Figure 2 also presents the split velocities, hydration status, and sleep periods of the athlete throughout the race.
Discussion
This case report describes the dietary profile of an athlete with type 1 diabetes mellitus during a 217-km ultramarathon, with a detailed description of the food and drink ingested. Only one other known study 16 has described examples of foods consumed by athletes during an ultramarathon; however, these authors evaluated healthy runners and only reported the usual foods, not how they were eaten during the competition.
The athlete’s nutritional strategies were compared to guidelines for ultraendurance sports, 16 and we can note that our athlete had insufficient energy, carbohydrate, and protein intake. The athlete in this study had nutritional intake similar to that of other triathletes who participated in a competition that lasted an average of 12 h, 17 less than a quarter of the time our athlete took to complete the ultramarathon, suggesting that there is great difficulty in consuming the recommended amounts during the race and in guaranteeing an energy intake compatible with the energy expenditure of the activity. The challenge observed is not only common for people with type 1 diabetes mellitus, but also for ultramarathon athletes in general. 16
Although total energy expenditure (TEE) was not evaluated in this study, authors who evaluated energy demands using doubly labeled water observed that ultramarathon runners have a high energy expenditure in the event, 18 reaching more than 8 times resting metabolism. 18 A study that evaluated athletes during a 161-km ultramarathon (Western States 100, with 25.9±3.0 h [mean±SD] to complete the race), observed that the TEE was 7040±1360 kcal per 12 h, or 587 kcal·h-1. 18 The literature describes that TEE may vary according to several factors, such as body composition, exercise, and environmental conditions, 16 but suggests values ranging from 48 to 72 kcal·km-1 during long races.16,19 According to these data, it is possible to note that the athlete in our study had an energy consumption far below the TEE estimated in the literature (17 kcal·km-1 and 70 kcal·h-1), reinforcing the difficulty of meeting caloric demands compatible with the high demand of the ultramarathon.
By specifically studying athletes with type 1 diabetes mellitus, authors 12 have reported carbohydrate intake rates of 0.4 to 0.6 g carbohydrate·kg-1 body mass·h-1 of exercise in an ultramarathon lasting more than 13 h, which was higher than that consumed by our volunteer.
Fluid consumption was also lower than that recommended for ultraendurance athletes 16 and for athletes with type 1 diabetes mellitus 4 and was below the water intake reported from stable isotope methodologies. 18 Despite this, the variation of –3% body mass observed during the race is compatible with the practical recommendations, 20 which suggest that body mass losses over 2% may be natural and necessary during ultraendurance events to avoid hyperhydration.
The literature 4 proposes glycemic targets for athletes with type 1 diabetes mellitus during competitions. Our results indicate that the athlete spent less time within the range of 3.9 to 10 mmol·L-1 (43%) and more time with his blood glucose above 13.9 mmol·L-1 (32%) than recommended (75 and 5%, respectively). We suspect that the athlete chose to spend more time in the highest targets of glycemia to avoid the negative consequences of hypoglycemia (<3.9 mmol·L-1), which only occurred in 1 glycemia measurement during the competition (∼11 h of race time, at 78 km).
The practice of reducing insulin and consuming additional carbohydrates to avoid hypoglycemia and enhance endurance performance in prolonged exercise has been observed in other athletes with type 1 diabetes mellitus. 8 In addition to managing insulin levels, athletes can manage their blood glucose by manipulating their macronutrient intake. 4 We observed that the increase in blood glucose was caused not only by carbohydrate intake, but also protein consumption. The high amount of this nutrient (around 60 g) consumed at around 18 h of race time and consumption of almost 40 g of protein at 37 h probably resulted in significant and sustained postprandial hyperglycemia. Authors 10 have reported that for individuals with type 1 diabetes mellitus, the addition of ≥28 g of protein to a mixed meal or ingestion of ≥75 g of protein alone could result in a significant and sustained increase in blood glucose in the postprandial period (2–5 h). It is worth mentioning that an insulin application could be a useful tool for adjusting the effects of protein-rich foods.
The impact of fat on blood glucose has also been discussed in the literature, 9 which described no significant effects of high-fat meals in relation to postprandial glycemia, but reported a tendency toward an increase in the long term. 9 We observed no evidence of an impact of fat on postprandial glycemia throughout the race. However, studies indicate that high-fat meals require more insulin than meals with less fat and the same carbohydrate content, supporting the need for alternative insulin dosing algorithms for high-fat meals.9,21
Finally, the athlete slept at 2 time points (after 116 and 195 km, totaling 7.6 h), which occurred between 0300 and 0600 of the 2 race days. Thus, he slept at night, which is the normal time for rest in humans, considering the circadian pressure for sleep, which possibly contributed to the successful performance of the athlete. 22 This pattern has also been observed in other ultramarathon runners. 23
The present case demonstrates that although the athlete with type 1 diabetes mellitus faced the physiological challenges of the ultramarathon and implemented strategies that differed from those recommended in the literature, the insulin management strategy, food and nutrient intake, and exercise and sleep distribution adopted allowed the athlete to successfully finish the race, suggesting that past personal experiences can be considered and that nutritional recommendations for athletes with type 1 diabetes mellitus should be individualized. Continuous glucose monitoring systems may provide a more detailed glycemic response throughout the race, which could constitute a possible limitation of the present investigation. Future studies should focus on this issue.
Footnotes
Acknowledgements
Author Contributions: Study concept and design (LMH, TB); acquisition of the data (LMH, TB); analysis of the data (NES, NVSD, LMH, AME, TB); drafting of the manuscript (NES, NVSD, AME, TB); critical revision of the manuscript (CAG, AME, TB); approval of final manuscript (all authors).
Financial/Material Support: None.
Disclosures: None.
