Abstract
Introduction
Our previous study showed that high-fat diet inhibited the increase in nitric oxide and endothelial nitric oxide synthase expression in the aortic endothelium of rats exposed to hypoxia, and hypoxia plus a high-fat diet led to earlier and more severe vascular endothelial dysfunction (VED) than hypoxia alone. The purpose of the present study was to investigate the effects of L-arginine on high-fat diet–induced VED of rats in hypoxia.
Methods
Forty male Sprague-Dawley rats were randomly divided into 4 groups and treated with hypoxia (H group), hypoxia plus high-fat diet (H+HFD group), hypoxia plus L-arginine (H+L-Arg group), and hypoxia plus high-fat diet and L-arginine (H+HFD+L-Arg group) for 1 wk. Hypoxia was simulated in a hypobaric chamber with an altitude of 5000 m. Aortic morphology and endothelium-dependent vasorelaxation were used to assess VED.
Results
High-fat diet impaired vascular remodeling and reduced endothelium-dependent vasodilator response to acetylcholine in rats exposed to hypoxia, secondary to dysregulation of the nitric oxide pathway. L-arginine supplementation significantly increased plasma nitrates and nitrites and endothelial nitric oxide synthase mRNA levels and improved ultrastructural changes in aortic endothelium and endothelium-dependent vasodilator response.
Conclusions
L-arginine prevents aortic ultrastructural changes and reverses VED induced by high-fat diet in rats exposed to hypoxia, which may have implications for VED induced by high-fat diet in high altitude dwellers.
Keywords
Introduction
Vascular endothelial dysfunction (VED) is characterized by impairment of the integrity of vascular endothelium and endothelium-dependent vasorelaxation. It is the initial event that can lead to the pathogenesis of many cardiovascular diseases, such as atherosclerosis, hypertension, coronary artery disease, and diabetes mellitus. 1 Alterations in the nitric oxide (NO) pathway have been implicated in the pathogenesis of VED. NO is continuously produced by vascular endothelial cells during NO synthase (NOS)-mediated conversion of L-arginine to L-citrulline. NO produced by endothelial NOS (eNOS) is crucial for vascular protection through vasodilatation, antiplatelet, anti-inflammatory, and antiproliferative properties, which help to regulate vascular structure and tone and thus maintain vascular homeostasis. VED is associated with downregulation and inactivation of eNOS, reduced production and bioavailability of NO, and increased generation of reactive oxygen species. 2 -4
The progression of VED can be induced by hypercholesterolemia and hypoxia. 5 Notably, chronic hypoxia can induce hypercholesterolemia, which suggests interactions between these 2 factors for the development of VED. 6 Our previous study showed that high-fat diet inhibited the generation of NO and eNOS mRNA and protein in aortic endothelial tissues of rats exposed to hypoxia, and hypoxia plus high-fat diet led to earlier and more severe VED than hypoxia alone. 7 L-arginine supplementation improved endothelium-dependent vasorelaxation and eNOS production in high-fat diet animal models and humans in normoxia. 8 -10 However, it remains unclear whether L-arginine supplementation can improve VED induced by high-fat diet in hypoxia. To address this problem, the objective of the present study was to examine the effects of oral administration of L-arginine on high-fat diet-induced VED of rats in hypoxia.
Methods
Animals
The protocols in this study were approved by the medical ethics committee of Qinghai University Medical College. Male Sprague-Dawley rats aged 4 to 6 wk (n=40) were purchased from Xi’an Jiaotong University (altitude of 400 m) and randomly divided into 4 equal groups: hypoxia (H) group, hypoxia plus high-fat diet (H+HFD) group, hypoxia plus L-arginine (H+L-arg) group, and hypoxia plus high-fat diet and L-arginine (H+HFD+L-arg) group. All rats were housed for 1 wk under a normal light-dark cycle in a hypobaric hypoxic chamber with a simulated altitude of 5000 m in our laboratory in Xining, China. The rats in H, H+HFD, H+L-arg, and H+HFD+L-arg groups were gavaged with saline solution 10 mL·kg-1, fat emulsion 10 mL·kg-1, saline solution 10 mL·kg-1, and fat emulsion 10 mL·kg-1 at 0800, respectively; they were then gavaged with saline solution 10 mL·kg-1, saline solution 10 mL·kg-1, L-arginine solution 50 mg·kg-1, and L-arginine solution 50 mg·kg-1 at 2000 every day, respectively. High-fat emulsion was prepared as previously reported. 11 Body weight was recorded.
Rats were anesthetized with urethane for the collection of blood from the abdominal aorta. Plasma malondialdehyde (MDA) level was measured using the thiobarbituric acid method. Plasma superoxide dismutase (SOD) activity was measured using the water-soluble tetrazolium salts method. Plasma NO was estimated by measuring the stable metabolite nitrates and nitrites (NOx) with the nitrate reductase method. The MDA, SOD, and NOx kits were provided by Nanjing Jiancheng Bio-Engineering Research Institute, China. Plasma triglyceride, total cholesterol (TCH), high-density lipoprotein, and low-density lipoprotein (LDL) levels were measured using the photoelectric colorimetric method (Aeroset-2000, Instrument Laboratory).
Isolation of Aortas
Rats treated for 1 wk were anesthetized and euthanized. Their aortas were removed and placed in ice-cold Krebs-Henseleit buffer consisting of NaCl (118.29 mM), KCl (4.69 mM), MgSO4 (2.39 mM), KH2PO4 (1.19 mM), NaHCO3 (25 mM), CaCl2 (2.52 mM), and glucose (12.11 mM). Connective tissues were carefully removed, and vessels were cut into rings of 5 mm in length for measurement of vascular tone.
Aortic rings were suspended on a pair of steel hooks in an organ bath connected to an isometric tension transducer (Multi Wire Myograph, Model MP150, BIOPAC System). The organ bath was filled with Krebs-Henseleit buffer (37°C, pH 7.4) and bubbled with a mixture of 5% CO2 and 95% O2. The aortic rings were allowed to equilibrate at a rest tension of 1 g for 60 min. To measure the viability of these aortic rings, phenylephrine (Phe; 10−6 M) was added to the bath and washed 3 times to make the tension to return to the baseline level. The aortic rings were contracted with Phe (10−6 M). When the constriction reached a plateau, acetylcholine (ACh, 10−5 M) or sodium nitroprusside (SNP, 10−5 M) (Sigma Aldrich Inc) was added to assess endothelium-dependent or endothelium-independent relaxation responses. The endothelium-dependent relaxation response to ACh was assessed in the presence or absence of Nω-nitro-L-arginine methyl ester (L-NAME; an NO synthase inhibitor; 5×10−6 M; pre-incubated for 20 min). The aortic relaxation was expressed as the change in tension with ACh or SNP relative to the maximal Phe contraction.
Transmission Electron Microscopy
Aorta tissue was fixed in 2.5% glutaraldehyde, dehydrated, and embedded in epoxy resin Epon812. The block was sliced to a thickness of 1 to 2 μm and stained with methylene blue, and then the sections were sliced to a thickness of 50 to 70 nm and stained with uranyl acetate and lead citrate. Aortic morphology was observed using a transmission electron microscope (HITACHI, Ibaraki prefecture, Japan).
Quantitative Real-Time Polymerase Chain Reaction Analysis of no Synthase
The total RNA of aortas was extracted by trizol (Takara Biotechnology, Dalian, China), and 1 microgram of total RNA was reverse transcribed to cDNA using random primers (Takara Biotechnology, Dalian, China). Real-time polymerase chain reaction was performed in 20 μL of reaction system consisting of 10 μL of premix, 1 μL of cDNA, (0.4 μL, 0.8 μL, and 1.0 μL of each primer for eNOS, inducible NOS [iNOS], and neuronal NOS [nNOS], respectively) and RNase-free water. The cycling conditions of eNOS were as follows: holding stage: 95°C for 90 s; cycling stage: 95°C 5 s, 60°C for 30 s, and 72°C for 60 s. The cycling conditions of nNOS were as follows: holding stage: 95°C for 120 s; cycling stage: 95°C for 10 s, 56°C for 30 s, and 72°C for 60 s. The cycling conditions of iNOS were as follows: holding stage: 95°C for 120 s; cycling stage: 95°C for 10 s, 62°C for 30 s, and 72°C for 60 s. Forward and reverse primers were as follows: eNOS 12 : 5′- CTACCGGGACGAGGTACTGG -3′ and 5′- GGAAAAGGCGGTGAGGACTT -3′; iNOS: 5′- TCTTTGCTTCTGTGCTAATG-3′ and 5′- CAGTAGTTGTTCCTCTTCCA-3′; nNOS: 5′- GGAATATGAGGAGTGGAAGT-3′ and 5′- CTGAGTGAGGAGAAGTGTAG-3′; β-actin 13 : 5′- AGTGTGACGTTGACATCCGT -3′ and 5′- GACTCATCGTACTCCTGCTT -3′. Real-time polymerase chain reaction results were analyzed by the 2-△△Ct method.
Statistical Analysis
Values were expressed as mean±SD and analyzed using SPSS 17.0 statistical analysis package (SPSS Inc., Chicago, IL). One-way analysis of variation followed by post hoc test was performed for multiple comparisons. P<0.05 was considered statistically significant.
Results
Effects of Treatment on Body Weight
There was no significant difference in body weight at the beginning of the study. However, body weight decreased in rats treated with hypoxia and high-fat diet compared to those treated with hypoxia alone. A further decrease in body weight was observed in the H+HFD+L-arg group. Similarly, body weight decreased in the H+L-arg group compared to the H group (Figure 1A).
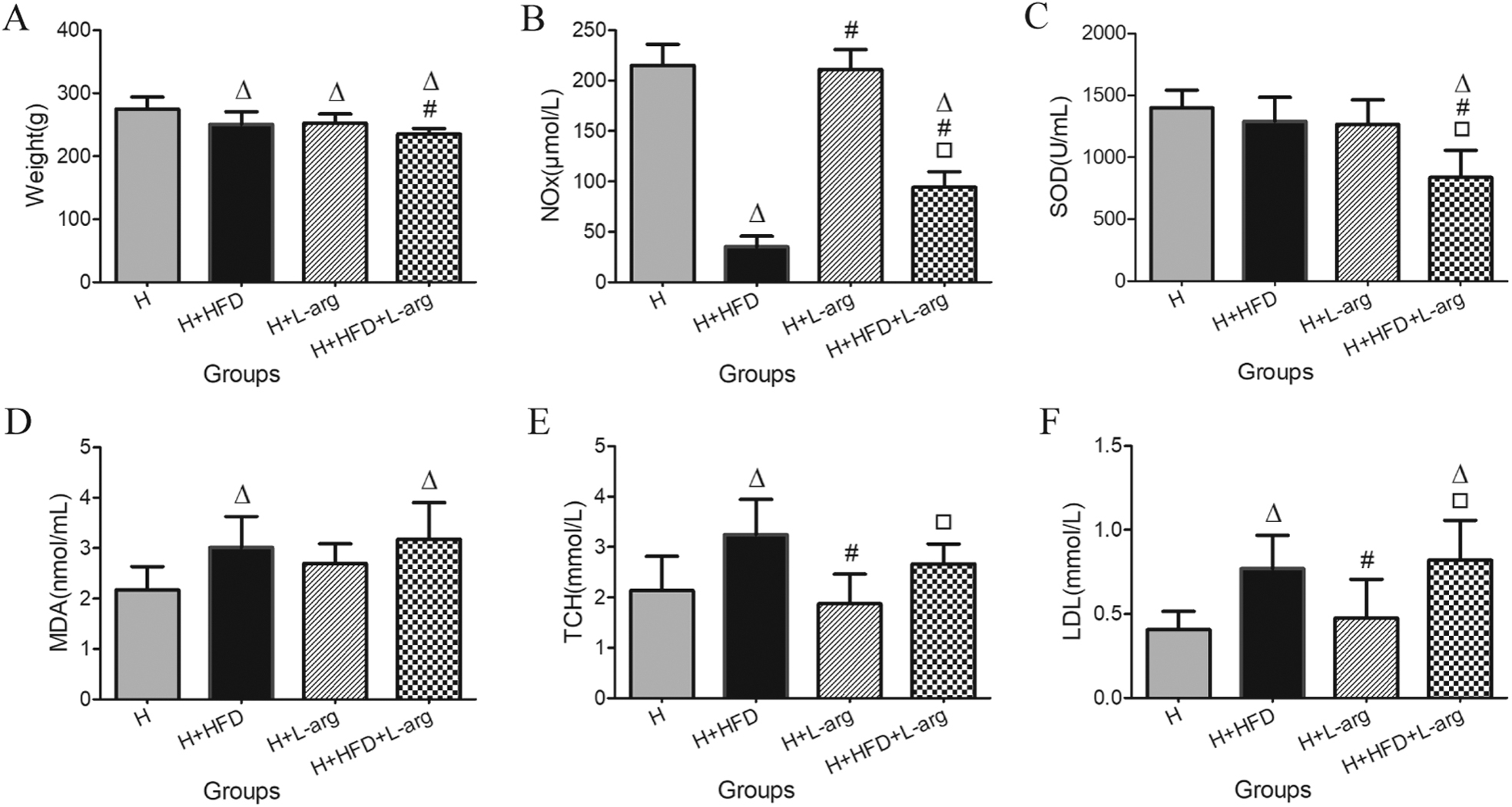
Changes in body weight (A), plasma levels of metabolic nitrates and nitrites (NOx) (B), superoxide dismutase (C), malondialdehyde (D), total cholesterol (E), and low density lipoprotein (F), in hypoxia (H) group, hypoxia plus high-fat diet (H+HFD) group, hypoxia plus L-arginine (H+L-arg) group, and hypoxia plus high-fat diet and L-arginine (H+HFD+L-arg) group. Values represent means±SD (n=8. △P<0.05 vs H group, #P<0.05 vs H+HFD group, □P<0.05 vs H+L-arg group).
Effects of Treatment on Plasma NOx, SOD, and MDA Levels
Plasma NOx level was significantly decreased in rats exposed to hypoxia and high-fat diet compared to the H group, suggesting an inability to compensate. However, it was significantly higher in the H+HFD+L-arg group than in the H+HFD group (Figure 1B). Compared to the H group, plasma MDA levels increased significantly in rats exposed to hypoxia and high-fat diet. However, there was no difference between H+HFD and H+HFD+L-arg groups (Figure 1D). Plasma SOD level did not differ among H, H+HFD and H+L-arg groups, but it was significantly lower in the H+HFD+L-arg group than in other groups (Figure 1C).
Effects of Treatment on Blood Lipids
Both TCH and LDL levels were significantly increased in rats exposed to hypoxia and high-fat diet compared to those exposed to hypoxia alone, but L-arginine supplementation had no effect on the TCH and LDL levels. The triglyceride and high-density lipoprotein levels were similar among the 4 groups (Figure 1E and F).
Ultrastructural Changes of Aortas
For the H group, endothelial cells were flattened, with loose connections to internal elastic lamina. Mitochondria, rough endoplasmic reticulum, microfilaments, and vesicles were also observed in the cytoplasm. Some endothelial cells were swollen with a decreased electron density, and internal elastic lamina was interrupted. Smooth muscle cells were irregular in shape with abundant mitochondria in the cytoplasm (Figure 2A1, 2A2, and 2A3). For the H+HFD group, cuboidal endothelial cells, endothelial denudation, swollen mitochondria, expanded rough endoplasmic reticulum, and few lipid droplets were observed. Connective structures were not observed, and internal elastic lamina was interrupted. Smooth muscle cells were irregular in shape with more lipid droplets and mitochondria in the cytoplasm, suggesting that high-fat diet may aggravate the impairment of aortic structure in rats exposed to hypoxia (Figure 2B1, 2B2, and 2B3). For the H+L-arg group, some swollen endothelial cells were observed, with loose connections to internal elastic lamina. The mitochondria were mildly swollen, and smooth muscle cells remained intact (Figure 2C1, 2C2, and 2C3). For the H+HFD+L-arg group, few swollen endothelial cells with loose connections to internal elastic lamina and swollen mitochondria were found, but most endothelial cells were flattened; tight junctions and gap junctions were seen between neighboring cells. Smooth muscle cells remained intact. Swollen mitochondria were observed, but no lipid droplets were observed (Figure 2D1, 2D2, and 2D3). L-arginine supplementation improved aortic alterations induced by high-fat diet in hypoxia.
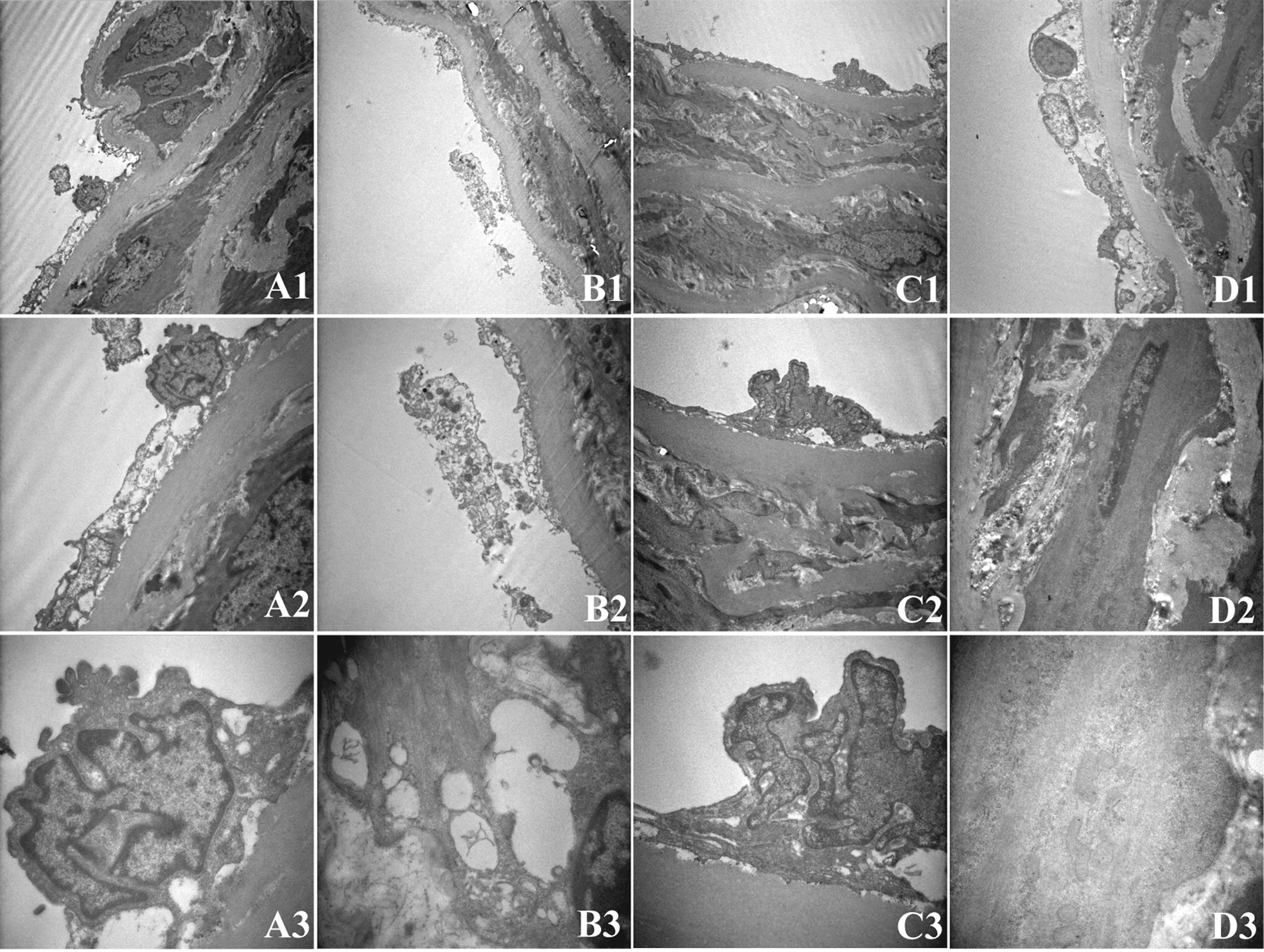
Transmission electron microscope images of aortic sections in rats of hypoxia (H) group (A), hypoxia plus high-fat diet (H+HFD) group (B), hypoxia plus L-arginine (H+L-arg) group (C), and hypoxia plus high-fat diet and L-arginine (H+HFD+L-arg) group (D) (A1, B1, C1, D1, ×4000; A2, B2, C2, D2, ×10000; A3, B3, C3, D3, ×30000).
Relaxation and Contraction in Aortas
All groups showed similar contraction of aortic rings in response to Phe (Figure 3A) and endothelium-independent relaxation in response to the NO donor SNP (Figure 3B). However, compared to the H group, endothelium-dependent relaxation in response to ACh was significantly decreased in the H+HFD group but significantly increased in the H+HFD+L-arg group (Figure 3C). ACh-mediated vasorelaxation was fully inhibited in all groups pre-incubated with the NOS inhibitor L-NAME, suggesting that the vasorelaxation in response to ACh was NO dependent.
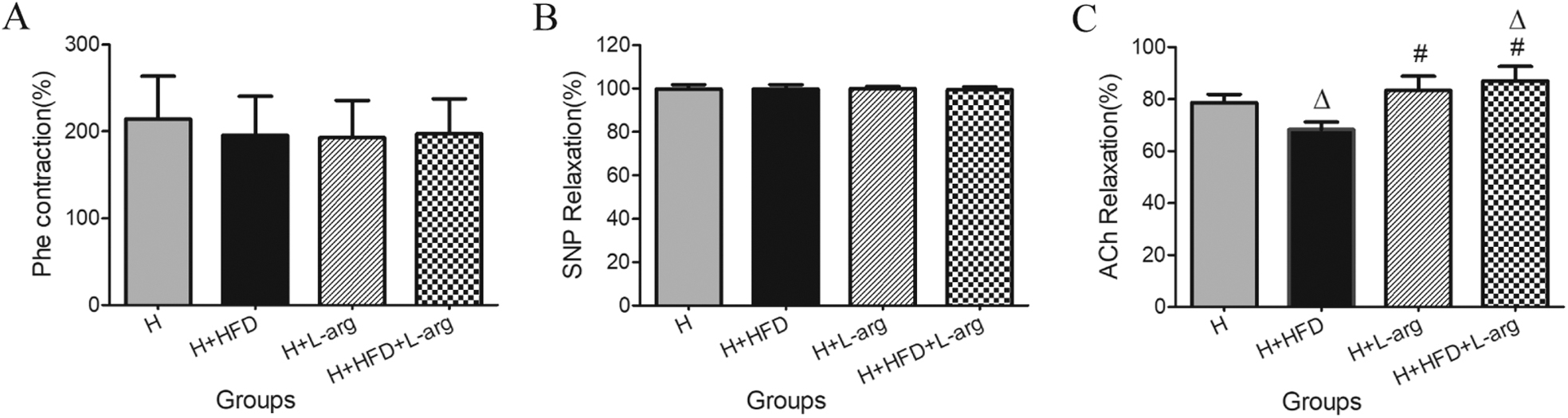
The effects of SNP and ACh on endothelium-independent relaxations and endothelium-dependent relaxations and Phe on contraction in aortic rings of rats in hypoxia (H) group, hypoxia plus high-fat diet (H+HFD) group, hypoxia plus L-arginine (H+L-arg) group, and hypoxia plus high-fat diet and L-arginine (H+HFD+L-arg) group. (A) Contraction response of aortic rings to Phe (10-6 M). (B) Endothelium-independent relaxation response of aortic rings to SNP (10-5M). (C) Endothelium-dependent relaxation response of aortic rings to ACh (10-5M). (n=6 animals/group). Values represent mean±SD (△P<0.05 vs H group, #P<0.05 vs H+HFD group).
NOS m RNA Levels
The eNOS mRNA levels were significantly lower in the H+HFD group compared to the H group, but there was no significant difference between H+HFD+L-arg and H groups (Figure 4A). The transcript expression levels of iNOS and nNOS were similar among the 4 groups (Figure 4B and C).
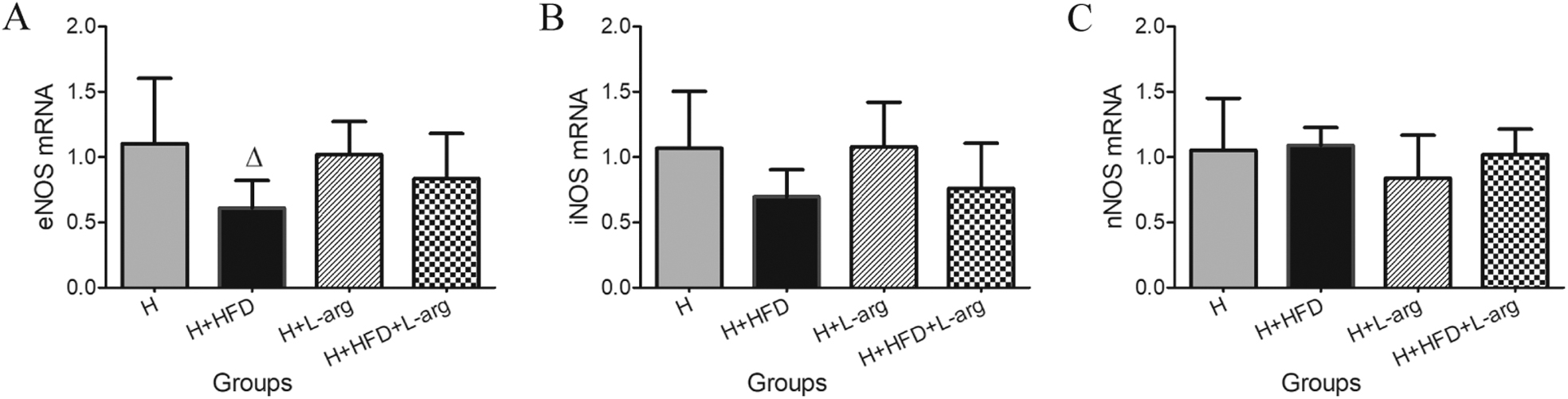
Expression levels of aortic eNOS mRNA (A), iNOS mRNA (B), and nNOS mRNA (C) in rats in hypoxia (H) group, hypoxia plus high-fat diet (H+HFD) group, hypoxia plus L-arginine (H+L-arg) group, and hypoxia plus high-fat diet and L-arginine (H+HFD+L-arg) group. Values represent mean±SD (n=5 animals/group. △P<0.05 vs H group).
Discussion
The purpose of this study was to investigate the effects of L-arginine on high-fat diet-induced VED of rats in hypoxia. We found that high-fat diet impaired vascular remodeling and reduced endothelium-dependent vasodilator response to ACh in rats exposed to hypoxia for 1 wk. L-arginine supplementation significantly increased plasma NOx levels and eNOS mRNA expression and improved ultrastructural changes in aortic endothelium and endothelium-dependent vasodilator response in rats exposed to hypoxia and high-fat diet.
The transmission electron microscope results revealed obvious impairment in the wall of the aorta in rats exposed to hypoxia and high-fat diet, which was characterized by endothelial morphological changes and denudation, absence of intercellular junctions, interrupted internal elastic lamina, morphological changes of smooth muscle cells, and more lipid droplets in endothelial cells and smooth muscle cells (Figure 2), suggesting that high-fat diet exacerbated aortic structure impairment in rats exposed to hypoxia. 7 L-arginine supplementation significantly improved morphological damage in hypoxia with normal or high-fat diet, and most endothelial cells and smooth muscle cells showed normal morphology and structure with intercellular junctions and without lipid droplets. 9 Thus, in hypoxia, L-arginine supplementation protected endothelial morphology and structure against impairment induced by high-fat diet.
Similarly, changes in vascular endothelial function were also observed. In hypoxia, high-fat diet induced severe VED. However, L-arginine supplementation improved this VED by increasing the endothelium-dependent relaxation to ACh (Figure 3). Ach-induced endothelium-dependent vasorelaxation was completely suppressed by the NOS inhibitor L-NAME, indicating that Ach-induced aortic relaxation in rats was dominated by NO. This finding was in agreement with our earlier study and the study by Balarini et al. 14 This is consistent with the changes we found in plasma NOx, which likely accounts for the endothelium-dependent relaxation to ACh. In summary, high-fat diet significantly decreased plasma NOx levels in hypoxia, and L-arginine supplementation prevented these changes and improved endothelium-dependent relaxation, similar to results in nomoxia.9,15
The NO-dependent mechanism in adaptive defense plays a key role in adaptation to hypoxia and prevention of cardiovascular diseases. Higher circulating NO products and NO-associated high blood flow via endothelium-dependent relaxation can offset high altitude hypoxia in Tibetans and lowlanders or experimental animals exposed to hypoxia. 16 -18 The same results were found in our study. The levels of plasma NOx increased significantly in hypoxia compared to those in normoxia. High-fat diet caused inability to increase plasma NO levels and induced VED in hypoxia.7,18 L-arginine supplementation increased plasma NOx levels and endothelium-dependent relaxation and thus could offset the impairment induced by high-fat diet in hypoxia (Figures 1B and 3C), which might be beneficial for individuals with a high-fat diet in adapting to high altitude hypoxia and preventing VED.
Adaptive increases in eNOS were found in hypoxia compared to normoxia. 7 High-fat diet caused significant downregulation of eNOS but no changes in iNOS and nNOS in rats exposed to hypoxia, resulting in impairment of NOx production and VED. However, these alternations could be reversed by L-arginine supplementation, which resulted in upregulation of eNOS and increased availability of substrate to NOS (L-arginine); consequently, an increase in plasma NO levels and improvement of the aortic endothelial structure and relaxation were observed.
High-fat diet induced alterations in rats exposed to hypoxia could be caused by a variety of factors. The present study showed that high-fat diet increased plasma TCH, LDL, and MDA levels in rats exposed to hypoxia, resulting in an increase in oxygen free radicals by lipid peroxidation and subsequently eNOS dissociation, eNOS uncoupling, decreasing generation and increasing inactivation of NO, and VED. 19 Lipid droplets were also observed in endothelial cells and smooth muscle cells in rats exposed to hypoxia and high-fat diet, which may be associated with VED and the early impairment of atherosclerosis as a direct risk factor. Previous studies have shown that L-arginine supplementation normalized lipid and lipoprotein levels and subsequently increased serum NO levels and improved the histological and ultrastructural profile of aortas in normoxic exposed rats with high-fat diet.9,20 The present results showed that L-arginine supplementation improved aortic endothelial structure in rats exposed to hypoxia and high-fat diet without significantly changing lipid and lipoprotein and MDA levels. The inconsistent results may be associated with the difference between normoxia and hypoxia and the dose and duration of L-arginine supplementation.
The present study has several limitations, including incomplete assessment of soluble factors secreted by vascular endothelium, such as intercellular adhesion molecule and vascular cell adhesion molecule. In addition to the lack of normoxic controls, we were unable to investigate the effects of L-arginine on high-fat diet induced VED of rats in normoxia simultaneously; thus, we were unable to evaluate the differences in effect between normoxia and hypoxia.
Conclusions
Through the NO pathway, L-arginine supplementation improves aortic ultrastructural changes and reverses VED induced by high-fat diet in rats exposed to hypoxia. Combined with our previous study, the reduced NO bioavailability accounting for VED induced by high-fat diet and hypoxia resulted mainly from depressed L-arginine utilization and/or bioavailability. Thus, L-arginine supplementation may be effective in preventing high altitude VED. These findings have possible implications for VED induced by high-fat diet in high altitude dwellers.
Footnotes
Acknowledgements
Author Contributions: Study design (ZYX, WP, TL); data acquisition and analysis (ZGM, ZXH, SYP, GQ); manuscript preparation and review (ZYX, LY, LDX); final approval of manuscript (ZYX, TL, ZGM, ZXH, SYP, LY, LDX, GQ, WP).
Financial/Material Support: None.
Disclosures: None.
Funding
Acknowledgments: This study is supported by the National Natural Science Foundation of China (No.81760823) and Science and technology project of Qinghai province (2016-ZJ-951Q).
