Abstract
Introduction
Cold-induced vasodilation (CIVD) is seen in the extremities during exposure to cold. A strong vasodilation response has been associated with a decreased risk of cold injury. Increasing CIVD might further decrease this risk. The calcium-channel blocker nifedipine causes vasodilation and is used to treat Raynaud’s syndrome and chilblains. Nifedipine is also used for high altitude pulmonary edema and could potentially serve a dual purpose in preventing frostbite. The effects of nifedipine on CIVD have not been studied.
Methods
A double-blind crossover study comparing nifedipine (30 mg SR (sustained release) orally twice daily) to placebo was designed using 2 sessions of 4 finger immersion in 5°C water, with 24 h of medication pretreatment before each session. Finger temperatures were measured via nailbed thermocouples. The primary outcome was mean finger temperature; secondary outcomes were mean apex and nadir temperatures, first apex and nadir temperatures, subjective pain ranking, and time of vasodilation onset (all presented as mean±SD).
Results
Twelve volunteers (age 29±3 [24–34] y) completed the study. No significant difference in finger temperature (9.2±1.1°C nifedipine vs 9.0±0.7°C placebo, P=0.38) or any secondary outcome was found. Pain levels were similar (2.8±1.6 nifedipine vs 3.0±1.5 placebo, P=0.32). The most common adverse event was headache (32% of nifedipine trials vs 8% placebo).
Conclusions
Pretreatment with 30 mg of oral nifedipine twice daily does not affect the CIVD response in healthy individuals under cold stress.
Introduction
Cold injury (eg, frostbite, frostnip, and chilblains) poses a significant risk to individuals working, living, and recreating in cold environments. Cold-induced vasodilation (CIVD) refers to the intermittent dilation of blood vessels in the extremities during exposure to cold, a physiologic mechanism that is hypothesized to affect an individual’s susceptibility to cold injury. A vigorous vasodilation response has been associated with a decreased rate of cold injury, and a poor response (ie, decreased degree or delayed onset of vasodilation) has been associated with an increased rate of injury. 1
Prior research has described the parameters of the CIVD response in a variety of experimental conditions, most commonly involving submersion of the fingers in a cold-water bath under controlled conditions. These studies have shown that the response has substantial individual and even digital variation and is affected by an individual’s core temperature. 2 –6 Several researchers have investigated the possibility of augmenting CIVD through various training regimens with the goal of preventing cold injury. Specifically, altitude acclimatization and exercise training have been shown to increase CIVD (ie, increase the amount of vasodilation),7,8 and a structured program of cold exposure has been shown to decrease CIVD. 9 No prior studies have investigated the use of a pharmacologic agent to augment the response.
Nifedipine causes peripheral vasodilation and is routinely used for the treatment of peripheral vasoconstrictive diseases such as Raynaud’s syndrome and chilblains. This suggests that nifedipine might affect the CIVD response and thus might be useful as a prophylactic measure in persons exposed to intense but limited periods of cold exposure (eg, expeditions, work assignments, or military deployments). The study of the peripheral effects of nifedipine in cold conditions has an additional benefit: Nifedipine is used for the treatment of high altitude pulmonary edema in persons traveling to high altitudes. The risk of frostbite at these altitudes is generally increased, and the effects of nifedipine on frostbite risk are not known.
This study investigated the effect of calcium-channel blockade with nifedipine on CIVD in the fingers of healthy individuals under experimental cold stress. It was hypothesized that nifedipine pretreatment would increase the CIVD response.
Methods
This study protocol was approved by the University of Utah institutional review board. An investigational new drug application regarding nifedipine use in cold injury research was submitted to the Food and Drug Administration and received a pre-investigational new drug exemption from that body.
Participants were recruited via the use of printed flyers and a call for participation through institutional email at the University of Utah School of Medicine. Volunteers between the ages of 18 and 65 y were considered for inclusion. Exclusion criteria were a history of Raynaud’s syndrome, current use of vasodilatory antihypertensives, and pregnancy.
Study Design
The magnitude of CIVD has been shown to vary significantly between individuals and even between different digits of the same individual. 2 Therefore, this study was designed using a double-blind crossover model to minimize the effect of individual variability in CIVD and tested multiple digits from each individual. Each participant completed 2 cold stress sessions as described later.
For the 24-h period before each session, participants received either nifedipine at a dose of 30 mg SR (sustained release) twice daily or placebo. Sessions were separated by a minimum of 1 wk to allow for washout of the drug. Participants were randomized to placebo-first or nifedipine-first groups by the staff of the investigational drug pharmacy at the University of Utah. Both nifedipine and placebo were identically encapsulated at the pharmacy before their delivery to the investigational team.
Cold Stress Protocol
Each cold stress session consisted of a warm-water standardization period and a cold-water stress period. The nondominant hand was used. Before immersion, wire thermocouples (Omega Engineering, Inc., Norwalk, CT) were affixed to digits 2 to 5 of the hand along the edge of the nailbeds using waterproof tape. These thermocouples were monitored using a USB interface (Phidgets, Inc., Calgary, AB, Canada) connected to a laptop computer. A fifth thermocouple was placed in the immersion basin to monitor water temperature. A SpotOn core temperature monitoring device (3M, Inc., Maplewood, MN) was affixed to the forehead to estimate core temperature. Cold stress sessions were carried out in an indoor climate-controlled room at 21°C with no perceptible wind at the University of Utah, at an altitude of 1505 m (4938 ft).
During the standardization period, fingers 2 to 5 of each participant’s hand were immersed up to the metacarpophalangeal joint in a basin of water at 35°C for 5 min. This was done to ensure a standard skin temperature before cold exposure. After this warm water exposure period, fingers 2 to 5 of each participant’s hand were immersed to the same level in a separate basin of water at 5°C for 30 min. Cold water was circulated via pump from an ice water reservoir as needed to maintain the basin temperature at 5°C (Figure 1). The 2 sessions for each participant were carried out during the same time of day (morning or afternoon). Before each cold stress session, participants were asked verbally about any side effects they experienced while taking the study medication.

Cold stress apparatus.
Temperature data from the thermocouples were recorded via Max software (Cycling ’74, Inc., San Francisco, CA), used to monitor the Phidgets interface. Temperature data from the fingers and water bath were recorded every 2 s. The core temperature and pain rating were recorded every 5 min.
The primary outcome of this study was mean finger temperature during the period of cold-water immersion. Secondary outcomes were the first nadir and apex skin temperatures, average apex and nadir temperatures, onset of vasodilation, and the average pain rating on a scale from 1 to 10 during cold water immersion.
Statistical Analysis
Effect sizes were estimated using the cross-over design module in PASS v. 11, based on the within-subject SD for the mean cold-water immersion temperature (SD=0.7) seen in prior research. 10 Twelve participants (24 measures) would achieve 80% power at a 0.05 significance level to detect an average temperature difference of 0.8°C or greater between groups. Data were analyzed using Microsoft Excel 365 (Microsoft Corporation, Redmond, WA). Relative maxima and minima and average temperature were calculated individually for each finger; these were then averaged to yield a single value of each outcome measure for each trial session. Average values were compared between the placebo and nifedipine conditions. Paired t-tests were used to determine statistical significance. The standard deviation of the change in mean finger temperature was calculated for comparison to prior research, and correlation between the 2 test conditions was examined. Average water temperature, pain ranking, and core temperature were also compared between the trial conditions using paired t-tests. Data are presented as mean±SD, with range as appropriate.
Results
Twelve volunteers (age 29±3 [24–34] y) completed the study. Nine volunteers were male and 3 were female. None reported any prior cold injuries. None dropped out due to adverse effects related to the medication or intolerable discomfort during the cold stress sessions. All participants completed both trial sessions.
No significant differences in mean finger temperature (9.2±1.1°C nifedipine vs 9.0±0.7°C placebo, P=0.38, SDCHANGE = 0.9, r = 0.58) or apex/nadir temperature were found (Figure 2). There was no significant difference in the time of response onset (5.4±1.2 min nifedipine vs 6.4±2.3 min placebo, P=0.16). Reported pain levels were similar between the 2 trial conditions (2.8±1.6 nifedipine vs 3.0±1.5 placebo, P=0.32).
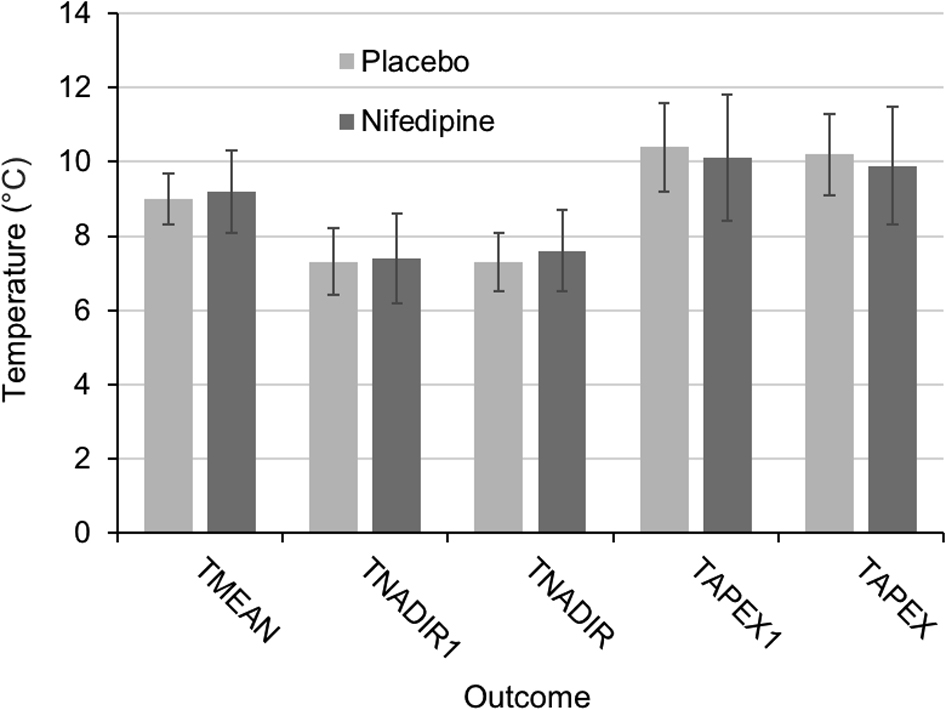
Temperature outcome results (mean±SD). Differences between means are nonsignificant throughout.
The most common adverse event was headache (32% of nifedipine trials vs 8% placebo). Other reported adverse events included lightheadedness, paresthesia, and fatigue (Table 1). No dangerous adverse events were observed.
Number of participants reporting adverse effects
No significant difference in water temperature (5.8±0.4°C nifedipine vs 5.6±0.4°C placebo, P=0.25) or core temperature (36.6±0.5°C nifedipine vs 36.7±0.4°C placebo, P=0.60) was observed between the trial conditions.
Three of the 12 participants showed a pronounced decrease in the time of onset of vasodilation with nifedipine treatment (5.2±0.7 min nifedipine vs 9.9±0.9 min placebo), whereas the remainder of the participants showed little change (5.5±1.3 min nifedipine vs 5.3±1.0 min placebo). These 3 participants had a delayed onset of vasodilation at baseline (ie, with placebo treatment) compared to the remainder (9.9±0.8 vs 5.3±1.0 min).
Discussion
This study showed no effect of nifedipine pretreatment on the parameters of the CIVD response, including mean finger temperature during cold stress and a number of secondary CIVD parameters that have been reported in prior studies. Nifedipine pretreatment also had no effect on the pain induced by cold stress. These results do not support a role for nifedipine pretreatment in the prevention of cold injury. However, this study does suggest that nifedipine may be taken in cold environments (eg, in high altitude pulmonary edema treatment) without adversely affecting the normal CIVD response.
This study was adequately powered to detect a mean temperature difference of 0.8°C between groups, and any treatment effect smaller than this threshold is unlikely to be clinically significant. Regarding dosage, nifedipine is used up to doses of 120 mg·d-1 for the treatment of Raynaud’s syndrome, and it is reasonable to believe that a higher dose would have a stronger vasodilatory effect. However, the effectiveness of higher doses in healthy individuals under cold stress is unknown. Furthermore, the use of a higher dose would likely lead to an increased rate of side effects, limiting the utility of this approach. An increased prevalence of headache was seen with nifedipine use in this study; this is not surprising because it is a common side effect of the medication.
Although this study demonstrated no effect of nifedipine pretreatment in healthy individuals, further investigation in those with an impaired vasoactive response may be warranted. Prior research in patients with Raynaud’s syndrome has demonstrated improved finger temperatures (although not CIVD specifically) with nifedipine at a dose of 20 mg 3 times per day under conditions of cold stress,11,12 and nifedipine might be useful to prevent cold injury in this population. Furthermore, in the current study a subgroup of patients with delayed CIVD onset experienced substantial improvement in onset times after nifedipine treatment. This suggests that those with a decreased onset of CIVD at baseline but without features of Raynaud’s syndrome might experience a benefit from the medication. However, the participant numbers in this group were too small for any specific conclusions to be drawn.
The physiologic reasons for the lack of a treatment effect in this study remain unclear. Nifedipine, as an L-type calcium channel blocker, acts directly on arterial smooth muscle to counteract the vasoconstrictive effect of adrenergic stimulation in conditions of cold stress. However, arteriovenous anastomoses in the fingers are thought to play a significant role in CIVD, and nifedipine might be less active at these sites than at arteries and arterioles. Although the results of this study and prior research in patients with Raynaud’s syndrome might seem to conflict, persons with Raynaud’s syndrome have pathologic arterial constriction in cold environments compared to those without the disease. 13 For this reason, nifedipine might be expected to have a larger effect on arterial blood flow and overall finger temperature in the population with Raynaud’s syndrome. This effect may or may not involve CIVD. Further research would be necessary to test these hypotheses and to better characterize the CIVD response in patients with Raynaud’s syndrome.
The CIVD response remains an intriguing area for investigation in cold injury research. Further preventative trials could investigate direct measurements of blood flow such as Doppler ultrasonography, or the use of topical rather than oral therapies. In addition, future research could explore the effect of vasodilatory therapies in those individuals with a delayed onset or decreased degree of CIVD at baseline; this group might be particularly prone to cold injury and benefit more from preventative therapies than the general population.
Limitations
The design of this study did not control for several potential confounders, such as the hormonal phase of female participants and the clothing worn during cold stress sessions. Cold stress was limited to 1 hand; although this methodology is similar to prior CIVD research, the results may not be generalizeable to whole body cold exposure. Doppler blood flow measurements were not obtained in this study, and this would provide an alternative means of assessing the CIVD response. Finally, CIVD in a field setting may be affected by many additional variables such as altitude, caloric state, and training. This study was not designed to examine any of these effects.
Conclusions
This study found that pretreatment with 30 mg of oral nifedipine twice daily does not affect the CIVD response in healthy individuals under cold stress.
Footnotes
Acknowledgements
Author Contributions: Study design (BHM, SEM); data collection (BHM, SEM); manuscript writing and editing (BHM, SEM, GBZ).
Financial/Material Support: This study was supported through research funding from the Division of Emergency Medicine at the University of Utah.
Disclosures: None.
