Abstract
Introduction
In recent years a number of publications have appeared describing noninvasive blood pressure (NIBP) measurement with 1 layer of light cloth between the patient's skin and the NIBP cuff. In this study we investigated the reliability of NIBP measurements performed through 2 and 3 layers of autumn/winter clothing in 2 research groups: healthy volunteers and patients.
Methods
Oscillometric NIBP measurements were made in a random order: 1) on the exposed arm; 2) on the arm covered by a standardized cotton and polar fabric test sleeve; and 3) with the arm covered by a cotton–polar fabric and down jacket test sleeve. The time taken for measurement was also recorded.
Results
We performed measurement in 101 volunteers and 50 patients and found no clinically or statistically significant differences in NIBP. Mean±SD values in the volunteer group were 1) 130/80±14/8 mm Hg; 2) 130/81±15/8 mm Hg; and 3) 128/81±14/8 mm Hg. In the patient group the values were 1) 140/82±34/15 mm Hg; 2) 140/84±33/15 mm Hg; and 3) 137/84±32/15 mm Hg. Measuring over a sleeved arm extended the time of measurement by an average of 3.5 s in comparison with bare arm measurement.
Conclusions
Our findings confirm that it is possible to perform reliable NIBP measurements over 2 and 3 layers of autumn/winter clothing. Measuring NIBP with a clothed arm does not show clinical or statistically significant differences in comparison with measurements performed on the bare arm.
Introduction
Noninvasive blood pressure (NIBP) measurement is an essential method of assessment of circulation in patients in prehospital emergency medicine, allowing medical and paramedical personnel to start and monitor appropriate treatment.
Recommendations for the appropriate measurements of NIBP are clear and precise. They were established many years ago and have not been changed since then.
1
–4 Among them, the following points are important: Create a calm and comfortable environment for both patient and medical responder; Ensure the patient is in a seated position, if possible, with supported back and legs parallel; Use a cuff that is appropriate for the size of the patient's arm; Position the patient's arm with the cuff at the level of the right atrium; and Allow a 5-min delay between application of the cuff and commencement of the measurement.
Most recommendations underscore the importance of direct contact between the cuff and the bare skin of the patient's arm as being essential for reliable readings. Leaving a layer of clothing between the arm and cuff is traditionally considered to lead to false readings. 3 ,5,6
In emergency prehospital settings we have no specific recommendations for NIBP measurement, and those quoted are often impossible to follow in full or may lead to significant delays in clinical assessment of the patient's condition.
In recent years, several publications have described reliable NIBP measurement performed in ambulatory conditions with 1 layer of light cloth (cotton shirt, light sweater) between the patient's skin and the cuff. The authors concluded that there were no clinical and statistical differences between NIBP measurements made on the sleeved and bare arm. 7 –13 Extending these findings with more layers of various, thicker clothing commonly used during trekking or mountaineering would be very useful in prehospital emergency care, particularly in low-temperature conditions, confined spaces, or multiple casualty scenarios where valuable time could be gained and future heat loss would be limited at the early stage of clinical assessment.
Initial observations collected by the authors in ambulance and rescue helicopter practice in Poland during the autumn and winter found that NIBP measurements performed using a multipurpose defibrillator fitted with an automated NIBP measurement device through several layers of winter clothing gave reliable data, confirmed subsequently by measurements on the bare arms of the same patients.
In this study, we hypothesized that the differences between NIBP readings obtained from measurements performed through 2 and 3 layers of winter/autumn clothing and those obtained from measurements performed on bare skin would not be of clinical significance and would not trigger any medical intervention.
Methods
We designed a prospective study in which participants (healthy volunteers and hospitalized emergency patients) had a set of 3 NIBP measurements made in a randomly selected order from the following: 1) bare arm; 2) arm covered by the first test sleeve; and 3) arm covered by the second test sleeve with the bare arm measurement serving as a control value. A randomization table of sequences among the set for each participant was generated using a statistical computation internet application. 14 The time gap between each measurement in the set was 1 min. Data collection consisting of systolic NIBP (sys) and diastolic NIBP (dias) measurements and recording of the time (T) taken for each measurement was performed.
All blood pressure measurements were performed on the left arm and were made by the same researcher (a senior paramedic). In the volunteer group, examinations were performed with the participant in a seated position, and in the patient group participants were in a supine or semisupine position (always the same for the 3 measurements for a given patient).
The size of the NIBP cuff was adjusted to fit the circumference of the arm covered by each testing sleeve. In the case of a significant difference between the size of the arm and the testing sleeve, the walls of the sleeve were overlapped but always positioned opposite to the brachial artery.
To avoid cross infection in the patient group, the skin beneath the cuff and testing sleeves was covered by a layer of thin transparent plastic film. In addition, in some intensive care unit (ICU) patients, constant direct arterial pressure values were also recorded (Drager Infinity Delta XL) and compared with NIBP values obtained from cuff measurements as another way to verify the accuracy of the automated blood pressure device used over the sleeves in seriously ill patients.
Because of the nature of the study, blinding of the participants and the examiner was not possible.
The research project was approved by the bioethics committee of the Medical University of Łódź, Poland (RNN/154/15/KE). The study was conducted in local hospital, medical school, and fire departments in Elbląg, Poland, during the autumn/winter season 2015-16.
PARTICIPANTS
In the first stage of the study, we planned to test the research hypothesis in the healthy volunteer group and verify whether any differences in the mean NIBP values between the covered and bare arm were apparent. To encourage volunteers to take part in the study, we announced information about it in a poster campaign and lectures in the field of NIBP measurements; in addition, the researchers personally contacted the managers of each institution and its employees. All volunteers who were willing to take part in the research came to the examination room on the research days and had their NIBP measured.
After collecting measurements from the volunteer group and confirming that there were no clinical differences between values obtained on the bare or covered arm, we performed NIBP measurements in a patient group using the same measurement protocol. Patients were drawn from ambulance responses and emergency department and ICU admissions. The number of patients was a result of measurements made by the examiner on selected wards and in the ambulance on previously indicated days in research months. This was part of an agreement with hospital management. Because NIBP measurement is a standard clinical practice, informed, written consent from the patient group was not sought. However, oral consent was obtained from all conscious participants.
The study design did not set any inclusion/exclusion criteria for participants, and the total number of participants was not previously calculated. We believe that in this manner we were able to gather a mix of participants who may be considered a reflection of the unpredictable and different physiological states of patients treated in the out-of-hospital setting.
TEST SLEEVES
Both test sleeves were previously prepared and cut in a rectangular shape from commercially available winter/autumn cloth commonly used during hiking/trekking/mountaineering, measuring 50 cm long and 28 cm wide. The first sleeve consisted of 1 layer of cotton textile and 1 layer of polar fabric shield sewn together, and the second had 1 layer of cotton textile, 1 layer of polar fabric, and 1 down-fitted double-textile layer (as used in winter jackets) sewn together. The thickness of a single layer of the first sleeve was 8 mm; the second sleeve 17 mm as measured by caliper.
To allow comparison of the thickness of our sleeves with results obtained by other authors, we also used a second technique. We compared the circumference of the bare arm with the NIBP cuff applied (in mm) with the circumference of the arm covered by the each of testing sleeves with the NIBP cuff firmly compressing the fabrics underneath. The ruler, in both cases, was placed in the middle of the cuff width on its outer face. This thickness measurement technique was applied in a subgroup of 20 participants (10 volunteers and 10 patients) picked randomly from the whole research group (table with participant numbers was generated by means of online application). 14
DEVICE
Blood pressure measurements were performed using a Welch Allyn automated sphygmomanometer integrated with a Zoll X-Series defibrillator with an appropriately sized cuff (Welch Allyn FlexiPort Reusable MQ with twist lock connector). This device uses an oscillatory method to determine both systolic and diastolic blood pressure automatically. During the cuff inflation phase, the first measurements of NIBP are obtained. The device then starts to deflate the cuff and takes the second measurement to ensure consistency of the results being displayed. This device does not need any specific calibration protocol before making measurements. In accordance with the maker's recommendations, only 2-min intervals with the power off were allowed between measuring different patients.
For statistical analysis, a standard statistical calculator
14
was used, and analysis of variance (ANOVA) tests for paired values were applied. The level of statistical significance was defined as
Results
One hundred one participants volunteered to take a part in the first stage of the study. In the second stage of the research, 50 patients were examined (Table 1).
Characteristics of participants
BP, blood pressure.
The patient group included the following: 1) 11 patients being transported by ambulance; 2) 20 patients in the emergency department; and 3) 19 patients in the ICU (Table 2).
NIBP and time results recorded in the study
NIBP, noninvasive blood pressure; sys, systolic; dias, diastolic; T, time of measurement.
Mean differences between sys and dias NIBP values in a particular set are presented with 95% CIs. Additionally, post hoc power analysis and mean±SD time values (s) are shown. Statistical significance
Two subsequent measurements using each technique were performed on 10 participants from both groups (picked randomly) to verify the reliability of the NIBP values obtained with a short time gap between the measurements (60 s). This was done to determine if using this method would indicate a significant difference in the readings (Tables 3 and 4; Figures 1 and 2).
Values of 2 subsequent measurements of NIBP
NIBP, noninvasive blood pressure.
The table shows differences between 2 subsequent measurements of NIBP made with a short time gap (60 s) in each of the research sets. Twenty participants were randomly enrolled into these analyses (10 volunteers and 10 patients).
NIBP and direct pressure results among ICU patients
NIBP, noninvasive blood pressure; ICU, intensive care unit; sys, systolic; dias, diastolic.
In a group of 8 ICU patients with constant direct arterial pressure monitoring, no clinically or statistically significant differences were detected between all performed cuff measurements and direct measurement (ANOVA
Mean NIBP values with 95% CI and mean time of measurement values are shown. Additionally, differences between values in particular sets and direct blood pressure values are also indicated.
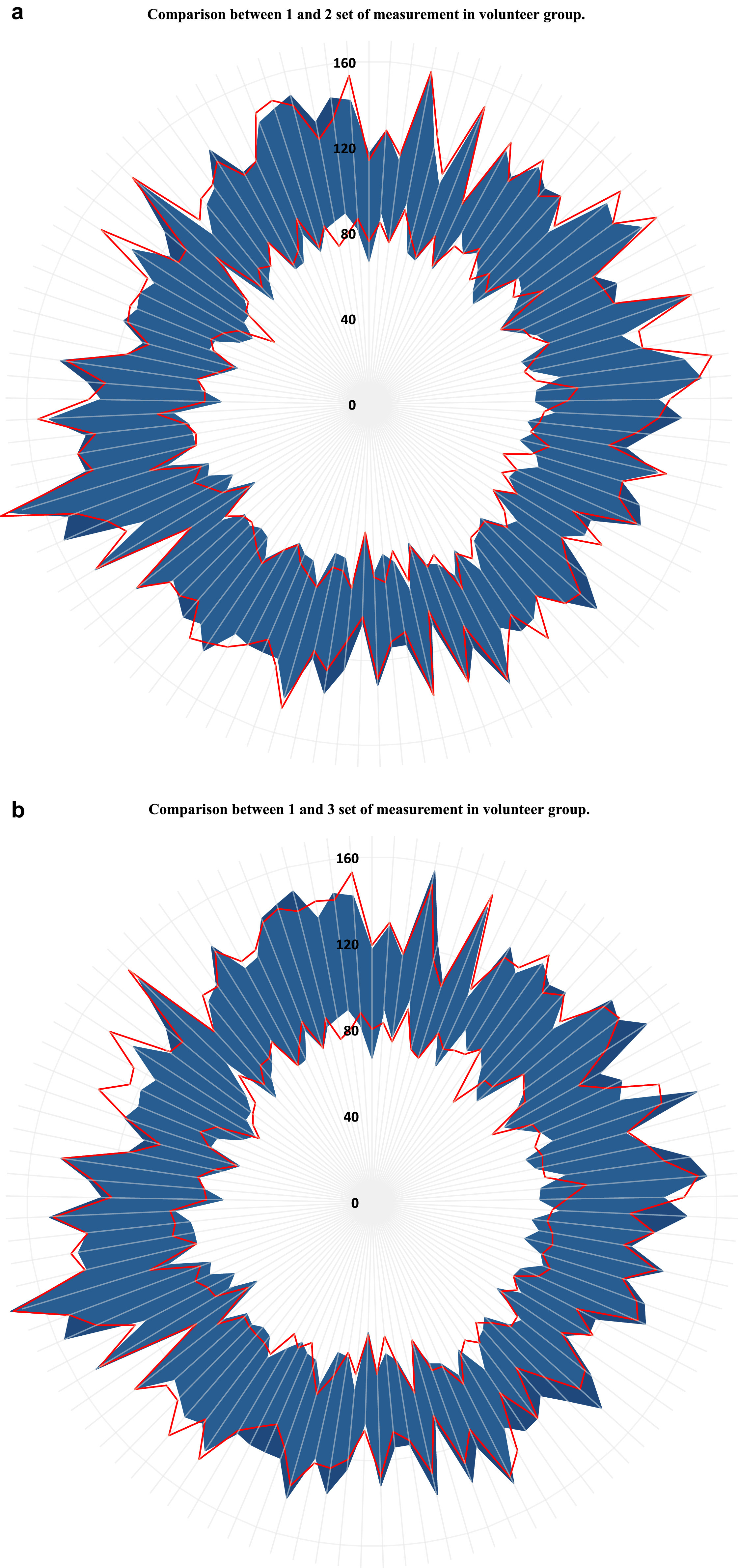
Radial diagrams for the volunteer group showing values obtained in set 1 in comparison to set 2 (A) and to set 3 (B) of noninvasive blood pressure (NIBP) measurements. The blue area presents reference values of NIBP made on a bare arm (set 1), where the inner edge shows diastolic and the outer edge systolic values of blood pressure. The red lines indicate values obtained in sleeved measurements (A, set 2; B, set 3). The inner red line shows diastolic and the outer line systolic NIBP. Each gray radius indicates one of the participants. Mean±SD values for NIBP in particular sets were as follows: set 1 = 130±14 / 80±8; set 2 = 130±15 / 81±8; and set 3 = 128±14 / 81±8 mm Hg.

Radial diagrams for the patient group showing values obtained in set 1 in comparison to set 2 (A) and set 3 (B) of noninvasive blood pressure (NIBP) measurements. The blue area presents reference values of NIBP made on a bare arm (set 1) with the inner edge for diastolic and the outer edge for systolic values of blood pressure. The red lines indicate values obtained in sleeved measurements (A, set 2; B, set 3), with the inner red line showing diastolic and the outer systolic NIBP. Each gray radius indicates one of the participants. Mean±SD values for NIBP in particular sets were as follows: set 1 = 140±34 / 82±15; set 2 = 140±33 / 84±14; and set 3 = 137±32 / 84±15 mm Hg.
The mean difference in circumference was 3±6 cm between the bare arm and the arm with the first test sleeve, and the difference was 8±6 cm for the second test sleeve, which corresponds with a widening gap between the arm and the cuff (cuff offset). In the case of the first sleeve, cuff offset was 0.5 mm; for the second sleeve, it was 1.25 cm.
The time taken for NIBP measurements in 92% of cases was within the manufacturer's range of 15 to 30 s. However, in 8% of participants (7 patients and 6 volunteers) the time needed to measure NIBP was significantly extended (average values were 32±4, 37±7, and 36±8 s;
In the group of volunteers the main cause of extension of the measurement time was the bigger size of the cuff (32–43 cm).
Discussion
In this study of 101 volunteers and 50 patients, we investigated the effect that 2 and 3 layers of winter/autumn clothing between the NIBP cuff and the arm had on the NIBP readings. We established the reliability of such measurements and found no clinically or statistically significant differences in values obtained when the arm was covered or bare.
In recent years a few published studies have assessed whether blood pressure readings vary significantly when measurements were taken 1) with a short time gap between each measurement; and/or 2) with a cuff positioned on a sleeved arm compared with a bare arm. 7 –13 Our results confirm and extend the current literature findings, but a few differences should be noted. In 2 studies8,11 subsequent measurements of NIBP in the same patient were analyzed. In the first, 8 a 4 mm Hg difference between first and second measurements was not statistically significant, and the latter 11 found no differences. In our study, although some differences were detected, these were of no clinical (from 1 to 2 mm Hg) or statistical significance. Therefore, we assumed that subsequent measurements with a 1 min gap between them, as a method used in this research protocol, had no influence on the reliability of the NIBP values obtained.
Another group of studies assessed whether blood pressure readings vary significantly when measurements are taken with a cuff positioned on a sleeved arm compared with a bare arm. The mean differences in NIBP values reported in each study were not statistically significant; these data and methodological information are shown in Table 5. 7 –9,11,12
NIBP measurements made through clothing: a comparison of previously described studies
NIBP, noninvasive blood pressure.
Some of the previous studies cited 8 –10 applied the same sequence for all measurements (on the bare or sleeved arm). In our protocol, the sequences for sets of all 3 measurements were randomly changed as in 2 other studies.11,12
Most published studies have used clothing belonging to volunteers: light cotton sleeves of various kinds (t-shirts, sweaters) with various parameters, including less or more than 2 mm thickness, 11 shirts and sweaters thinner than 2 mm, 12 clothing 1.7 cm thick, 7 and a 0.8 cm difference between the circumferences of the bare arm and sleeved arm. 9
In our study we used standardized test sleeves made of 2 and 3 layers of structurally different fabrics for all measurements. These sleeves were thicker with a wider range of density than those previously described. During the measurements, we customized the cuff size to the arm circumference, as did authors of 1 cited study. 9 Other authors do not mention the cuff size or indicate that only 1 cuff size was used, which may be considered as a methodological weakness.
Importantly, previous studies did not record the time needed for NIBP measurement made on sleeved arms, 7 –13 although this seems to be an important factor in prehospital care. In this respect, our findings indicate that most NIBP measurements made on a clothed arm do not take considerably longer (average 3.5 s) than measurements on a bare arm. This is an important factor if compared with the time needed for cutting or removing 2 or 3 layers of clothing. Our results have shown that reliable NIBP measurements can be made through fabric, eliminating the need for undressing the patient and therefore eliminating unnecessary heat loss, especially in the early stage of medical intervention. The time needed for this procedure, although longer in some cases than the measurement on a bare arm, would be shorter than that required to expose the arm and subsequently take a measurement.
Moreover, it is worth noting that in contrast to other published articles, 7 –13 we studied both seriously ill patients and healthy volunteers.
The intra-arterial values of 8 direct pressure measurements gave a measure of the accuracy of the automated sphygmomanometer used in the study.
The study reported here has a number of limitations, including the following: We used only 1 specific automated NIBP measurement device; The volunteer group was homogenous (76% normotensive, age 26±5 y); Hypotensive participants accounted for only 10% of the total patient group; The research methodology did not allow blinding of the researcher and participants; and In comparison to other studies, we had a lower number of healthy volunteers taking part in the research.
Conclusions
Our findings confirm that with use of a specific automatic NIBP device it is possible to perform reliable NIBP measurements over 2 and 3 layers of winter/autumn clothing among healthy volunteers in a test environment and among patients in wide range of emergency circumstances. NIBP values obtained on the covered arm are not clinically or statistically significant different when compared with NIBP measurements performed on the bare arm in the same participants, indicating that they did not trigger any medical intervention in the patient group.
In addition to these findings, we noticed that in 92% of cases measuring NIBP on a sleeved arm extends the time of measurement by an average of 3.5 s compared to the bare arm method but still remains within the declared device range. In an emergency situation, given the time needed to undress the patient's arm to perform a bare arm NIBP measurement, we consider this extension to be of no clinical significance.
In summary, these findings, together with previous studies, show that NIBP can be reliably measured through layers of sleeves on the arm even if autumn/winter clothing is being worn. This is of importance in the efficient early management of emergencies, particularly in moderate and cold climate conditions.
This study should be regarded as preliminary and should be followed up by a larger, multicenter study using a variety of automated NIBP devices.
Footnotes
Acknowledgements
Author Contributions: Analyzed the relevant literature, supervised acquisition of the data, drafted the manuscript, and prepared the figures and tables (PW); identified the problem, designed the trial, and prepared the application to the ethics committee (IB); provided some important methodological suggestions, critically revised the manuscript, and adjusted it according to scientific and publishing standards (DB).
Financial/Material Support: None.
Disclosures: None.
