Abstract
Bites by the European adder (Vipera berus) are a rare medical emergency in the UK with 20 to 50% of an estimated 50 to 200 cases per year necessitating treatment with antivenom. We present a case demonstrating both severe systemic and local effects necessitating out of hospital resuscitation, vasopressor support, and prolonged rehabilitation. Hemodynamic stability was restored promptly after administration of ViperaTAb antivenom, the first published case of its use in the UK.
Background
UK national guidelines for antivenom administration for V berus envenoming from National Poison Infor- mation Service at
After the cessation in manufacture of Zagreb antivenom, Public Health England recommends the use of ViperaTAb (MicroPharm, Newcastle Emlyn, UK) for the management of V berus envenomation. ViperaTAb is a monovalent FAB antivenom derived from sheep hyperimmunized against V berus venom. Animal models demonstrate cross reactivity across the European Vipera spp. 2 This is supported by clinical data from small cohorts of patients treated for Vipera aspis and Vipera ammodytes envenomation.3,4
Although this is the first published case of ViperaTAb use in the UK, data are available from Scandinavia, where it has been in use in the management of V berus envenomation for 20 y. 5 Because of a lack of randomized controlled trials, comparison of antivenoms used for management of European Vipera envenomation is difficult. A systematic review assessing current antivenoms in Europe found clinical outcomes to be better in those given intravenous (IV) ViperaTAb and ViperFAV when compared with Biomed, Bulbio, and Zagreb antivenoms, which are predominantly administered intramuscularly. 6
Although favorable outcomes have been seen in patients administered ViperaTAb in Scandinavia, variations in venom composition and envenomation syndromes within European viper species demonstrate a need for further data describing antivenom use in new geographical cohorts. 7
Case report
A 62-y-old woman with a minimal medical history was bitten while walking in the New Forest, UK in June 2016. The patient recalled feeling a sharp sting on her right ankle and described seeing a snake “slithering” away. Together with her friend, she identified the markings of the snake on her smartphone and found it to be an adder. She had no previous snake venom exposure. Within a few minutes of the envenomation, she developed progressive pain and rapid swelling of the affected leg. Over the next 30 min she became progressively more unwell with vomiting, diarrhea, and prostration and was incontinent of both urine and feces.
The air ambulance service arrived near the location of the bite 53 min after the envenomation. On arrival the patient was pale and clammy. Her pulse was faint and blood pressure (BP) was 65/30 mm Hg. The paramedic team suspected an anaphylactic reaction and administered 2 doses of 500 microgram intramuscular epinephrine, 20 mg IV chlorphenamine, 150 mg IV hydrocortisone, and 1 L IV normal saline. Because only modest improvement in BP (85/52 mm Hg) was noted, a further 100 microgram IV epinephrine (1:10,000) was administered.
The patient arrived at Southampton General Hospital 2 h after envenomation. She felt generally unwell, her Glasgow Coma Scale was 15, and arterial blood gas demonstrated lactic acidosis (pH 7.22, lactate 3.6 mEq·L-1). Her admission venous blood tests demonstrated marked leukocytosis and thrombocytopenia (hemoglobin 173 g·dL-1, white blood count 26.4×109/L, platelet count 103×109/L, neutrophil count 23.5×109/L, international normalized ratio 0.9, creatinine 114 μmol·L-1, and creatine kinase 92 units·L-1). The envenomated leg was grossly swollen to above the knee. After a telephone discussion with the UK National Poison Information Service, 2 vials of ViperaTAb in 100 mL of normal saline were administered intravenously. After administration of the antivenom, the patient began to feel better in under an hour. Her BP remained above 120/80 mm Hg without further doses of epinepherine or IV fluid. The leg remained swollen and tender but did not increase further in size.
The next day the leg was warm and tender, and the patient was unable to bear weight. Her clinicians commenced flucloxacillin and clindamycin IV because of concerns of cellulitis (Figure 1). She was monitored for evidence of compartment syndrome with frequent orthopedic assessment. Compartment pressure measurements were not obtained. The patient received 2 units of packed red blood cells because of a decrease in hemoglobin from 173 g·L-1 on admission to 74 g·L-1 after 4 d. The patient did not receive any anticoagulation. The decrease in hemoglobin was attributed to a combination of extensive ecchymosis and dilutional anemia after fluid resuscitation. Over the course of the next week, the pain eased and her mobility slowly returned with the assistance of physiotherapy. She was discharged home on day 11. On follow-up at 2 mo, the patient reported intermittent swelling and continued to walk with the assistance of a walking stick.
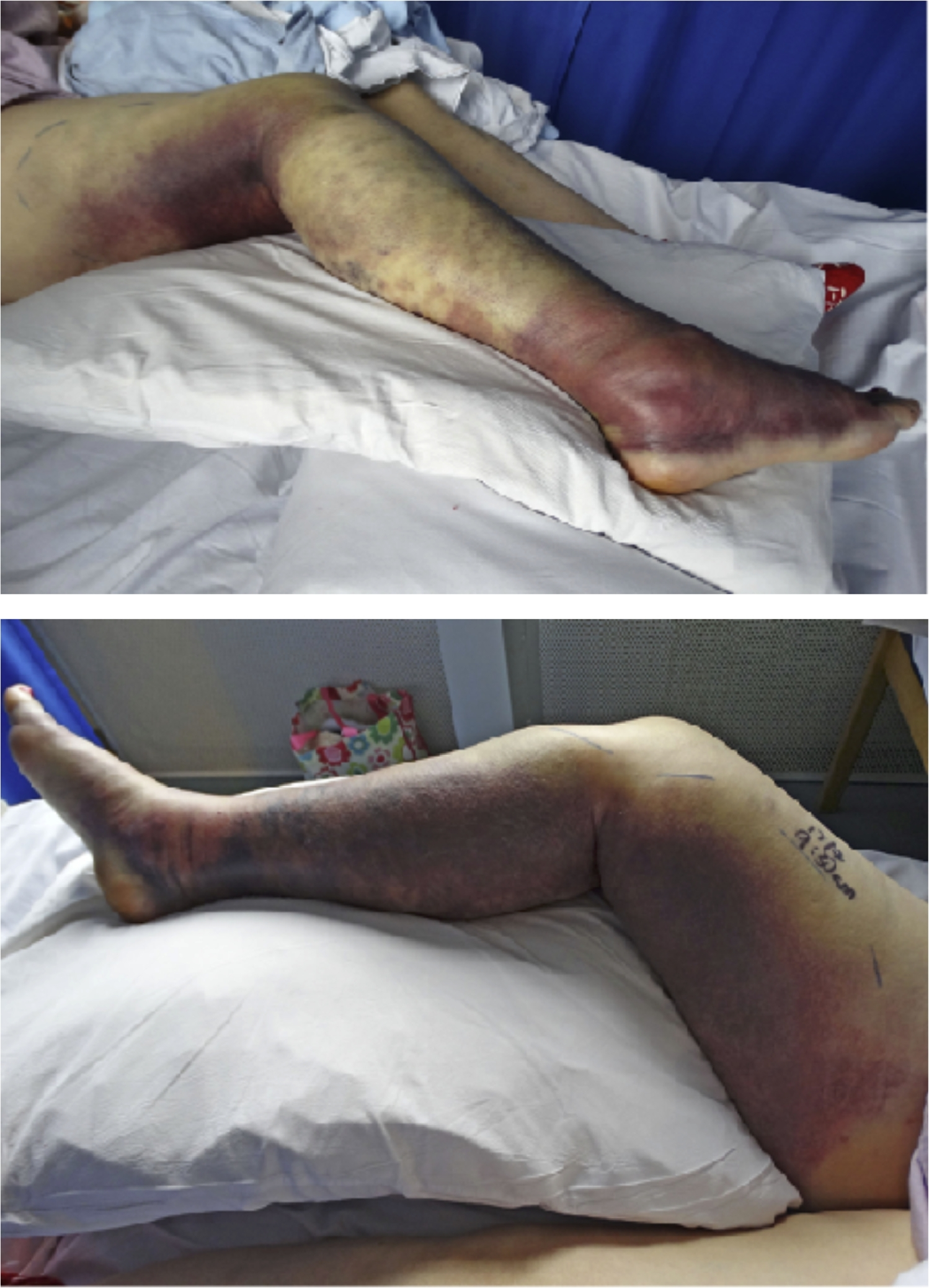
Photographs taken on day 3 of admission demonstrating significant ecchymosis and limb edema.
Discussion
The case report describes severe early systemic V berus envenomation followed by local effects characterized by significant limb edema and prolonged hospital admission. Life-threatening hypotension related to V berus envenomation is rare but well described. 8 In this case, out-of-hospital resuscitation, vasopressor support, and prompt assessment and administration of antivenom resulted in resolution of life-threatening hemodynamic instability.
The etiology of hypotension in snakebite is complex and multifactorial. Snake venom contains a diverse array of proteins and peptides that cause hypotensive effects such as angiotensin converting enzyme inhibition, histamine release, and extravasation of plasma resulting in severe localized edema. 9 A recent assessment of the V berus venom proteome has identified 4 bradykinin-potentiating peptides, which could further explain the hypotension seen with severe systemic envenomation. 10 The role of anaphylactoid symptoms in snake envenomation is poorly understood.
Prolonged functional impairment (greater than 14 d) as described in this case is a common feature of severe Vipera envenomation, occurring in 24% of patients in a cohort of envenomated French patients. Early administration of antivenom (<10 h after bite) was associated with reduced frequency of functional impairment, whereas low-molecular-weight heparin was associated with increased functional impairment. 11
Historically, there has been reluctance to use antivenom for Vipera spp envenomation after a fatal anaphylactic reaction to subcutaneous administration of the now out-of-production Pasteur antivenom. 12 This resulted in a statement in the British National Formulary 1974−1976 that “the bite is less dangerous than the antisera.” This is no longer the case with greater purity techniques used in the production of modern antivenoms. Adverse reactions to ViperaTAb have been reported in 0.5% of cases, with 1 reported case of anaphylaxis. 6
Until 2016, the UK had been reliant on the importation of Zagreb antivenom, an F(ab’)2 antivenom raised against horse sera, for the management of moderate to severe V berus envenomation. Although anecdotally there appeared to be good clinical response to Zagreb antivenom administered IV in the UK, there is a scarcity of clinical data. After cessation of its manufacture, Public Health England now recommends the use of ViperaTAb for the management of V berus envenomation.
The rapid improvement in clinical condition after administration of ViperaTAb in this case is consistent with prior literature. 6 This suggests that the known efficacy and safety profile of ViperaTAb is applicable locally in the UK. Further research would be helpful to determine who would benefit, when to administer, and what dose antivenom is most efficacious.
Conclusion
Although uncommon, V berus envenomation is potentially life threatening and can require resuscitation, close monitoring, and prompt administration of antivenom. Further prospective data on the use of ViperaTAb in the UK would be useful.
Footnotes
Acknowledgments
The authors thank Professor Michael Eddleston, Professor of Toxicology, University of Edinburgh.
Author Contributions: Study concept and design (JH, TL); acquisition of the data (JH, TL, JK); analysis of the data (JH, TL); drafting of the manuscript (JH); critical revision of the manuscript (JH, TL); and approval of final manuscript (JH, TL, JK).
Financial/Material Support: None.
Disclosure: None.
