Abstract
Background
Chronic hypoxia-induced pulmonary hypertension and vascular remodeling have been shown to be associated with ornithine decarboxylase 1 (ODC1). However, few animal studies have investigated the role of ODC1 in acute hypoxia.
Objectives
We investigated ODC1 gene expression, morphologic and functional changes, and the effect of L-arginine as an attenuator in lung tissues of rats exposed to acute hypobaric hypoxia at a simulated altitude of 6000 m.
Methods
Sprague-Dawley rats exposed to simulated hypobaric hypoxia (6000 m) for 24, 48, or 72 hours were treated with L-arginine (L-arginine group, 20 mg/100 g intraperitoneal; n=15) or untreated (non–L-arginine group, n=15). Control rats (n=5) were maintained at 2260 m in a normal environment for the same amount of time but were treated without L-arginine. The mean pulmonary artery pressure was measured by PowerLab system. The morphologic and immunohistochemical changes in lung tissue were observed under a microscope. The mRNA and protein levels of ODC1 were measured by real-time polymerase chain reaction and Western-blot, respectively.
Results
Hypobaric hypoxia induced pulmonary interstitial hyperemia and capillary expansion in the lungs of rats exposed to acute hypoxia at 6000 m. The mean pulmonary artery pressure and the mRNA and protein levels of ODC1 were significantly increased, which could be attenuated by treatment with L-arginine.
Conclusions
L-arginine attenuates acute hypobaric hypoxia-induced increase in mean pulmonary artery pressure and ODC1 gene expression in lung tissues of rats. ODC1 gene contributes to the development of hypoxic pulmonary hypertension.
Introduction
Ornithine decarboxylase (ODC), a rate-limiting enzyme in the metabolism of polyamine, plays an essential role in cell growth, differentiation, and proliferation. 1 ODC can be divided into ODCl and ODC2, and ODC1 is the major functional gene widely expressed in various tissues.2,3 Previous studies have found that polyamine contents are elevated in lung tissues under chronic hypoxia. 1 The administration of α-difluoromethylornithine, a specific irreversible inhibitor of ODC, prevented pulmonary hypertension, pulmonary edema, and right ventricular hypertrophy induced by monocrotaline in rats. 4 L-arginine treatment could result in a decrease in ODC expression and the protein levels of inducible nitric oxide synthase and an increase in the nitric oxide (NO) concentration, indicating that the activation of L-arginine/NO pathway attenuated cardiac hypertrophy and protected the heart. 5
Hypoxic pulmonary vasoconstriction is the response of the pulmonary vasculature to acute or sustained alveolar hypoxia, by which lung perfusion is matched with ventilation. Under local alveolar hypoxia, perfusion and ventilation are matched, which results in improved gas exchange; under global alveolar hypoxia, hypoxic pulmonary vasoconstriction may lead to pulmonary hypertension. In response to alveolar hypoxia, a mitochondrial sensor dynamically changes reactive oxygen species and redox couples in pulmonary artery smooth muscle cells. At high altitudes, global hypoxia can lead to diffuse pulmonary venous hypertension and then to increased pulmonary artery pressure and pulmonary hypertension. 6 Thus, exaggerated or heterogeneous hypoxia pulmonary hypertension contributes to high-altitude pulmonary edema (HAPE). 7
Our unpublished data have shown that ODC1 mRNA is highly expressed, and the mean pulmonary artery pressure (mPAP) is significantly increased in patients with HAPE. Thus, it is critical to understand how ODC1 leads to pulmonary vasoconstriction, and subsequently capillary inflammation and leakage, and finally HAPE. We assumed that ODC1 may be involved in pulmonary artery contraction and pulmonary hypertension in acute hypoxia. As such, the main purpose of this study was to evaluate ODC1 gene expression in lung tissues and mPAP of rats exposed to acute hypobaric hypoxia for different times. In addition, we also investigated whether L-arginine as the inhibitor of ODC1 attenuated pulmonary artery contraction and ODC1 gene expression.
Methods
Animals
Thirty-five male Sprague-Dawley rats of the same age (about 6 weeks) and weight (180–210 g) were provided by the Animal Center of Xi’an Jiao Tong University (Xi’an, China) and were maintained under the same conditions in Xi’ning (2260 m ), China, with free access to food and water. They were divided into 3 groups at random: 5 rats were maintained at a moderate altitude of 2260 m at 18–20°C (control group [CTRL], n=5); 15 rats were exposed to a simulated altitude of 6000 m in a hypoxic chamber for 1–3 days at 18–20°C and then treated with 20 mg/100 g of L-arginine (intraperitoneal) 24 hours before discharge (L-arginine [ARG] group, n=15); and the remaining 15 rats were also exposed to the simulated altitude of 6000 m but received no L-arginine (non–L-arginine [NARG] group, n=15). Those rats exposed to the simulated altitude of 6000 m were further randomized into 3 subgroups of 5 rats each (n=5) according to the exposure duration: 1) 24 hours; 2) 48 hours; and 3) 72 hours. Afterward, all rats were removed from the hypoxic chamber and intraperitoneally anesthetized for the measurement of mPAP, and lung tissues were separated and cryopreserved. Rats died after the separation of lung tissues. All experimental protocols were approved by the Ethics Committee of Qinghai University.
Design and Synthesis of Primers
The primers of ODC1 (target gene) and 18S (reference gene) were designed by Primer Premier 5.0. All primer sequences were synthesized by TSINGKE Biotech Company (Beijing, China) (Table).
Primer sequences for reverse transcription polymerase chain reaction analysis
Measurement of mPAP
The mPAP was measured by right-heart catheterization using a PowerLab system (ADInstruments, Bella Vista, Australia).
Hematoxylin-Eosin Staining and Immunohistochemistry of Lung Tissues
Right lung tissues were harvested, chopped into small pieces, washed with saline, fixed with 4% paraformaldehyde, and embedded in paraffin. Hematoxylin-eosin staining was performed, and the sections were examined and photographed under an Olympus microscope. Immunohistochemistry was performed using specific fluorescent antibodies against ODC following the standard procedure. 8 A rabbit polyclonal antibody against ODC1 (Abcam, Cambridge, MA) was used at a dilution of 1:250 in blocking buffer. The slides were incubated with the primary antibody at +4B°C overnight, washed with phosphate-buffered saline, and incubated with Alexa Fluor 488 conjugated anti-rabbit IgG secondary antibody (1:500 dilution, Abcam, horseradish peroxidase) for 2 hours at room temperature. The stained sections then were observed and photographed.
Determination of Lung Tissue mRNA
Right lung tissues were harvested and homogenized to extract the total RNA. The extracted total RNA was stored at –80°C until further use. First-strand cDNAs were synthesized using a Tiangen FastQuant RT Kit according to the manufacturer’s instructions. The polymerase chain reaction (PCR) system (20 μL) consisted of 10 μL of 2 × Taq PCR Master Mix (Applied Biosystems, Foster City, CA), 1 μL of cDNA, ODC1, 0.1 uL of upstream and downstream primers, and sterilized double-distilled water. Real-time PCR was performed in duplicate on an ABI Prism 7500 Sequence Detection System (Applied Biosystems). The PCR plate was incubated at 95˚C for 6 minutes, followed by 40 cycles of 95˚C for 15 seconds, and 60˚C for 1 minute.
Measurement of ODC1 Protein Levels
Lung tissues were dissected and homogenized in lysis buffer. Proteins were separated by sodium dodecylsulfate polyacrylamide gel electrophoresis and then blotted onto nitrocellulose membranes. They were incubated with primary antibodies (anti-rat ODC1; Abcam) and then with horseradish peroxidase–conjugated secondary antibodies (goat anti-rat ODC1; Abcam) after washing. Immunoreactive proteins were visualized with ECL detection reagents. Immunopositive bands were scanned by an imaging densitometer.
Statistical Analyses
All data are presented as mean±SD and were analyzed using the Statistical Package for Social Sciences for Windows (SPSS 18.0). The normality test was performed by the Kolmogorov and Smirnov method. The significance of difference among different groups was determined by one-way analysis of variance and repeated measures analysis of variance, followed by the least significant difference post hoc test. The fold change of the ODC1 gene was calculated using the delta delta CT method, in which the ODC1 gene was adjusted to corresponding reference gene (18S) levels. Statistical significance was accepted at P < .05.
Results
The mPAP and Morphologic Changes of Lung Induced by Hypobaric Hypoxia
Figure 1 shows that the mPAP is significantly higher in rats exposed to hypobaric hypoxia for 24–72 hours (ARG and NARG groups) than moderate altitude (control) (P < .05). However, it is important to note that intraperitoneal injection of L-arginine leads to a significant decrease in mPAP (P < .05 for NARG-1 group vs ARG-1 group; NARG-2 group vs ARG-2 group; and NARG-3 group vs ARG-3 group).
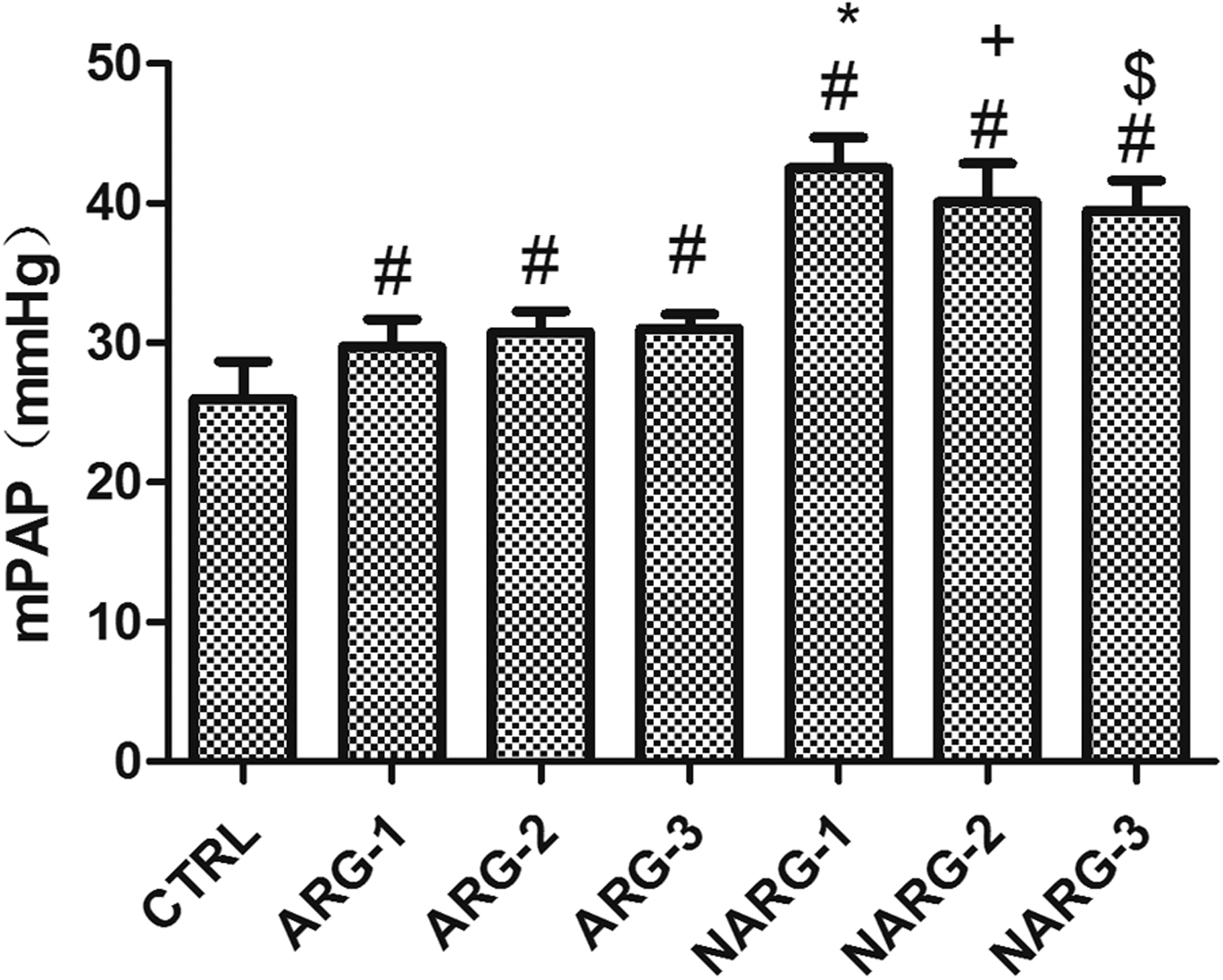
The mean pulmonary artery pressure (mPAP) changes in different groups. Data are presented as mean±SD. #, P<.05, all hypoxia groups (ARG and NARG) vs control group; *, P < .05, NARG-1 group vs ARG-1 group; +, P < .05, NARG-2 group vs ARG-2 group; $, P < .05, NARG-3 group vs ARG-3 group.
Intact alveoli and pulmonary interstitium with no liquid leakage were found in rats in the control group. No significant morphological changes in lung are observed in rats in the ARG groups. However, clear swelling of pulmonary interstitium, hyperemia, and telangiectasis were found in rats in the NARG group; their alveolar cavities deform slightly and become smaller, but with no liquid leakage (Figure 2A–G).
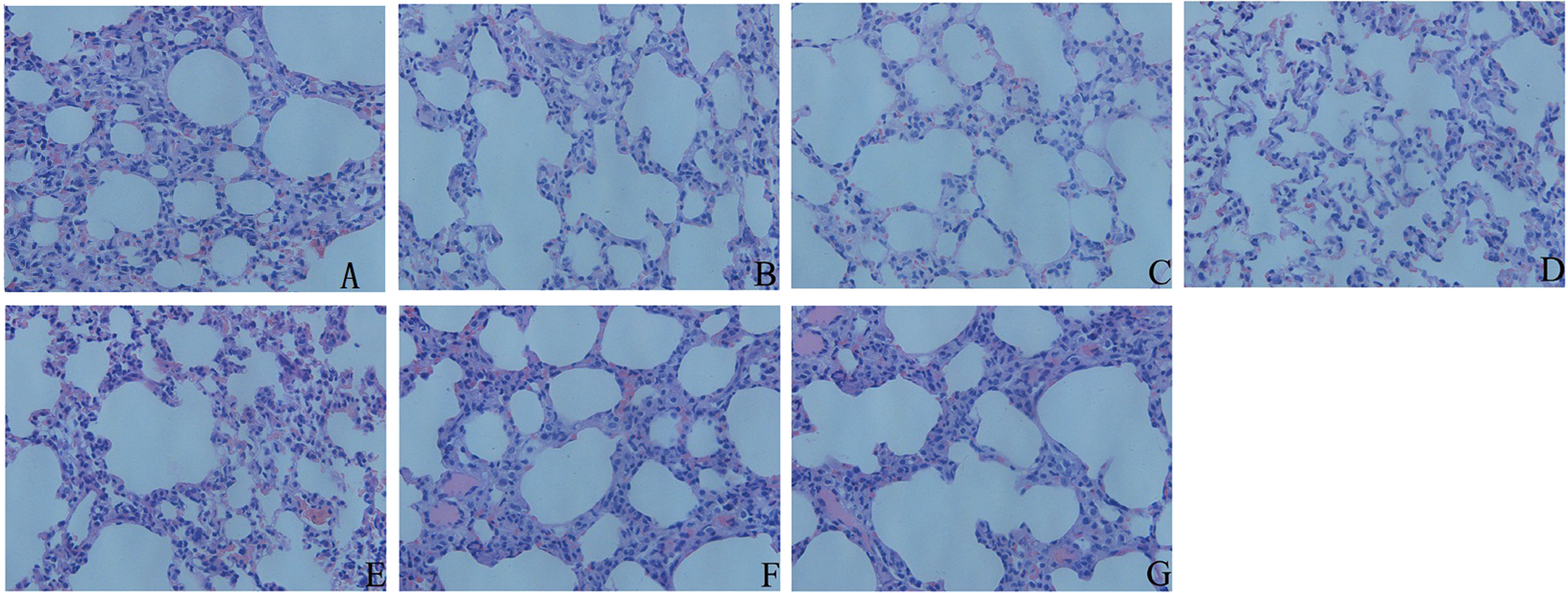
The hematoxylin-eosin staining results in all hypoxia groups (ARG and NARG) and control groups at ×40.
Alterations in ODC1 of Rats Exposed to High Altitude
Figure 3 shows increased fluorescence intensity in the ARG-2, NARG-1, and NARG-2 groups compared with the control group. In addition, intensity is higher in the NARG-1 and NARG-2 groups than in the ARG-1 and ARG-2 groups, respectively (P < .05). However, there is no significant difference between the NARG-3 and ARG-3 groups (Figure 3A–H).
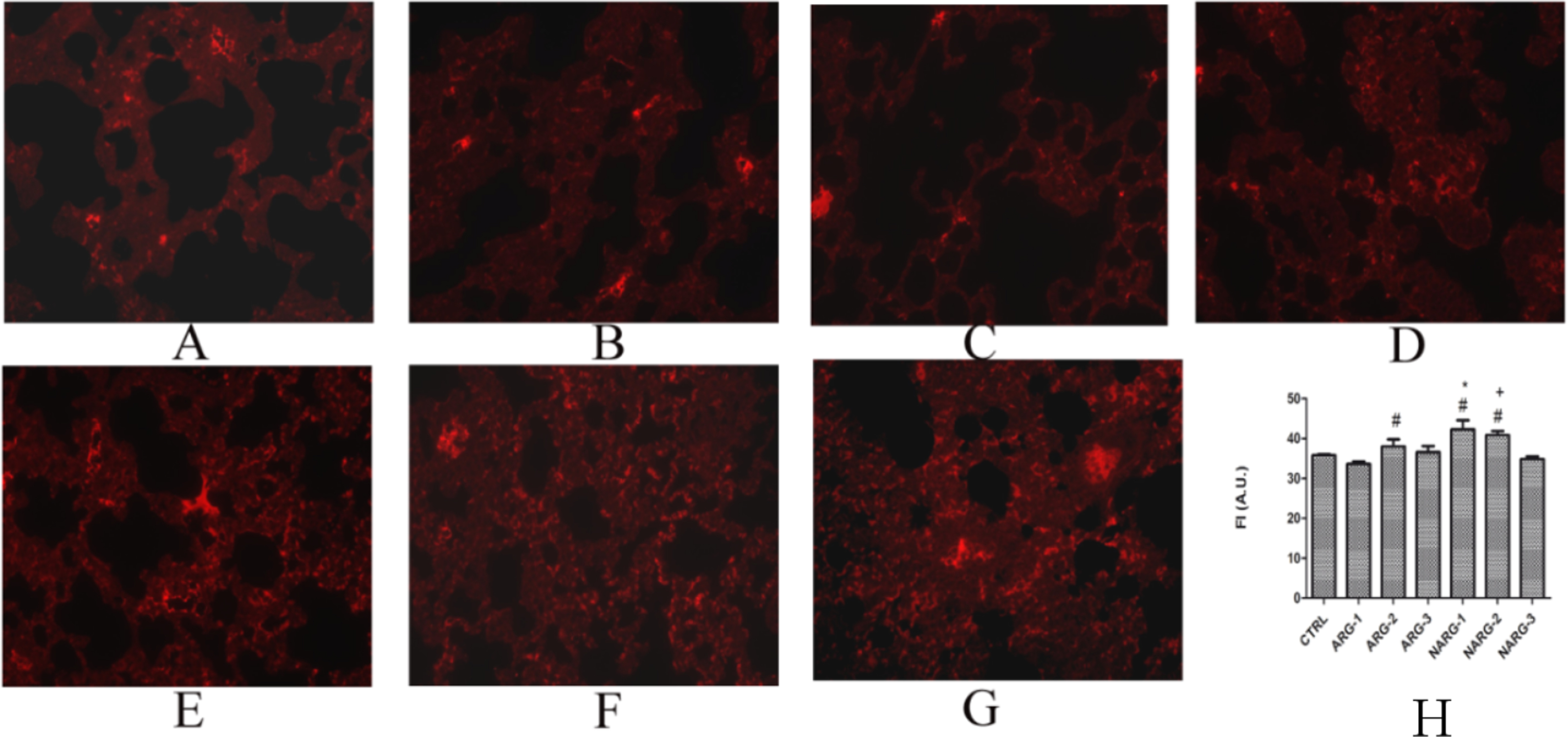
The immunohistochemical results of ODC1 in different groups at ×40.
ODC1 mRNA expression is significantly increased in the ARG-1, NARG-1, and NARG-2 groups. In contrast, the administration of L-arginine results in a significant decrease in ODC1 mRNA expression in the ARG-1 and ARG-2 groups compared with the NARG-1 and NARG-2 groups (P < .05). The mRNA expression reaches a peak at 24 hours and then decreases gradually at 48 and 72 hours (P < .05) (Figure 4).
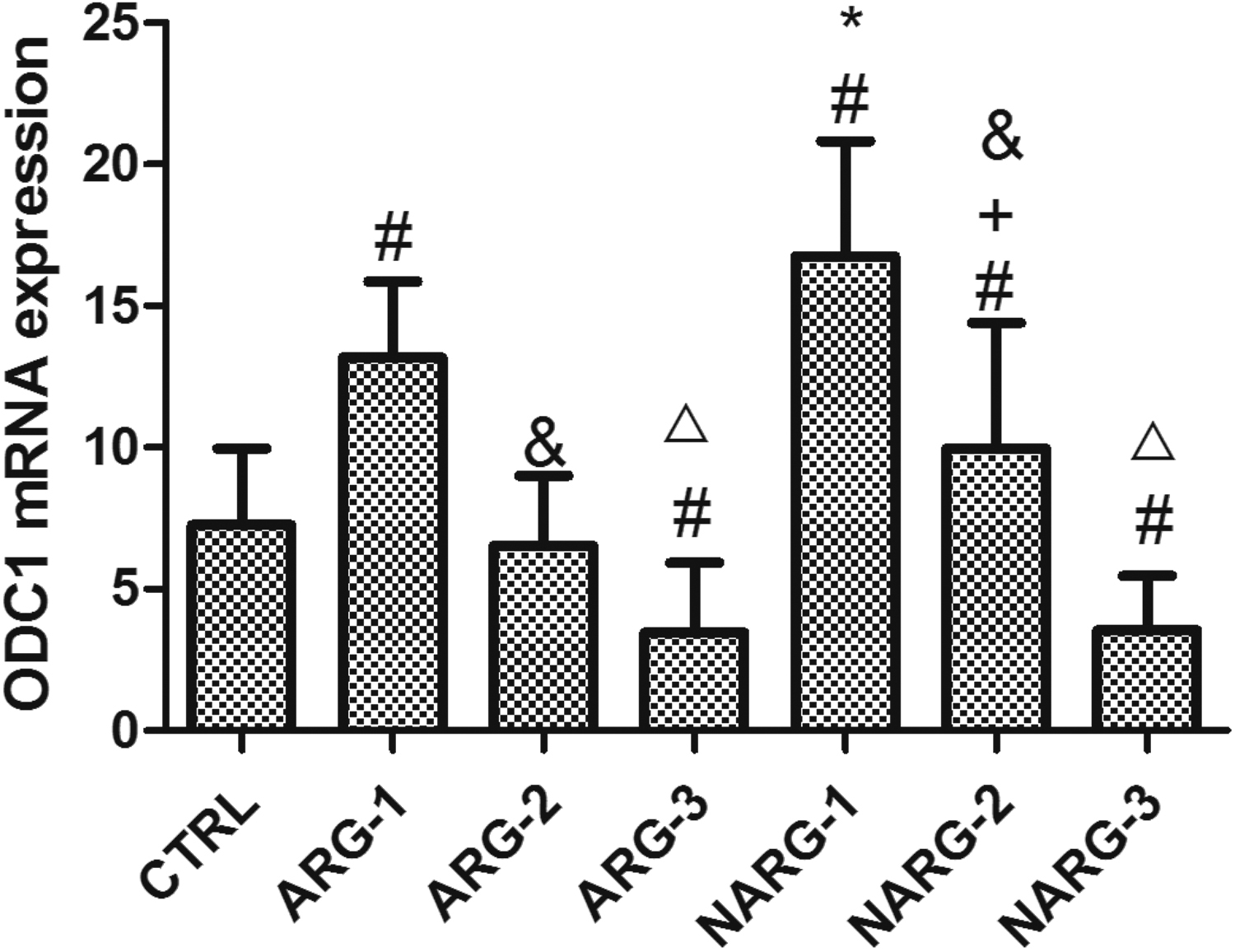
ODC1 mRNA expressions in different groups. Data are presented as mean±SD. #, P < .05, ARG-1, ARG-3, and all NARG groups vs control groups; *, P < .05, NARG-1 group vs ARG-1 group; +, P < .05, NARG-2 group vs ARG-2 group; &, P < .05, 48 hours vs 24 hours; △, P < .05, 72 hours vs 24 hours; and ▲, P < .05, 72 hours vs 48 hours in ARG and NARG groups, respectively.
The ODC1 protein level is significantly higher in the NARG groups than in the control group. In addition, it is higher in the NARG-1 and NARG-2 groups than in the ARG-1 and ARG-2 groups, respectively (P < .05). However, there is no significant difference among rats exposed to hypoxia for 24, 48, and 72 hours (Figure 5).
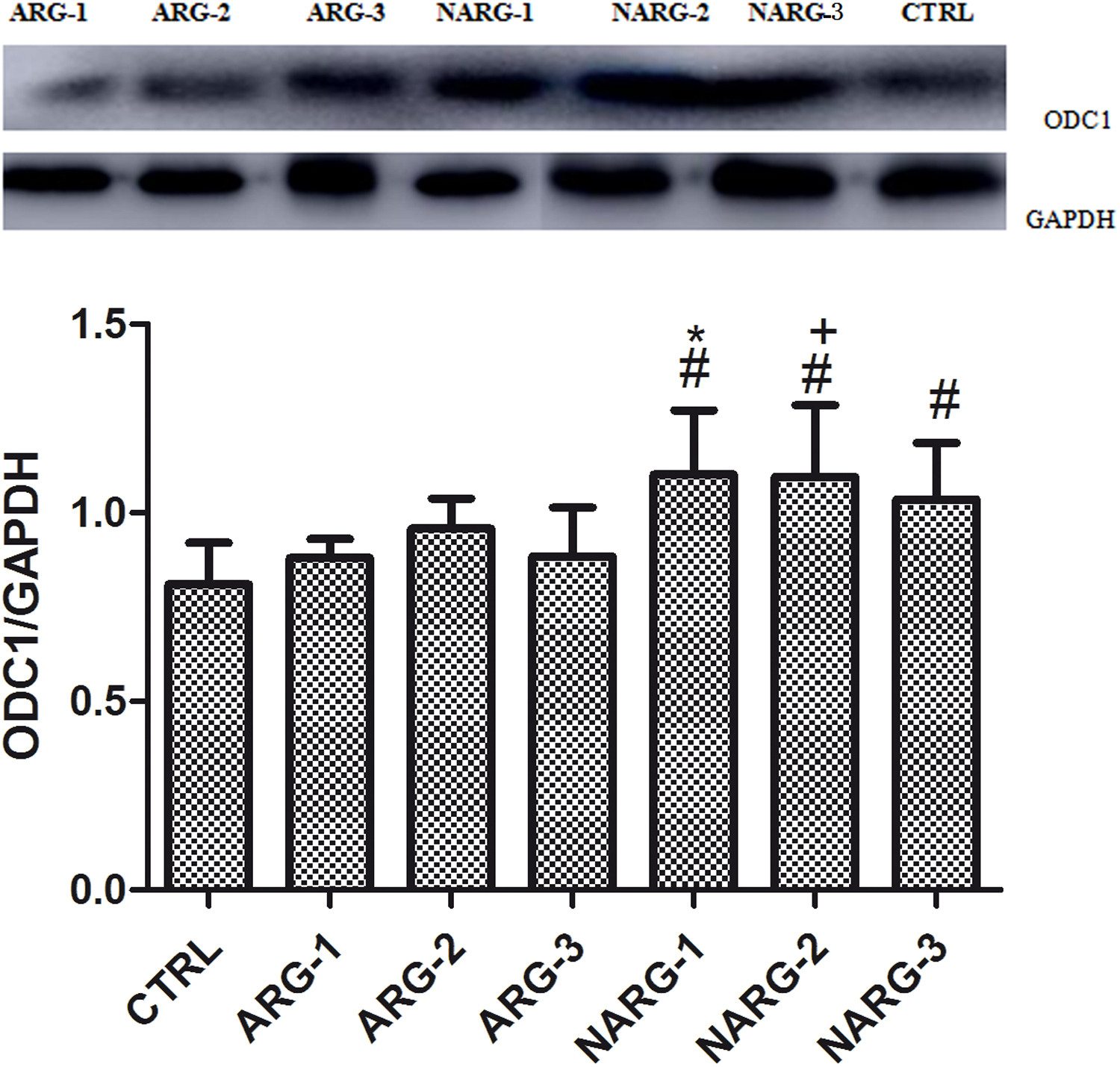
ODC1 protein levels in different groups. The gray level comparison of ODC1 and GAPDH in the same group at the top of the figure, and the densitometry values are shown below gray level. Data are presented as mean±SD. Densitometry values are represented as ratio of ODC1 to GAPDH protein. #, P < .05, all NARG groups vs control group; *, P < .05, NARG-1 group vs ARG-1 group; +, P < .05, NARG-2 group vs ARG-2 group.
Discussion
In this study, we found that ODC1 gene expression and mPAP were increased in the lung tissues of rats exposed to acute hypobaric hypoxia; however, this could be attenuated by L-arginine. Notably, these results imply that ODC1 may be mediated in pulmonary arterial hypertension in hypobaric hypoxia.
Acute exposure to hypoxia can cause an increase in mPAP within 1–21 days.9,10 In this study, we found that mPAP was significantly increased in rats exposed to acute hypoxia for 1–3 days and decreased after treatment with L-arginine. The pathologic mechanism of HAPE is pulmonary arterial hypertension and increased activity of sodium ion channel, water channel proteins, and genetic changes.11,12 This can explain the mechanism of HAPE. Acute exposure to high altitude can also lead to swelling of the pulmonary interstitium, hyperemia and telangiectasis, and alveolar cavities in the lung tissues of rats. The arginine pathway and the inflammatory response are interregulated, and the repair phase of the arginine pathway will attenuate the inflammatory response. 13 Our results have verified previous results and reveal that these changes could be attenuated by treatment with L-arginine.
Previous studies have reported that polyamine contents are elevated in lung under chronic hypoxia and that pharmacologic inhibition can increase hypoxic pulmonary vascular remodeling and hypertension. 1 ODC1 mRNA levels were increased in the brain tissues of rats exposed to acute hypoxia for 2–4 hours, and thus the ODC1 activity may be related to the protective mechanism of brain to hypoxia. 14 Hypoxia-induced increase in ODC1 expression is related not only to those factors that can downregulate the functions of membrane receptors but also to some indirect factors, such as hypoxia stress reaction and signal transduction of adrenergic response. 15 The increased ODC1 and polyamine activity may be involved in cell damage in HAPE. 13 The increase in ODC1 may lead to systemic inflammation and alveolar damage, such as hypoxia stress reaction, immune response and transcriptional regulation, signal transduction and RNA metabolism, and protein metabolism. 16 Our study has also suggested that the mRNA and protein levels of ODC1 are significantly increased in rats exposed to acute hypoxia at 24 hours and then decreas gradually at 48–72 hours. The mRNA level decreases more quickly than the protein level, probably because mRNA is less stable and thus prone to degradation. We found a decrease in ODC1 gene expression in the ARG-3 group, which was attributed to the hypoxic acclimatization of rats. 17
However, the mPAP and the ODC1 gene expression could be reduced by L-arginine.
Limitations
This study has several limitations. The first limitation concerns the small number of participants, which may limit the generalizability of our findings. Further studies with a larger number of participants are warranted to confirm these findings. Difluoromethylornithine is the best inhibitor of ODC1, but it is delisted in China. Furthermore, chronic hypoxia is not addressed in the experimental design, and the upstream and downstream of ODC1 gene are not measured.
Conclusions
L-arginine can attenuate the increase in mPAP and ODC1 gene expression in lung tissues of rats exposed to acute hypoxia. Our study contributes to better understanding the pathogenesis of HAPE at the genetic level.
Acknowledgments: The authors sincerely thank all the study subjects.
Author Contributions: Study concept and design (LY); acquisition of the data (BZ, TF, YQ); analysis of the data (LY); drafting of the manuscript (LY); critical revision of the manuscript (BZ); and approval of final manuscript (RL-G).
Financial/Material Support: This research was supported by the National Basic Research Program of China (No. 2012CB518200), National Natural Science Foundation of China (No. 31571231).
Disclosures: None.
