Abstract
Objective
Symptoms consistent with neurological decompression sickness (DCS) in commercial breath-hold (Ama) divers has been reported from a few districts of Japan. The aim of this study was to detect circulating intravascular bubbles after repetitive breath-hold diving in a local area where DCS has been reported in Ama divers.
Methods
The participants were 12 partially assisted (descent using weights) male Ama divers. The equipment (AQUALAB system) consisted of continuous-wave Doppler with a 5-MHz frequency, and the Doppler probe was placed in the precordial site with the ultrasonic wave directed into the pulmonary infundibulum. We carried out continuous monitoring for 10 minutes at the end of the series of repetitive dives, and the recordings were made on numerical tracks and graded in a blind manner by 2 experienced investigators, according to the Spencer Doppler code.
Results
Depths and number of dives were 8 to 20 m and 75 to 131 times. Mean diving duration and surface interval were 64 ± 12 seconds and 48 ± 8 seconds, respectively (mean ± SD). We detected the lowest grade of intravascular bubbles (Spencer’s grade I) in an Ama diver whose mean surface interval was only 35.2 ± 6.2 seconds. His mean descending, bottom, and ascending times were 10.4 ± 1.6 seconds, 39.2 ± 8 seconds, and 18.2 ± 3.0 seconds, respectively, over the course of 99 dives.
Conclusions
Intravascular bubbles may be formed after repetitive breath-hold dives with short surface intervals or after a long breath-holding session in Ama divers. Symptoms consistent with neurological accidents in repetitive breath-hold diving may be caused in part by the intravascular presence of bubbles, indicating the need for safety procedures.
Introduction
Records in breath-hold (BH) diving continue to be broken, with dives that are deeper and longer. Nevertheless, this rise in performance is not without risk. Independent reports from around the world suggest that repeated BH dives may result in symptoms consistent with decompression sickness (DCS), and an on-line search resulted in a total of 141 cases of these accidents in BH divers, 1 –3 despite the past assumption that such accidents do not occur in these divers. Although there is now objective evidence that such accidents occur in BH divers as well, the mechanisms are still unknown. Among BH divers, the Ama divers of Japan have been particularly difficult to study because access to these working divers is often limited.
Ama divers are professional fishermen who catch fish during BH dives, and they have 2 distinct types of practice. Some are “Cachido” divers who dive without assistance to between 3 m and 6 m, and others are “Funado” divers who dive assisted by a weight during the descent phase to between 15 m and 25 m. In the northern district of Yamaguchi, Japan, 40% of the Funado divers have experienced accidents similar to cerebral vascular diseases during or after their working day with symptoms that were numerous and various. 4 The most common symptoms were sensory numbness or motor weakness of the hand and foot on one side, speech disturbance, and dizziness after more than 3 hours of consecutive dives. Diving accidents in Funado divers are thus more likely than in Cachido divers according to Tamaki et al. 4 These accidents are similar or identical to the “Taravana” syndrome of Polynesia, suggesting that DCS is caused by a cumulative increase in the tissue and blood nitrogen tension (bubbles). 5 The estimation of DCS risk after repetitive shallow BH dives is therefore important to advance insight into human diving physiology. Moreover, to the best of our knowledge, no studies have yet assessed the impact of repetitive BH dives on the production of venous bubbles in Ama divers.
We hypothesized that 1) the nitrogen gas bubbles formed during a BH diving session could be imaged using a continuous-wave Doppler apparatus, 2) Funado Ama divers performing repetitive shallow BH dives may be at risk for gas bubble formation, and 3) the Funado diving pattern would increase the risk of DCS in BH divers. The aim of this study was therefore to assess the impact of an ordinary working day on the production of bubbles in the Ama of Japan. We also tried to characterize their diving pattern to better understand BH-related DCS and whether any risk factors predispose them to experiencing this condition.
Methods and Materials
Study Population
This study was performed in the province of Yamaguchi, Japan. Twelve Ama professionals agreed to participate in the study. The study protocol was approved by the human research ethics committee of Kurume University School of Medicine, Japan. Morphological parameters were taken before their day of fishing, at the same time in the morning and under resting conditions.
Procedures
First, weight, size, and percentage of fat mass estimated by skinfold thickness 6 were measured a few hours before departure. Second, monitoring was conducted with a continuous-wave Doppler apparatus (AQUALAB system) equipped with a 5-MHz probe. To improve the accuracy of the cardiac signal, the divers were placed in left lateral decubitus position, and they lay at rest for 1 minute before the beginning of detection. Because the Funado divers were in real diving conditions (they had equipped themselves, swum, and then climbed out of the sea onto the boat), they were probably affected by cold and immersion (diuresis, hemoconcentration, dehydration). Thus, their level of bubble release was assumed to reflect the actual occurrences in Funado divers.1,3 The probe was placed in the precordial site, with the ultrasonic wave being directed into the pulmonary infundibulum. Thirty minutes before the working day and thus the dives, the 12 Funado divers were monitored by Doppler for at least 1 minute to establish a reference. After their working day and their final exit from the water, we again sought to detect bubbles with the same equipment and the same experimenter, one time continuously for 10 minutes. This time was chosen because fast tissue would be the first to be saturated in BH divers, as suggested by Wong. 3
The bubbles were graded in a blind manner by 2 experienced investigators according to the Spencer Doppler scale 7 : grade 0, no bubble signal; grade I, an occasional bubble signal with the great majority of the cardiac cycles free of signals; grade II, many but less than half of the cardiac cycles containing bubble signals; grade III, bubbles in most of the cardiac cycles but not obscuring the heart sounds; and grade IV, numerous bubbles obscuring the heart sounds. If any discrepancy in the interpretation of the signals occurred, the recordings were studied again until consensus was reached. To avoid modifying the Funado divers’ habits (diving depth and time, the interval of surface recovery), no particular instructions were given to them. The divers were also equipped with a Suunto D4 (Suunto Oy, Vantaa, Finland) diving computer to record their diving parameters. They used their personal diving equipment, including fins, mask, and a 20-kg weight that was connected to a windlass on the boat. The diving profile was analyzed using Suunto Dive Manager 3 (Suunto Oy) software.
Statistics
The results are presented as means and SD. Diving parameters were compared using a Wilcoxon matched-pairs signed-rank test. Pearson correlations were also performed. A probability value of less than 0.05 was considered significant. Analyses were performed with Statview software (Abacus Concepts, Inc, Berkeley, CA; 1992).
Results
The anthropometric parameters of the Ama divers are presented in Table 1. According to their diving histories, 4 of the 12 Ama divers had experienced symptoms consistent with neurological incidents before this experiment (10 years earlier); these included sensory numbness, motor weakness, and speech disturbance during or after 3 to 4 hours of repetitive BH dives. Two Ama divers had a medical history of disease (diabetes).
Anthropometric parameters of the Ama divers
The diving pattern is presented in Table 2. The diving histories of the Cachido and Funado divers were 29.8 ± 7.9 years and 26.2 ± 10.5 years, respectively. All Ama accomplished their habitual work in the morning (in 2.5 to 3.75 hours), and they continued their diving work for 1 to 2 hours after a 30-minute lunch break. More than half of the Ama divers started their profession as Cachido divers and changed to Funado diving with a weight for descent. The Ama divers carried a weight belt of 5.0 ± 1.7 kg to maintain neutral buoyancy, and all were partially assisted BH divers who descended passively using a weight of 20 ± 1.5 kg and then swam to the surface. A total of 1241 dives were analyzed for the 12 divers. Ama dives were made between 8 m and 20 m for BH times of 42 seconds to 2 minutes for 94 ± 16 dives. The ascent rate was 0.8 ± 0.2 m/s and the descent rate was 1.4 ± 0.3 m/s.
Diving pattern of the Funado divers analyzed from 1241 dives
BH, breath-hold; SI/dive, ratio between surface interval and diving duration.
P < .05.
P < .01.
The search for venous bubbles allowed us to identify 1 case of grade I bubbles during the cardiac cycle in an Ama diver, according to the Spencer code. 8 These bubbles were identified after 2 minutes of detection for about 10 seconds. However, this diver had never reported an experience of neurological deficit during or after diving over the course of a 17-year career in Funado Ama diving. Nevertheless, he had the deepest (20.6 m for 1 minute 18 seconds) and also the longest dive (2 minutes 26 seconds at 18.9 m) of the Funado diver-subjects. His mean maximum depth (15.8 ± 1.8 m vs 12.5 ± 2.8 m; P < .05) and his mean dive duration were also greater (68 ± 9 seconds vs 64 ± 12 seconds; P < .05). This particular diver also had the shortest mean surface interval of all the other divers (35 ± 6 seconds vs 48 ± 7 seconds; P < .05) for 48% of his BH dives and a total of 99 BH dives (Table 2). His ascent rate was 0.8 ± 0.3 m/s, and the descent rate was 1.5 ± 0.2 m/s.
The mean maximal depth reached during the diving session was positively correlated with the mean BH duration, the bottom BH duration, and also the ascent duration of the dives (P < .001; Figure 1). The surface interval was positively correlated with the mean BH duration of the dives (P < .001; Figure 2). No correlation was found between the anthropometric parameters and other parameters. The individual diving pattern of the BH diver with the grade I score is also presented in Figure 3.
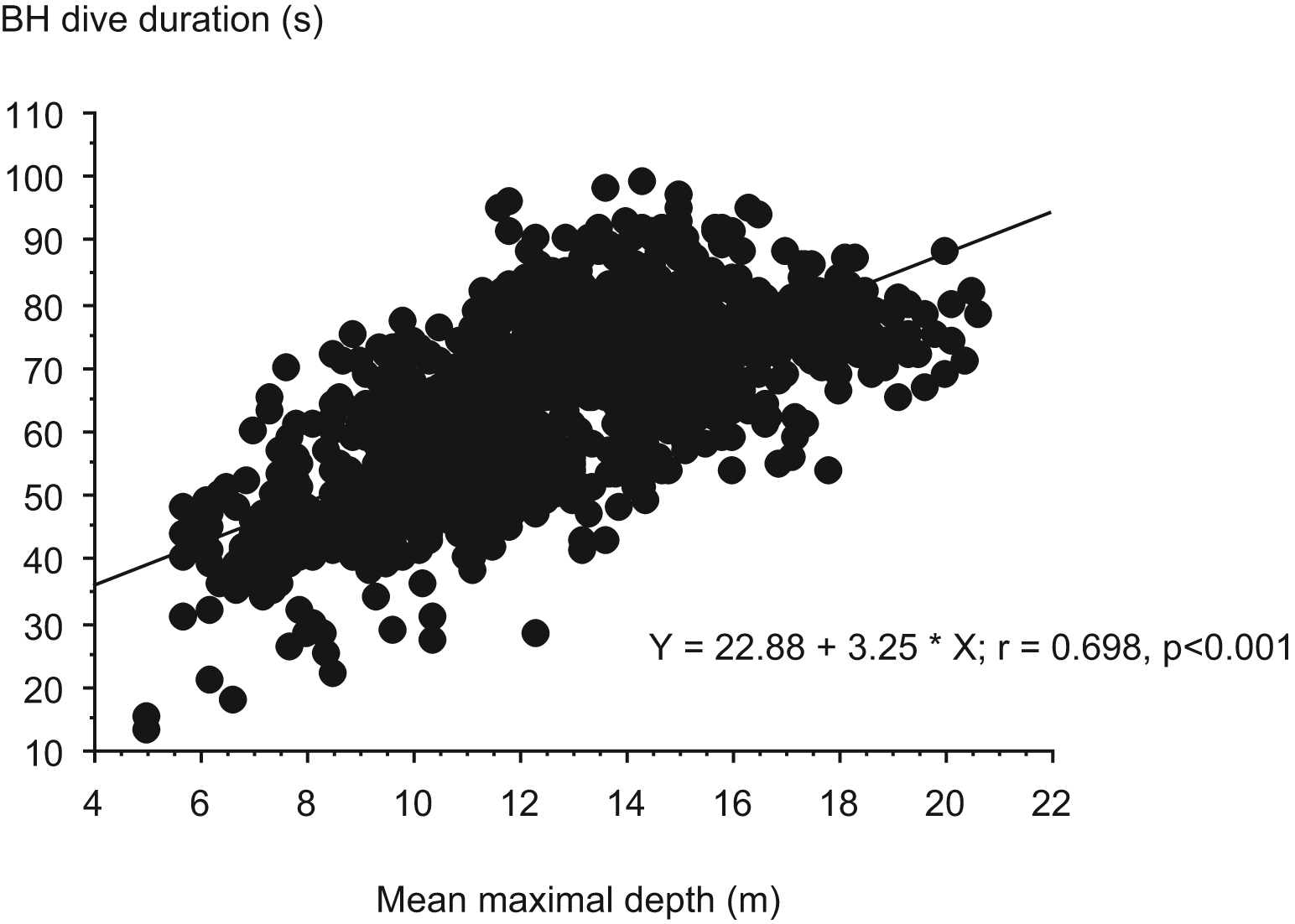
Correlation of mean maximal depth reached during the diving session with the mean breath-hold (BH) duration.
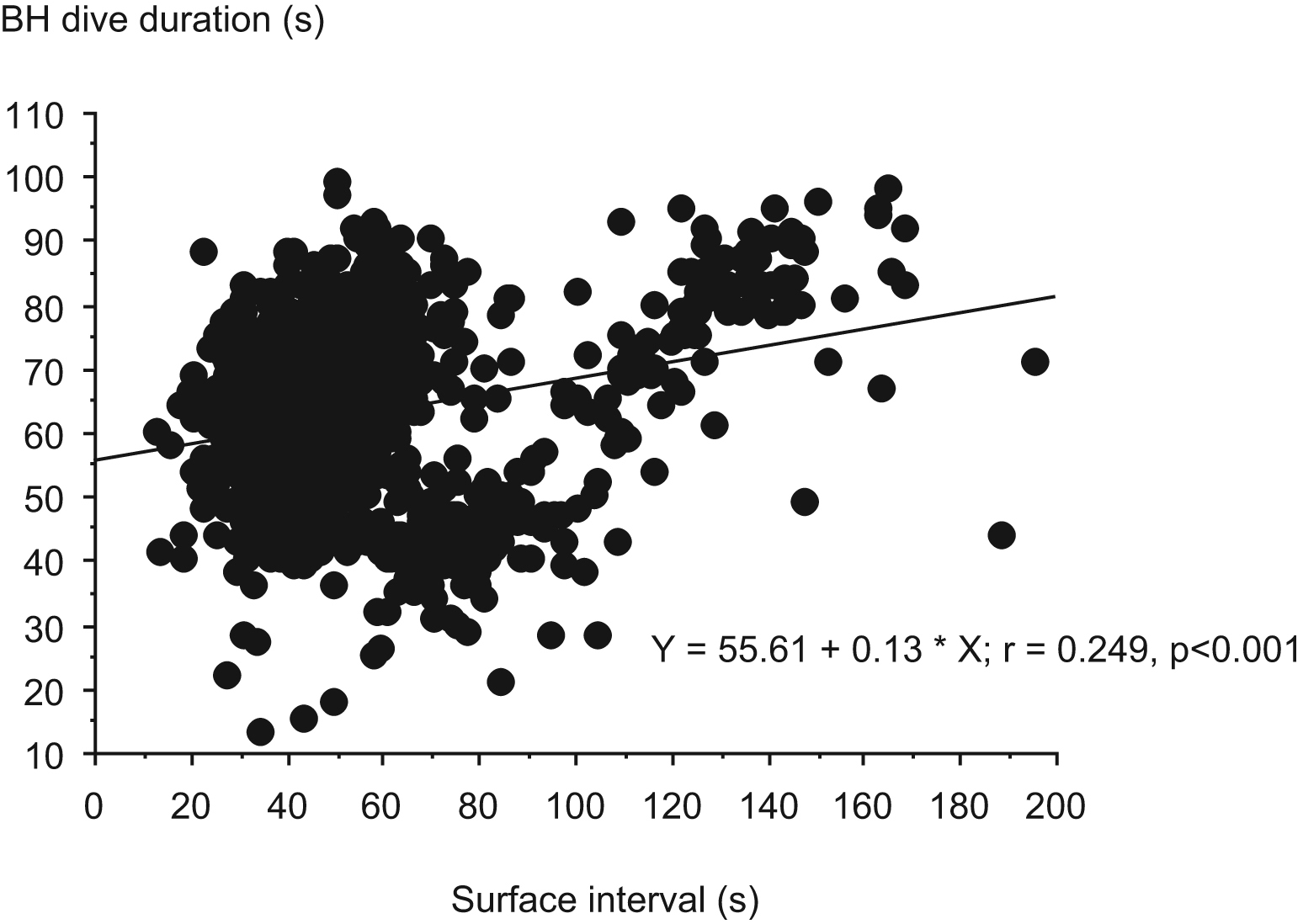
Correlation of surface interval with the mean breath-hold (BH) duration of the dives.
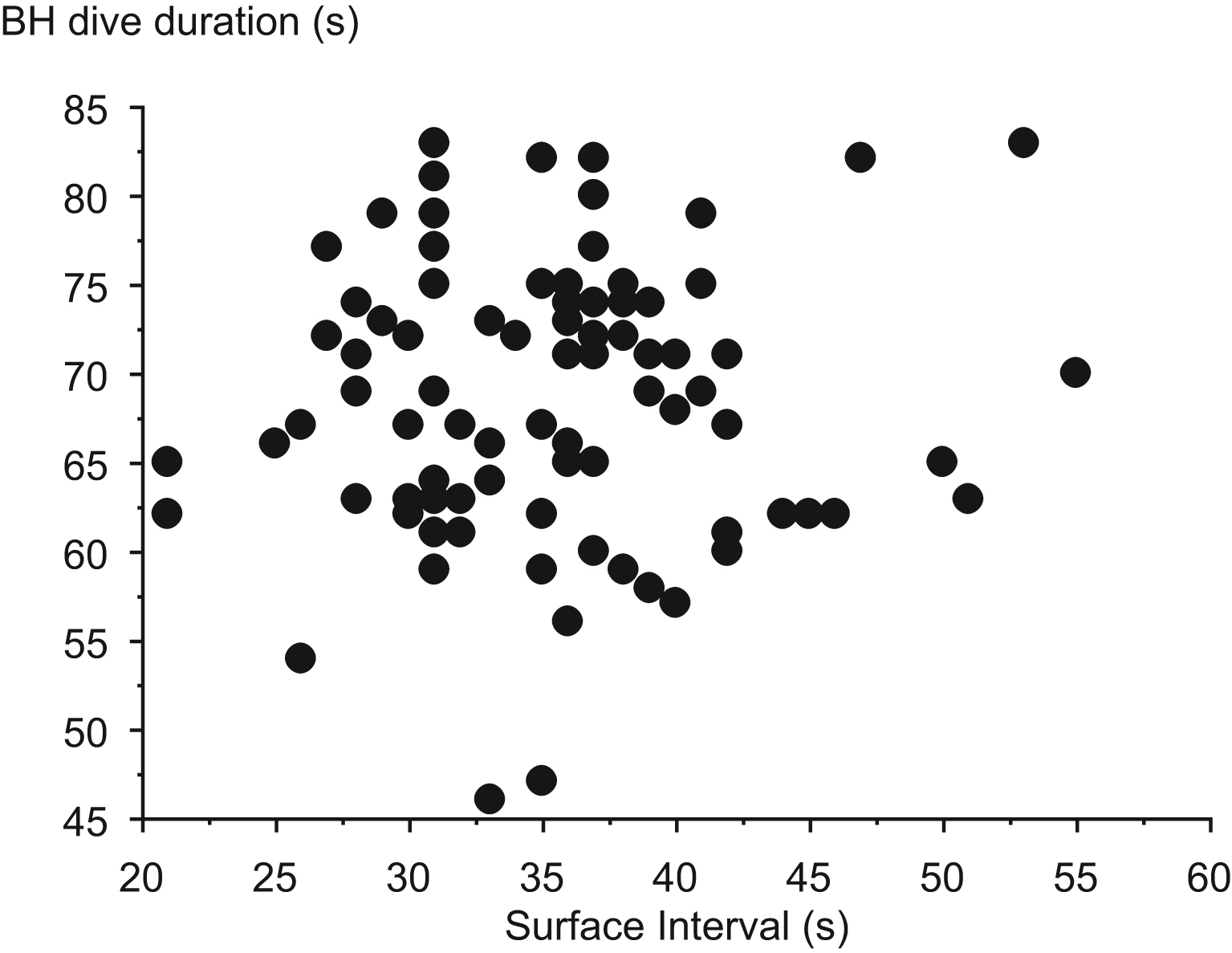
Individual diving pattern of the breath-hold (BH) diver with the grade I bubble score.
Discussion
The main result of the present study was the finding of the lowest possible bubble grade only once in a single Ama diver, after a series of 99 repetitive BH dives.
In this district, the male Funado Ama divers were partially assisted and continued diving work for 3 to 4 hours. 4 Although their diving patterns were quite similar to those of the completely assisted female Ama divers on Hegura Island, Japan, whose mean maximum diving depth and time were 16.8 m and 58.4 seconds, respectively, no DCS among Hegura Ama divers has been reported in medical records or by rumor. 1 Despite partial assistance, the male Ama divers in the present study had a longer bottom time (39.2 ± 7.8 seconds) compared with that of the female Hegura Ama divers (23.6 ± 6.4 seconds), but this duration was not different in the Ama diver with the grade I bubbles. However, this Ama diver did have the greatest mean depth and the lowest surface interval to dive ratio, indicating a particular exposure pattern. A long bottom time in BH diving may be a major risk factor of DCS, in addition to the depth and total dive time. Moreover, repeated BH diving was thought to cause neurological DCS in pearl divers. The pearl divers made 40 to 60 dives/d to depths of 30 m or more, spent an average of 2 min/dive underwater, and remained at the surface for 3 to 4 minutes between dives. Many of them experienced Taravana diving syndrome, the symptoms and signs of which include vertigo, nausea, partial or complete paralysis, unconsciousness, and even death. Motor paralysis was the most common neurological sign, but it was transient and disappeared completely. Although the Japanese Ama divers did not dive deeper than 20 m, repetitive BH diving for more than 3 hours with short surface intervals can cause stroke-like symptoms consistent with neurological DCS. The Ama diver with the grade I intravascular bubbles in our study had shorter surface intervals and deeper dives than the other BH divers for 48% of his dives. In theory, this higher exposure may have elevated the saturation of fast tissues (i.e., tissues that have a fast dissolution of gas bubbles) in this diver, and the short surface interval may have reduced the restoration of tissue equilibrium. It is notable that despite shorter dives of less depth and fewer repetitions than in our study, a case was reported of a 21-year-old man showing neurological signs of DCS. 1 Other parameters such as ascent and descent rates may also play a role in the risk of DCS. In our grade I BH diver, the descent phase was rapid (1.5 vs 1.4 m/s) and to a greater depth, indicating a possibly higher risk than to the other BH divers.
The potential mechanisms of cerebral involvement after repetitive BH diving are still unclear. The marked response to recompression treatment cannot be explained readily on any other basis than the presence of bubbles in the brain. What could cause the cerebral events in BH divers? In situ bubble formation as a result of progressive inert gas loading during dives is possible. Theoretical calculations indicate that significant nitrogen loading can indeed occur if enough repetitive BH dives are performed with short surface intervals.1,2 Nevertheless, regarding nitrogen kinetics and the relatively extensive cerebral perfusion, the development of autochthonous bubbles in the brain is difficult to explain. Nitrogen gas bubbles probably form in the intravascular and extravascular spaces of systemic organs after multiple repetitive BH dives. Air compression in the lungs during BH diving increases the partial pressure of nitrogen in the alveoli, inducing nitrogen to be taken up by the blood. Because of a smaller gradient in nitrogen tension from tissues to blood during the ascent, nitrogen is not released into the alveoli as rapidly as it is taken up by the blood and tissues during descent. Nitrogen accumulation in venous blood has been described in unassisted Ama divers3,4; hence, multiple deep BH dives with short intervals can theoretically cause DCS.
The radiological findings in Ama divers with neurological DCS show multiple cerebral infarcts in the areas corresponding to the symptoms and elicited signs. The brain lesions are localized in the basal ganglia, internal capsule, brainstem, and deep and subcortical white matter. 4 The types of brain lesions in Ama divers are identical to those caused by compressed air diving. Ischemic lesions in the basal ganglia are situated in the terminal zone, and lesions involving deep or superficial white matter correspond to border zone or watershed regions. These are the so-called low-flow cerebral infarctions as the result of low perfusion pressure in terminal supply areas, and these lesions are considered to be an arterial gas embolism of the brain.
An unresolved question is why the lesions in BH divers mainly involve the brain. Several possible mechanisms for DCS have been identified in BH divers. The passage of venous bubbles through the lungs would be possible during dives because “trapped” bubbles are compressed at the bottom under increasing pulmonary arterial pressure caused by the accumulation or lodging of bubbles. 4 When Ama divers descend, the trapped bubbles would be compressed and thus able to pass through the pulmonary capillaries.1,2 Arterialized bubbles expand during each ascent and gather in the terminal supply areas of the brain, border zones, and watershed regions. On the surface or during ascent, these arterialized microbubbles could expand as a result of decompression and cause a cerebral gas embolism. Multiple and large cerebral infarcts are probably formed by the repetition of this phenomenon. According to this hypothesis, all venous bubbles could be arterialized and then result in cerebral arterial gas embolism.1,2 Although this is our hypothesis of the neurological DCS in Ama divers, the exact mechanisms of this condition are in dispute.
Conclusions
Intravascular bubbles were detected after a long series of repetitive BH dives (grade I) but only in one Ama diver without any symptom of accident. The pattern of diving and especially the short surface intervals may play a particular role in the development of these bubbles, increasing the DCS risk. The mechanism underlying neurological accidents in BH divers and their relationship to bubble formation, if they exist, remains to be elucidated.
Footnotes
Acknowledgments
We are grateful to the physicians of Tamaki Hospital for their assistance in this study. We are also indebted to the Ama divers, who so enthusiastically took part in the study. We also thank Cathy Carmeni for help in preparing the manuscript. This project was supported by our laboratory (CETAPS EA 3832), the COMEX S.A., and the “Imersion Society.”
