To the Editor:
We present the case of a 58-year-old woman who presented to a major emergency department in Canberra, Australia, with an 8-hour history of acute-onset diarrhea and vomiting. She had arrived to Australia from China 1 day previously. On presentation she was afebrile and normotensive with a normal clinical examination. Her initial hematology and biochemistry results were unremarkable (Table), as was her electrocardiogram.
Selected biochemistry results at presentation and at 96 hours in intensive care unit
ALT = alanine transaminase; APPT = activated partial thromboplastin time; AST = aspartate transaminase; GGT = γ-glutamyltransferase; HCO3 = bicarbonate; INR = international normalized ratio; PT = prothrombin time.
The patient confessed that she had ingested 6 mushrooms from her daughter's front garden at 1 o'clock the previous afternoon. She had boiled the mushrooms before eating them, and no one else at home had consumed them. At 10 o'clock that evening she developed acute onset of watery diarrhea and nonbilious vomiting. Her daughter was asked to return home and bring back a sample of the mushrooms she had consumed. Subsequent consultation with a local mycologist confirmed the mushroom was Amanita phalloides (Figure).
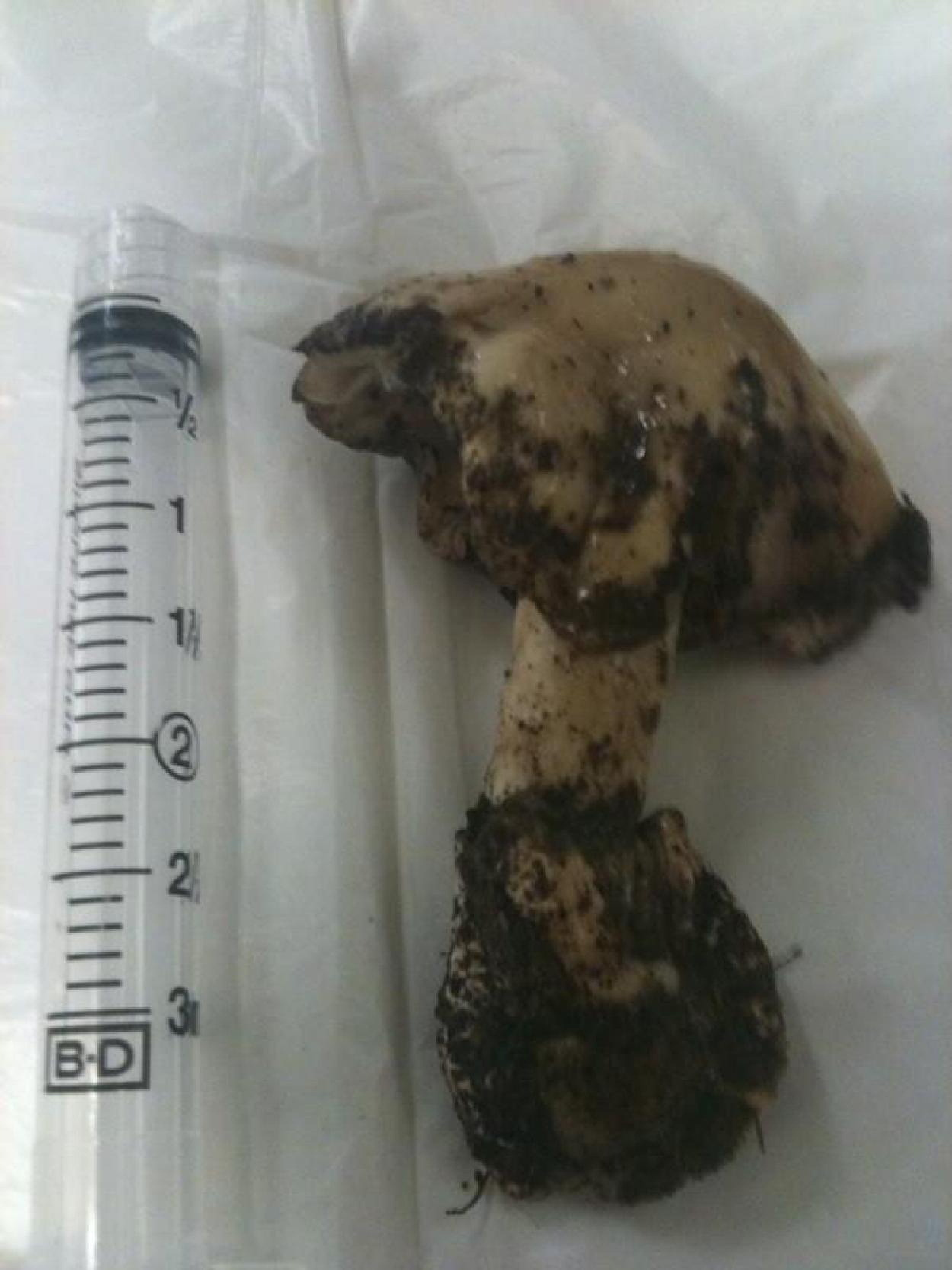
Amanita phalloides ingested by our patient. Note the green-white color and the pronounced volva.
Treatment was initiated with silibinin 5 mg/kg intravenously 4 times a day, as well as high-dose benzylpenicillin (1,000,000 IU · kg–1 · day–1) and N-acetylcysteine as per the Australian guidelines for hepatoprotection in paracetamol overdose. She was aggressively resuscitated with fluids to replace her gastrointestinal losses.
Forty-eight hours after admission the patient developed decompensated coagulopathic liver failure (Table) and became acutely encephalopathic. She also required renal replacement therapy. She was transferred to the intensive care unit at the Royal Prince Alfred Hospital in Sydney, a quaternary critical care unit with both liver and renal transplantation units on site. She died of fulminant liver failure 5 days after ingestion of the mushrooms.
A phalloides, more commonly known as death cap mushrooms, are native to Europe. 1 They have a symbiotic relationship with certain trees. The species was incidentally introduced to temperate regions of Australia by European settlers with the cultivation of nonnative species of oak, chestnut, and pine trees. A similar spread has occurred in North America. 1
Death caps are highly toxic. The lethal dose is debated, but may be as small as 0.1 mg/kg (approximately one mushroom). They resemble several edible species of mushroom, including Caesar's mushroom and the straw mushrooms. They have a white-green to white cap, free white gills, and a volva (cuplike structure) at the base of the stem (Figure). Identification by a non-mycologist is extremely unreliable.
The primary toxin potentiating A phalloides poisoning is amatoxin, a multicyclic peptide. 2 A secondary agent named phallotoxin is present in the mushrooms and is highly hepatotoxic, but it is poorly absorbed through the gastrointestinal wall and probably contributes little to toxicity. Amatoxin inhibits RNA polymerase 2, and the result is catastrophic cessation of cell metabolism, with subsequent nuclear and cytoplasmic disruption, cell death, and tissue necrosis. Primarily, the liver is the affected organ because of extensive first-pass metabolism. Fundamentally there is a large similarity between amatoxin-mediated free radical damage and the pathophysiology of paracetamol (acetaminophen) induced free radical hepatic necrosis. Amanita phalloides poisoning is a multistage process. 2 An initial latent period of 8 to 12 hours is followed by a gastrointestinal phase characterized by watery diarrhea. A high index of suspicion must be maintained as gastrointestinal decontamination during this phase may be an important determinant of survival. A second latent phase follows for 12 to 24 hours. Although the patient may be asymptomatic during this phase, biochemical investigations may reveal a transaminitis and coagulopathy. Finally, in the delayed phase of poisoning, fulminant hepatic failure develops, with progressive encephalopathy, coma, convulsions, and cardiovascular collapse. The hepatorenal syndrome may also develop, requiring dialysis. Unless a liver transplant is available, death usually occurs 7 to 10 days after ingestion. The mortality rate even with modern treatment may be as high as 40%. 3
Management of phalloides syndrome centers around decontamination, reduction of toxic load, and trying to prevent initial uptake and hepatic injury. 3 Meticulous supportive care is also indicated. There is no high-quality evidence base for any particular treatment. 2 Activated charcoal, gastric lavage, and whole bowel irrigation are strongly indicated for acute ingestion, and there may be a role for decontamination of delayed presentations. 4 Amatoxin circulates in the serum of poisoned patients for up to 30 hours after ingestion. 4 Repeated administration of activated charcoal may be of benefit up to 3 days after poisoning. Other methods of detoxification such as forced diuresis have also been advocated. 4 During the initial gastrointestinal phase, volume losses may be large, and attention must be paid to fluid and electrolyte balance. 4
Early administration of silibinin,2,3 N-acetylcysteine, 2 and high-dose benzylpenicillin2,5 have been associated with increased survival in death cap poisoning. Penicillin displaces amatoxin from albumin, and decreases its uptake by hepatocytes. 5 It is administered as doses of 1 000 000 U · kg–1 · day–1 (note that 3 g of intravenous penicillin G is approximately 5 000 000 U). 5 Silibinin, an extract from the blessed milk thistle (Silybum marianum), inhibits amatoxin penetration of liver cell walls by competing with amatoxin for bile salt transport systems. 3 It is administered in 4 equal doses and is calculated at 20 mg · kg–1 · day–1, although this is a consensus rather than evidence-based dose. 2 Both benzylpenicillin and silibinin should be given to patients with confirmed or suspected poisoning as soon as possible, preferably in the gastrointestinal phase of the illness. Treatment should continue based on the clinical condition of the patient, usually until transaminase levels normalize. N-acetylcysteine is thought to provide nuclear protection via glutathione-mediated protection from free radical toxic intermediates. 2 Cimetidine has also been suggested as an antidote because it modifies cytochrome p450 metabolism, which may decrease formation of toxic amatoxin metabolites. 2
Even with early and optimal treatment, meticulous supportive care, and renal replacement therapy, survival remains uncertain. Treatment failure and progression of the syndrome will mandate liver transplantation or death.
A phalloides has a long and sordid history; the fungus is highly toxic and easily accidentally ingested. For the modern physician, the harms associated with administering silibinin, N-acetylcysteine and penicillin are all minimal, especially when compared with the high probability of death from amatoxic hepatic failure. When A phalloides toxicity is suspected, easily available therapies may be the most efficacious: decontamination, the use of easily available drugs (penicillin and N-acetylcysteine), and rapid transport to a tertiary center for specialist toxicological management.
Above all, when hungry in the wilderness, we would suggest that it is wise to be very careful what one eats.
