Abstract
High altitude pulmonary edema is a life-threatening condition that remains a concern for climbers and clinicians alike. Within the last decade, studies have shown ultrasonography to be valuable in the accurate diagnosis of a variety of lung pathologies, including cardiogenic pulmonary edema, pleural effusion, pneumothorax, and lung consolidation. Recently, studies conducted in remote areas have demonstrated that ultrasound lung comets can be used as a measure of subacute pulmonary edema and high altitude pulmonary edema in climbers ascending to altitude. This clinical review article provides an overview of lung ultrasonography and its relevance as a diagnostic aid to respiratory pathology. In addition, we describe a standardized technique for identifying ultrasound lung comets and its utility in recognizing the presence of extravascular lung water, as well as the results of studies that have used this approach at sea level and high altitude.
Introduction
High altitude pulmonary edema (HAPE) is a life-threatening condition that remains a concern for climbers and clinicians alike. HAPE can affect between 0.2% and 15% of persons who ascend to between 2500 m and 5000 m and is the most common cause of death from high altitude sickness.1,2 HAPE is associated with increased pulmonary artery pressures and the breakdown of alveolar capillary membranes, resulting in the accumulation of extravascular lung water (EVLW). 3
Historically, the diagnosis of HAPE has been based on the results of a well-taken history and an appropriate physical examination. Although this approach has often proved successful, it has the potential to overlook other respiratory conditions such as pneumonia, pneumothorax, and pulmonary effusion that may require alternative treatments. In this article, we describe the role ultrasonography (US) can play in the wilderness environment in differentiating among these lung pathologies.
Lung Ultrasonography
Ultrasonography has been used as an imaging technique for more than 50 years. However, until recently, the role of US in the diagnosis and management of respiratory diseases was limited. That limitation was primarily due to the presence of air in the respiratory tract and the solid structures of the thoracic cage that impeded the passage of US waves and created considerable artifact. However, it is exactly one of these artifacts, caused by the air-fluid interface in the presence of extra-alveolar fluid, that has now been shown to be an accurate sign in detecting the presence of EVLW. This artifact is called an ultrasound lung comet (ULC), B-line, or “comet tail” artifact (Figures 1 and 2). 4
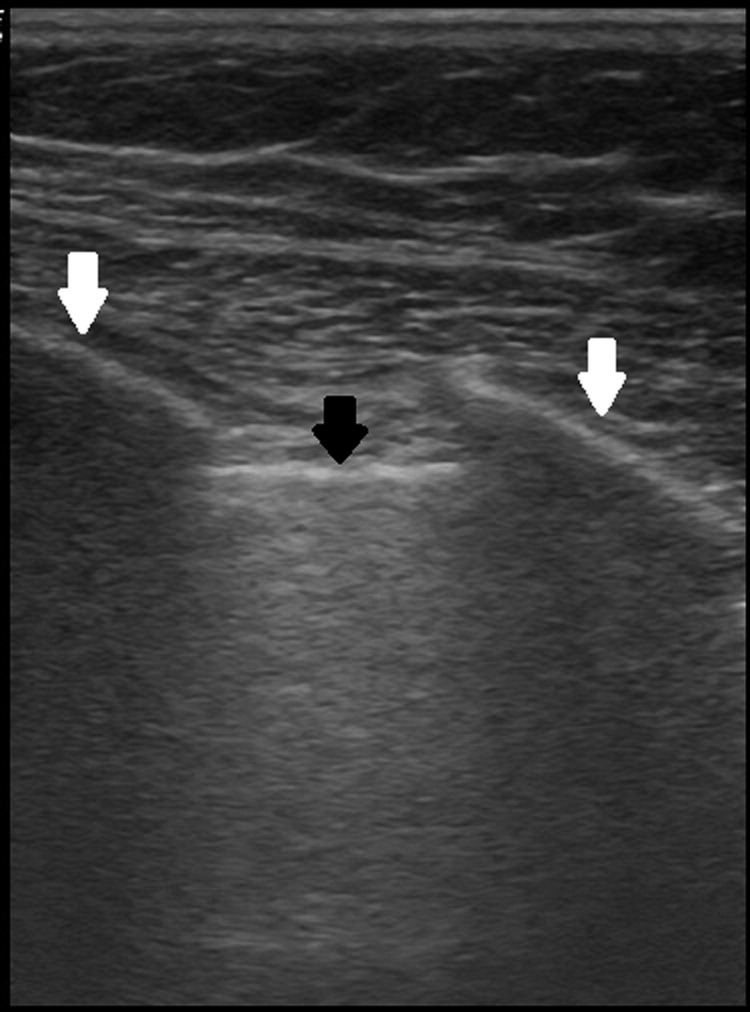
A normal lung ultrasonogram of a single intercostal space with no ultrasound lung comets. The black arrow indicates the pleural line, and the white arrows indicate the ribs as these form the “bat wing sign.”
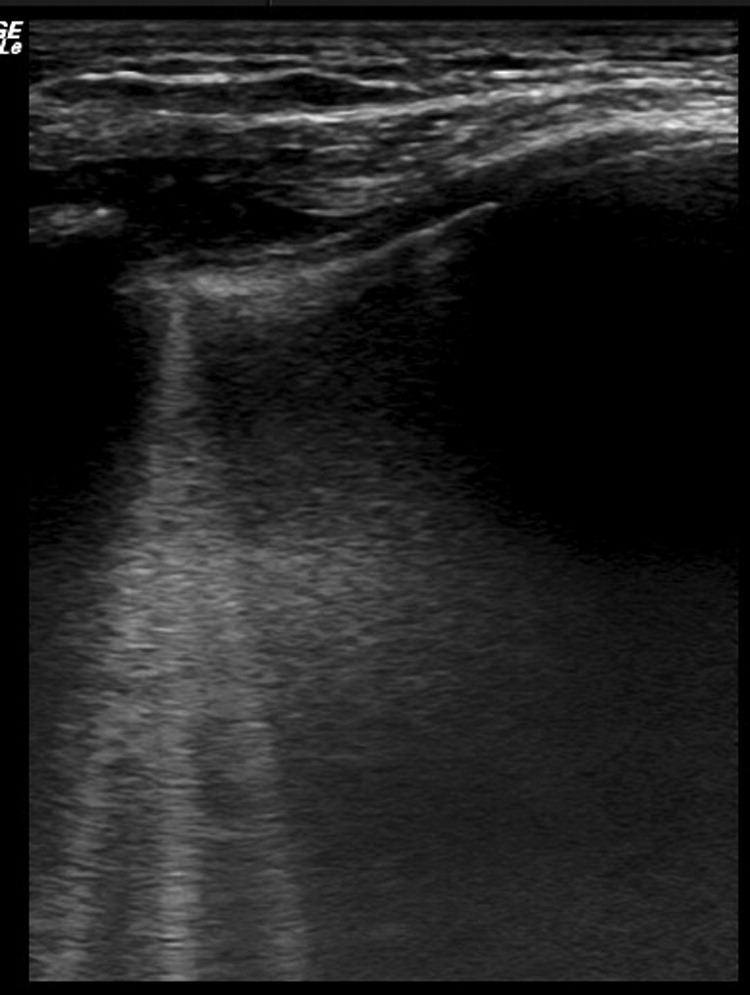
A lung ultrasonography scan of a single intercostal space showing multiple ultrasound lung comets (white vertical lines).
Within the last decade, studies have shown US to be an accurate adjunct in the diagnosis of a variety of lung pathologies including cardiogenic pulmonary edema (CPE), pleural effusion, pneumothorax, and lung consolidation. 5 –7 More recently, ULCs have been used to quantify EVLW to aid in the diagnosis of HAPE.8,9
Lung US in Clinical Practice
Toward the end of the 1980s, it had become clear that identifying the cause of acute respiratory compromise could not be done by clinical examination alone. 10 –12 Since portable chest radiography results were often found to be misleading, 12 –15 other modalities were sought. The ULC was first described as a diagnostic sign for pulmonary edema by the French intensivist Daniel Lichtenstein. His first study in 1997 demonstrated that lung US could be used to diagnose the presence of EVLW, with high levels of specificity and sensitivity. 16 In a further study, the same investigator was also able to show that the use of lung US was effective in differentiating between CPE and chronic obstructive pulmonary disease. 17
Subsequent studies carried out by other researchers validated the ULC as an accurate sign for detecting pulmonary edema by comparing it to results obtained from chest radiogram, computed tomography scan, and invasive measurements of EVLW. 14 ,18–20,26 In 2007, a study of 340 patients by Frassi et al 21 demonstrated that ULCs were significantly related to the severity of heart failure measured by the New York Heart Association (NYHA) functional classification I to IV. Successful treatment resulted in falls in both NYHA class and ULC score, thereby demonstrating that ULCs could be used as a tool for monitoring changes in EVLW. 21 Further work by Lichtenstein et al 7 in 2008 demonstrated that lung US alone can diagnose the etiology of acute respiratory failure in 90.5% of cases. Recently, this was confirmed by Xirouchaki et al, 22 who compared the diagnostic performance of lung US to bedside chest radiography for the detection of various lung pathologies. Using computed tomography as the gold standard, chest radiography was shown to have a sensitivity of 46% and a specificity of 80% in diagnosing pulmonary edema, whereas lung US was shown to have a sensitivity of 94% and specificity of 93%. 22 A summary of hospital based studies that have utilized ULCs in the detecation of EVLW is shown in Table 1.
Summary of hospital-based clinical studies on the use of lung ultrasonography in diagnosing cardiogenic pulmonary edema
US, ultrasonography; NA, not available; ICD, implanted cardioverter-defibrillator.
Pulse contour continuous cardiac output (PiCCO [Pulsion Medical Systems, Phillips, Munich, Germany]) is an invasive measure of extravascular lung water using transpulmonary thermodilution and arterial pulse contour analysis.
N-Terminal pro–B-type natriuretic peptide (NT-proBNP) is the atrial natriuretic peptide used as a measure of congestive cardiac failure.
Final clinical diagnosis—these trials used a composite gold standard of final clinical diagnosis reached by conducting chest roentgenography (CXR), computed tomography (CT), and other investigations.
Lung US in Hape
Ultrasound lung comets were first identified at high altitude in a study of HAPE victims conducted at the Himalayan Rescue Association clinic in Pheriche, Nepal (4240 m). 8 Lung US performed on 11 patients diagnosed with HAPE and 7 healthy control subjects revealed that patients with HAPE had a significantly higher ULC score (mean 31 vs 11) and lower oxygen saturations (61% vs 87%). 8 In patients with HAPE, mean ULC score fell (35 vs 11) and oxygen saturations increased (60% vs 84%) after treatment. An inverse correlation was shown to exist between ULC score and oxygen saturation. For every 1-point increase in ULC score, oxygen saturation fell by 0.67% (95% CI:, 0.41% to 0.93%, P < .001). 8
In 2010, Pratali et al 9 conducted a study in Nepal on 18 healthy Italian trekkers ascending to an altitude of 5130 m. Lung US was conducted at sea level and at various points during the ascent. Their results showed that ULC appeared in 15 of 18 subjects (83%) at 3440 m and in 18 of 18 subjects (100%) at 4790 m. Using the 28-zone examination, ULC increased during the ascent, from 1.06 ± 1.3 at 1350 m to 16.5 ± 8 at 5130 m (P < .001) and decreased on descending to 1355 m (2.9 ± 1.7). Of the 18 subjects, 3 climbers were diagnosed with HAPE, 2 at 4790 m and 1 at 5130 m. All 3 exhibited a much higher number of ULCs at 5130 m (64, 30, and 38, respectively) compared with others in the study group (mean of 16.5). With treatment, their ULC scores fell to 16, 17, and 14, respectively. 9
These 2 studies conducted at high altitude have shown that ULCs are common in healthy lowlanders who ascend to altitude and that the number of ULCs has an inverse relationship with oxygen saturation. In patients diagnosed with HAPE, ULCs have been shown to be significantly greater than in healthy controls, and resolve with treatment. However, it is still not known what number of ULCs is diagnostic for HAPE as opposed to the presence of subclinical pulmonary edema.
Performing Lung US
The probe most widely used in the current literature is the microconvex phased array low frequency (2 to 5 MHz) probe. 16 ,18,27–29 Other studies have utilized higher frequency linear transducers (5 to 10 MHz) 30 –32 or cardiac phased arrays (2 to 4 MHz).33,34 However, in the experience of the authors, lung US can be performed with most US probes.
Lung US is conducted with the patient in a supine or 45-degree position. Ultrasonography gel should be applied to each intercostal space that will be examined. The transducer is set at a depth of 4 cm to 10 cm, and the lungs are visualized through the intercostal spaces. To help identify the intercostal space, the probe should be oriented longitudinally. The “pleural line,” which represents the parietal and visceral pleural interface, can typically be seen 1.5 cm below and between 2 rib shadows. Together, the upper rib, pleural line, and lower rib form a characteristic pattern, the “bat wing sign” (Figure 1). Once the pleura and underlying lung are identified, the probe should be turned to the transverse position to visualize a larger pleural area. The number of lung zones scanned can vary from a comprehensive 28-zone examination to an abbreviated 8-zone study. For out-of-hospital clinical use, the 8-zone examination is often preferred as it has been shown to be effective in detecting EVLW and avoids the need for undressing. 18 In the 8-zone technique, 2 anterior and 2 lateral intercostal spaces are scanned on each hemithorax.
The initial examination is normally conducted using the default 2-dimensional B mode; however, if a pneumothorax or pleural effusion is suspected, a M-mode (time-motion) study is needed to look for further signs commonly seen in these 2 conditions (Table 2). 35 The number of ULCs seen per area scanned is noted, and a total ULC score recorded. The presence of 3 or more ULCs per intercostal space is evidence of EVLW, and if seen diffusely in more than 1 intercostal space, it is indicative of pulmonary edema. 16 In hospital trials conducted on the efficacy of lung US in the diagnosis of CPE, lung US was shown to take less than 3 minutes to perform and produce interpretable images in nearly 100% of cases. 16 ,17,19,20 Table 2 and Figures 2 through 6 provide a summary of US signs that may be seen when performing a lung US examination and explain their significance.
Summary of lung ultrasonography signs and their significance
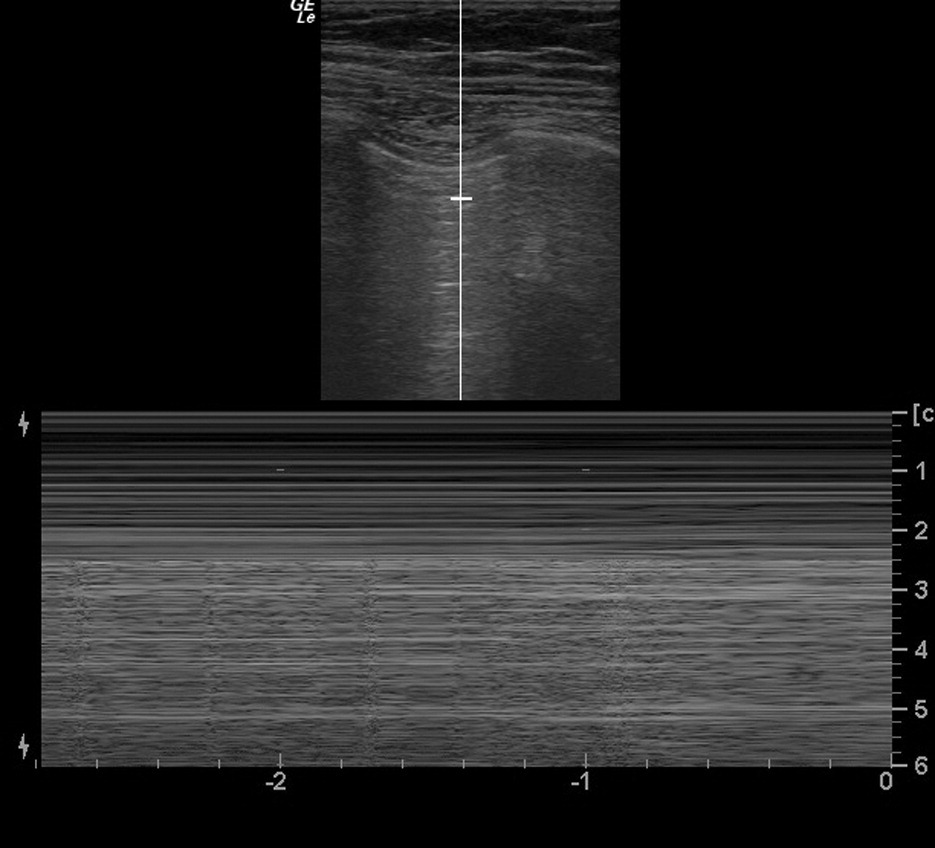
Ultrasonography B and M mode scan of normal lung.
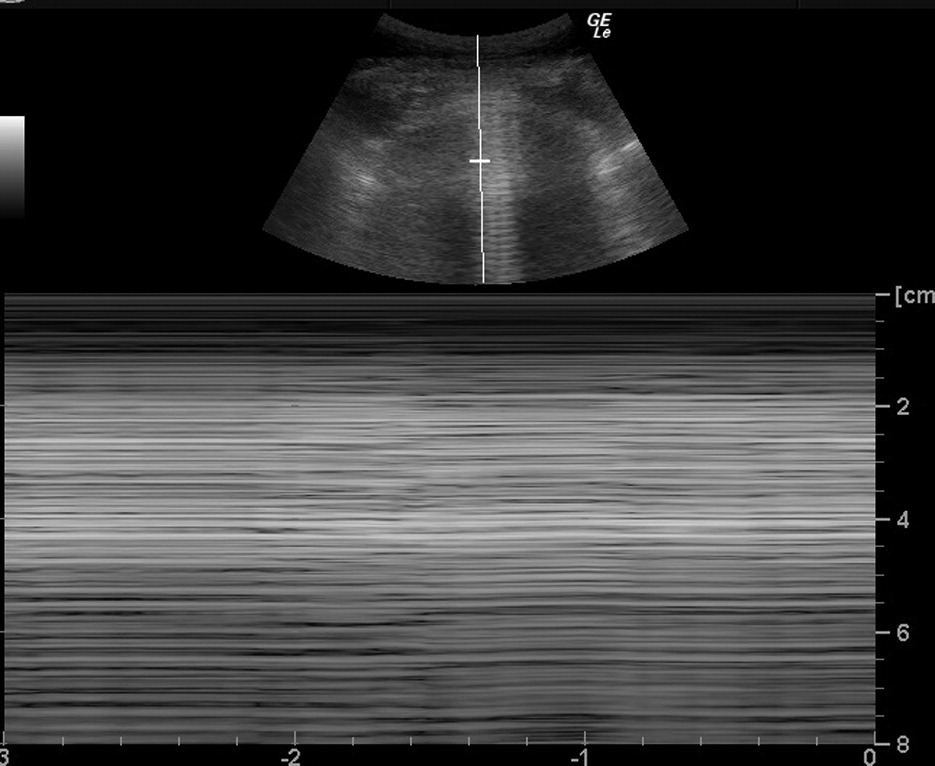
Ultrasonography B and M mode depiction of a pneumothorax.
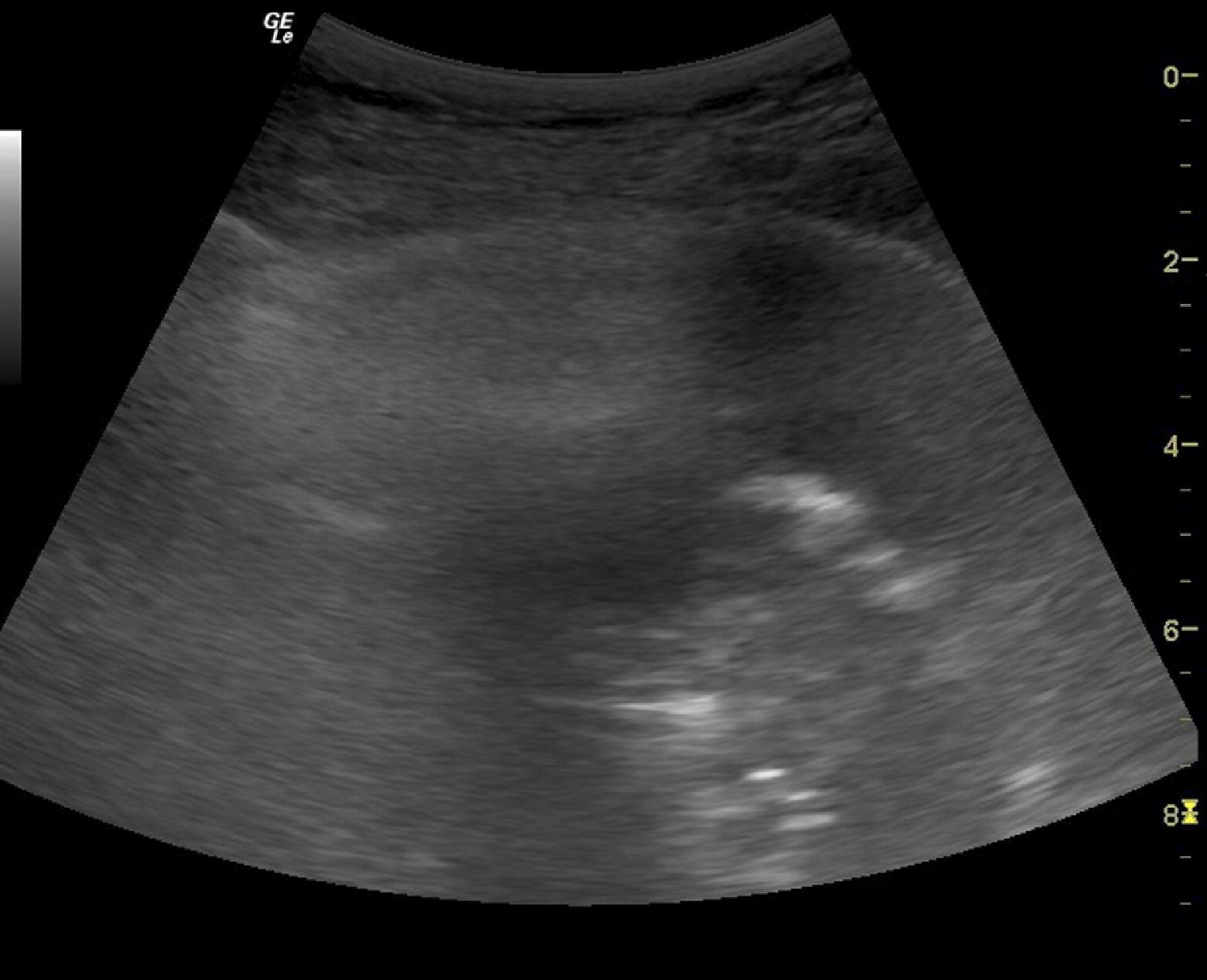
Lung ultrasonography scan showing lung consolidation.
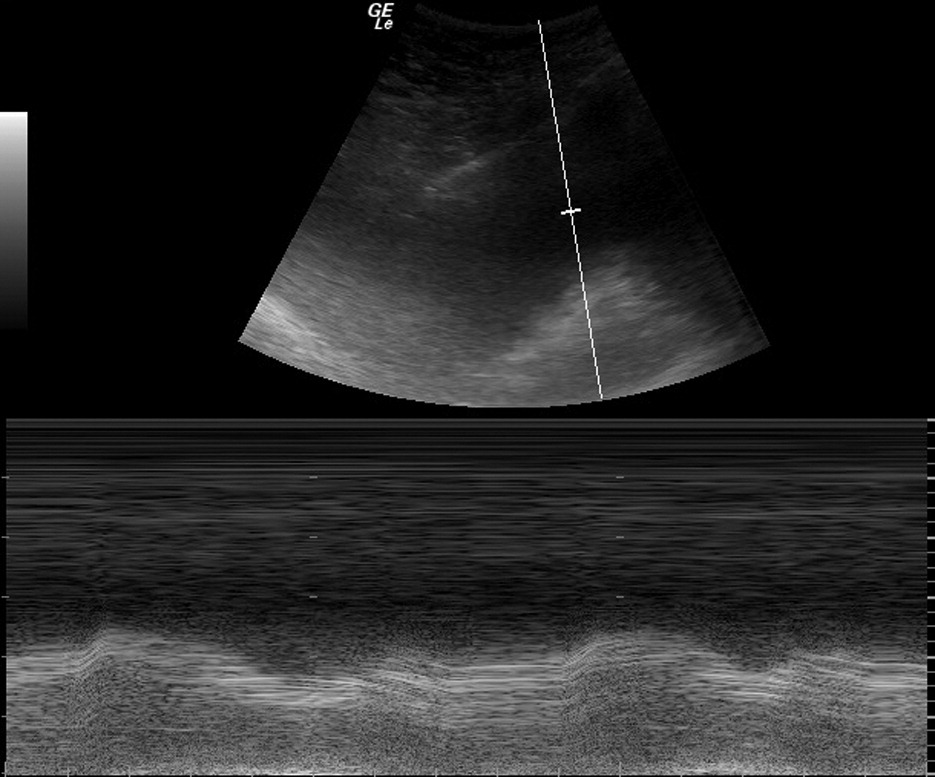
Ultrasonography B and M mode view of pleural effusion.
Advantages of Using Lung US as Diagnostic Tool in the Remote Environment
The major advantage of ultrasonography as a diagnostic aid in remote areas is its portability. Modern laptop-sized ultrasound machines are more than sufficient for performing accurate lung scans. Even with batteries, power source, image storage (USB or laptop), and ultrasound gel, the total weight should not exceed 15 kg, which therefore permits it to be carried by a single person. 36 Other advantages include that it is nonionizing, repeatable, and immediate, providing the clinician with images in real time. Lung US can also be performed within a few minutes, and hence reduces the time of patient exposure, which is especially important in cold mountainous environments.
Limitations of Using Lung US for Diagnosing Hape
As shown by studies done by Pratali et al9,37 in 2011 and 2012, subclinical pulmonary edema is now an accepted phenomenon with altitude gain. Their 2011 Khumbu study showed that all climbers demonstrated subclinical edema at 4750 m but only 3 were diagnosed with HAPE. This presence of subclinical pulmonary edema was further shown in a study conducted in 2012 where it was associated with exercise limitation in patients with chronic mountain sickness. These studies have demonstrated that ULCs are common at altitude in the absence of other classic features of HAPE, leading to questions about the clinical relevance of this sign at altitude. However, it should be noted that the climbers diagnosed with HAPE in both the Pratali and Fagenholz studies showed significantly higher numbers of ULCs compared with the climbers who were asymptomatic.8,9 Despite this, it is as yet unclear exactly what quantity of ULCs are diagnostic of HAPE; therefore, more studies and more experience are required to understand the pathophysiology of subclinical pulmonary edema developing into HAPE and to determine whether ULCs can aid in detecting this pathological conversion.
Other limitations of lung US lie in the areas of training, operator variability, and reliability. Although most studies showed lung ultrasonography to have low intraobserver and interobserver variability, the majority of these scans were performed by clinicians with considerable experience in sonography. Whether these results can be replicated in remote environments remains to be seen. Most high altitude remote areas are cold and have no electricity. In this type of environment, ultrasound machine batteries deplete rapidly, and lack of provisions for charging can be a major limitation.
Conclusions
With an increasing number of people trekking to high altitudes around the world, it is becoming increasingly important to accurately diagnose and treat pathologies associated with altitude exposure. Although no imaging technology or monitor can replace the value of good history-taking and clinical examination, lung US has been showed to be both sensitive and specific in the diagnosis of a wide range of pulmonary pathologies.
